Experimental acute arsenic toxicity in Balb/c mice: organic markers and splenic involvement
Abstract
Introduction: Arsenic is an environmental toxic present worldwide. In men and animals, various organs and tissues are targets of its deleterious effects including those of the immune system.
Objective: To determine acute arsenic toxicity in tissues and target cells of Balb/c mice using an in vivo methodology.
Materials and methods: We injected Balb/c mice intraperitoneally with 9.5 or 19 mg/kg of sodium arsenite (NaAsO2), or an equivalent volume of physiological solution as a control (with 3 per experimental group). After 30 minutes, the animals were sacrificed to obtain spleen, thymus, liver, kidneys, and blood. We determined arsenic, polyphenols, and iron concentrations in each sample and we evaluated the oxidative markers (peroxides, advanced products of protein oxidation, and free sulfhydryl groups). In splenocytes from the spleen, cell viability and mitochondrial potential were also determined.
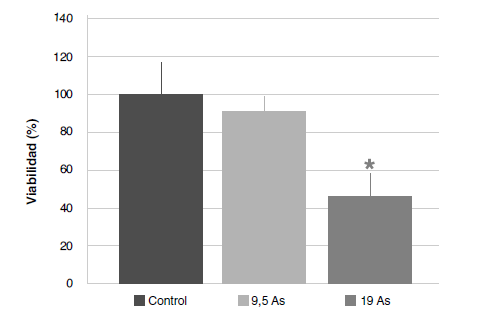
Results: The exposure to an acute dose of NaAsO2 reduced the mitochondrial function of splenocytes, which resulted in cell death. Simultaneously, the confirmed presence of arsenic in spleen samples and the resulting cytotoxicity occurred with a decrease in polyphenols, free sulfhydryl groups, and an alteration in the content and distribution of iron, but did not increase the production of peroxides.
Conclusion: These findings provide scientific evidence about changes occurring in biomarkers involved in the immunotoxicity of arsenic and offer a methodology for testing possible treatments against the deleterious action of this compound on the immune system.
Downloads
References
Grinyó JM, Cruzado JM, Bestard O, Vidal-Castañeira JR, Torras J. Immunosuppression in the era of biological agents. Adv Exp Med Biol. 2012;741:60-72. https://doi.org/10.1007/978-1-4614-2098-9_5
Ferrario D, Gribaldo L, Hartung T. Arsenic eExposure and immunotoxicity: A review including the possible influence of age and sex. Curr Environ Health Rep. 2016;3:1-12. https://doi.org/10.1007/s40572-016-0082-3
Cordero H, Morcillo P, Martínez S, Meseguer J, Pérez-Sirvent C, Chaves-Pozo E, et al. Inorganic arsenic causes apoptosis cell death and immunotoxicity on European sea bass (Dicentrarchus labrax). Mar Pollut Bull. 2018;128:324-32. https://doi.org/10.1016/j.marpolbul.2018.01.052
Hughes MF, Beck BD, Chen Y. Arsenic exposure and toxicology: A historical perspective. Toxicol Sci. 2011;123:305-32. https://doi.org/10.1093/toxsci/kfr184
Acharya S, Chaudhuri S, Chatterjee S, Kumar P, Begum Z, Dasgupta S, et al. Immunological profile of arsenic toxicity: A hint towards arsenic-induced carcinogenesis. Asian Pac J Cancer Prev. 2010;11:479-90.
Ng JC. Environmental contamination of arsenic and its toxicological impact on humans. Environ Chem. 2005;2:146-60. https://doi.org/10.1071/EN05062
Miranda AR, Soria EA. Análisis proteasomal y multidimensional en quimioencefalopatía aguda inducida por arsénico trivalente en ratones y neuroprotección por ácido clorogénico. En: INC. Becas de investigación en cáncer 2015-2016: Informes finales de estudiantes avanzados y graduados. Primera edición. Ciudad Autónoma de Buenos Aires: Instituto Nacional del Cáncer; 2017. p. 73-84.
Pérez RD, Falchini GE, Cometto-Vincente F, Soares L, Poletti ME, Sánchez HJ. A new spectrometer using a crystal monochromator and parallel plates beam guides. Nuc Istr Meth. 2019;440:48-53. https://doi.org/10.1016/j.nimb.2018.11.030
Chen JL, Steele TWJ, Stuckey DC. Metabolic reduction of resazurin; location within the cell for cytotoxicity assays. Biotechnol Bioeng. 2018;115:351-8. https://doi.org/10.1002/bit.26475
Ait Baddi G, Cegarra J, Merlina G, Revel JC, Hafidi M. Qualitative and quantitative evolution of polyphenolic compounds during composting of an olive-mill waste-wheat straw mixture. J Hazard Mater. 2009;165:1119-23. https://doi.org/10.1016/j.jhazmat.2008.10.102
Pieroni G, Khalil L, Charlotte F, Poynard T, Piton A, Hainque B, et al. Comparison of bathophenanthroline sulfonate and ferene as chromogens in colorimetric measurement of low hepatic iron concentration. Clin Chem. 2001;47:2059-61.
Soria EA, Goleniowski ME, Cantero JJ, Bongiovanni GA. Antioxidant activity of different extracts of Argentinian medicinal plants against arsenic-induced toxicity in renal cells. Hum Exp Toxicol. 2008;27:341-6. https://doi.org/10.1177/0960327108092192
Piwowar A. Advanced oxidation protein products. Part I. Mechanism of the formation, characteristics and property. Pol Merkur Lekarski. 2010;28:166-9.
InfoStat. InfoStat versión 2018. Córdoba: Universidad Nacional de Córdoba; 2018.
Rubatto-Birri PN, Pérez RD, Cremonezzi D, Pérez CA, Rubio M, Bongiovanni GA. Association between As an Cu renal cortex accumulation and physiological and histological alterations after chronic arsenic intake. Environ Res. 2010;110:417-23. https://doi.org/10.1016/j.envres.2009.09.002
Soria EA, Pérez RD, Queralt I, Pérez CA, Bongiovanni GA. Immunotoxicological effects of arsenic bioaccumulation on spatial metallomics and cellular enzyme response in the spleen of male Wistar rats after oral intake. Toxicol Lett. 2017;266:65-73. https://doi.org/10.1016/j.toxlet.2016.12.014
Patra PH, Bandyopadhyay S, Bandyopadhyay MC, Mandal TK. Immunotoxic and genotoxic potential of arsenic and its chemical species in goats. Toxicol Int. 2013;20:6-10. https://doi.org/10.4103/0971-6580.111533
D´Archivio M, Filesi C, Varí R, Scazzocchio B, Masella R. Bioavailability of the polyphenols: Status and controversias. Int J Mol Sci. 2010;11:1321-42. https://doi.org/10.3390/ijms11041321
Mahmud H, Fӧller M, Lang F. Arsenic-induced suicidal erythrocyte death. Arch Toxicol. 2009;83:107-13. https://doi.org/10.1007/s00204-008-0338-2
Wang L, Xu ZR, Jia XY, Han XY. Effects of dietary arsenic levels on serum parameters and trace mineral retentions in growing and finishing pigs. Biol Trace Elem. 2006;113:155-64. https://doi.org/10.1385/BTER:113:2:155
Cui X, Okayasu R. Arsenic accumulation, elimination and interaction with copper, zinc and manganese in liver and kidney of rats. Food Chem Toxicol. 2008;46:3646-50. https://doi.org/10.1016/j.fct.2008.09.040
Montelescaut E, Vermeersch V, Commandeur D, Huynh S, Danguy des Deserts M, Sapin J, et al. Acute arsenic poisoning. Ann Biol Clin (Paris). 2014;72:735-8. https://doi.org/10.1684/abc.2014.1012
Watanabe T, Hirano S. Metabolism of arsenic and its toxicological relevance. Arch Toxicol. 2013;87:969-79. https://doi.org/10.1007/s00204-012-0904-5
Xu H, Medina S, Lauer FT, Douillet C, Liu KJ, Stýblo M, et al. Genotoxicity induced by monomethylarsonous acid (MMA+3) in mouse thymic developing T cells. Toxicol Lett. 2017;279:60-6. https://doi.org/10.1016/j.toxlet.2017.07.897
Xu H, McClain S, Medina S, Lauer FT, Douillet C, Liu KJ, et al. Differential sensitivities of bone marrow, spleen and thymus to genotoxicity induced by environmentally relevant concentrations of arsenite. Toxicol Lett. 2016;262:55-61. https://doi.org/10.1016/j.toxlet.2016.09.008
Zhou X, Sun X, Mobarak C, Gandolfi AJ, Burchiel SW, Hudson LG, et al. Differential binding of monomethylarsonous acid compared to arsenite and arsenic trioxide with zinc finger peptides and proteins. Chem Res Toxicol. 2014;27:690-8. https://doi.org/10.1021/tx500022j
Pelicano H, Feng, L, Zhou Y, Carew JS, Hileman EO, Plunkett W, et al. Inhibition of mitochondrial respiration: A novel strategy to enhance drug-induced apoptosis in human leukemia cells by a reactive oxygen species-mediated mechanism. J Biol Chem. 2003;278:37832-9. https://doi.org/10.1074/jbc.M301546200
Belzacq AS, El Hamel C, Vieira HL, Cohen I, Haouzi D, Metivier D, et al. Adenine nucleotide translocator mediates the mitochondrial membrane permeabilization induced by lonidamine, arsenite and CD437. Oncogene. 2001;20:7579-87. https://doi.org/10.1038/sj.onc.1204953
Skulachev VP. Bioenergetic aspects of apoptosis, necrosis and mitoptosis. Apoptosis. 2006;11:473-85. https://doi.org/10.1007/s10495-006-5881-9
Moe B, Peng H, Lu X, Chen B, Chen L, Gabos S, et al. Comparative cytotoxicity of fourteen trivalent and pentavalent arsenic species determined using real-time cell sensing. J Environ Sci (China). 2016;49:113-24. https://doi.org/10.1016/j.jes.2016.10.004
Rahman MT, De Ley M. Arsenic induction of metallothionein and metallothionein induction against arsenic cytotoxicity. Rev Environ Contam Toxicol. 2017;240:151-68. https://doi.org/10.1007/398_2016_2
Some similar items:
- Juliana Buitrago, Sandra Viviana Santa, Acute intermittent porphyria presenting as spontaneous hemothorax , Biomedica: Vol. 29 No. 3 (2009)
- Marcela Varona, Gloria Lucía Henao, Sonia Díaz, Angélica Lancheros, Álix Murcia, Nelcy Rodríguez, Víctor Hugo Álvarez, Effects of aerial applications of the herbicide,glyphosate and insecticides on human health , Biomedica: Vol. 29 No. 3 (2009)
- Gerzaín Rodríguez, Rafael Pinto, Fernando López, Yenny Gómez, Persistent type 2 lepra reaction and clofazimine-induced lethal enteropaty , Biomedica: Vol. 29 No. 1 (2009)
- Ana María Perilla, Camilo González, Sandra Liliana Valderrama, Natasha Vanegas, Bibiana Chavarro, Luis Carlos Triana, José Roberto Támara, Carlos Arturo Álvarez, Necrotizing pneumonia by community-acquired, methicillin-resistant Staphylococcus aureus in Colombia , Biomedica: Vol. 29 No. 4 (2009)
- Liliana Alejandra Chicaíza-Becerra, Mario García-Molina, Óscar Gamboa, Cost-effectiveness of iso- versus low-osmolality contrast media in outpatients with high risk of contrast medium-induced nephropathy , Biomedica: Vol. 32 No. 2 (2012)
- Andrés F. Londoño, Silvana Levis, Juan D. Rodas, Hantavirus as important emerging agents in South America , Biomedica: Vol. 31 No. 3 (2011)
- Alvaro Javier Idrovo, Possible health effects associated with Pre-Columbian metallurgy. , Biomedica: Vol. 25 No. 3 (2005)
- Marcela E. Varona, Jorge E. Tolosa, Omayda Cárdenas, Carlos H. Torres, Darío Pardo, Gabriel Carrasquilla, Howard Frumkin, Pesticides use by flower companies associated with the Colombian Association of Flower Exporters. , Biomedica: Vol. 25 No. 3 (2005)
- Jeison Monroy-Gómez, Orlando Torres-Fernández, Calbindin and parvalbumin distribution in spinal cord of normal and rabies-infected mice , Biomedica: Vol. 33 No. 4 (2013)
- Jefferson Antonio Buendia, Andrés F. Zuluaga, Physicians' insight about adverse drug reaction to frequently used medication groups in Bogotá (Colombia) , Biomedica: Vol. 34 No. 3 (2014)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























