Detection of Trypanosoma cruzi strains circulating in Córdoba department (Colombia) isolated from triatomines (Hemiptera: Reduviidae) collected by the community
Abstract
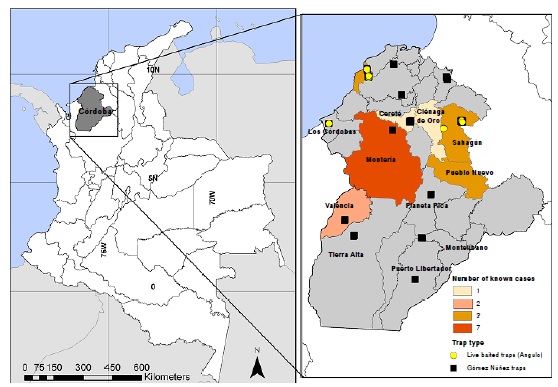
Introduction: From 2011 to 2016, 24 cases of Chagas disease were reported in Córdoba according to the national public health surveillance system (Sistema Nacional de Vigilancia en Salud Pública, Sivigila), but the information regarding Trypanosoma cruzi circulating strains and infection rates are unknown.
Objectives: To establish the triatomine species with which people come in contact and recognize as Chagas disease vectors, as well as to assess the infection with trypanosomes and make an exploratory approach to host feeding preferences with the participation of the local community.
Materials and methods: Triatomines sampling was conducted in 12 municipalities between 2011 and 2016; T. cruzi infection was established by k-PCR, SAT-PCR, while strain genotyping was done by mini-exon and SL-IR (spliced-leader intergenic region) sequence characterization. We also screened for blood sources.
Results: Local community members collected the majority of triatomines and we identified three species: Rhodnius pallescens, Panstrongylus geniculatus, and Eratyrus cuspidatus. The overall T. cruzi infection rate in collected triatomines was 66.6% and we detected the TcIDOM and TcI sylvatic strains. Community-based insect collection allowed reporting the presence of P. geniculatus in two new disperse rural settlements, T. cruzi infection of P. geniculatus in Córdoba, and the first report of triatomines infected with T. cruzi in Montería municipality.
Conclusions: These results revealed the presence of triatomines infected with T. cruzi inside dwellings in five municipalities of Córdoba. The dominant circulating T. cruzi strain was TcIDOM, a genotype associated with human Chagas disease and cardiomyopathies in Colombia. Our results highlight the importance of local community participation in entomological surveillance tasks.
Downloads
References
Chagas C. Nova tripanozomiaze humana: estudos sobre a morfolojia e o ciclo evolutivo do Schizotrypanum cruzi n. gen., n. sp., ajente etiolojico de nova entidade morbida do homem. Mem Inst Oswaldo Cruz. 1909;1:159-18. https://doi.org/10.1590/S0074-02761909000200008
Teixeira A, Nitz N, Guimaro M, Gomes C, Santos-Buch C. Chagas disease. Postgrad Med J. 2006;82:788-98. https://doi.org/10.1136/pgmj.2006.047357
Jannin J, Villa L. An overview of Chagas disease treatment. Mem Inst Oswaldo Cruz. 2007;102 (Supl. 1):95-7. https://doi.org/10.1590/S0074-02762007005000106
Zingales B, Andrade SG, Briones MR, Campbell DA, Chiari E, Fernandes O, et al. A new consensus for Trypanosoma cruzi intraspecific nomenclature: Second revision meeting recommends TcI to TcVI. Mem Inst Oswaldo Cruz. 2009;104:1051-4. https://doi.org/10.1590/S0074-02762009000700021
Zingales B, Miles M, Campbell D, Tibayrenc M, Macedo A, Teixeira M, et al. The revised Trypanosoma cruzi subspecific nomenclature: Rationale, epidemiological relevance and research applications. Infect Genet Evol. 2012;12:240-53. https://doi.org/10.1016/j.meegid.2011.12.009
Añez N, Crisante G, da Silva F, Rojas A, Carrasco H, Umezawa E, et al. Predominance of lineage I among Trypanosoma cruzi isolates from Venezuelan patients with different clinical profiles of acute Chagas disease. Trop Med Int Health. 2004;9:1319-26. https://doi.org/10.1111/j.1365-3156.2004.01333.x
Guhl F, Ramírez JD. Trypanosoma cruzi I diversity: Towards the need of genetic subdivision? Acta Trop. 2011;119:1-4. https://doi.org/10.1016/j.actatropica.2011.04.002
Guhl F, Aguilera G, Pinto N, Vergara D. Actualización de la distribución geográfica y ecoepidemiología de la fauna de triatominos (Reduviidae: Triatominae) en Colombia. Biomédica. 2007;27(Supl. 1):143-62. https://doi.org/10.7705/biomedica.v27i1.258
Rassi A Jr, Rassi A, Marcondes De Rezende J. American trypanosomiasis (Chagas disease). Infect Dis Clin North Am. 2012;26:275-91. https://doi.org/10.1016/j.idc.2012.03.002
Instituto Nacional de Salud. Enfermedad de Chagas. Fecha de consulta: 30 de enero de 2017. Disponible en: http://simposiovirologia.ins.gov.co/temas-de-interes/Paginas/chagas.aspx
Moncayo A, Silveira AC. Current epidemiological trends for Chagas disease in Latin America and future challenges in epidemiology, surveillance and health policy. Mem Inst Oswaldo Cruz. 2009; 104(supl. 1):17-30. https://doi.org/10.1590/S0074-02762009000900005
Zambrano P. Protocolo de vigilancia en salud pública: Chagas. Fecha de consulta: 14 de noviembre de 2016. Disponible en: http://simposiovirologia.ins.gov.co/lineas-de-accion/Subdireccion-Vigilancia/sivigila/Protocolos%20SIVIGILA/PRO%20Chagas.pdf
Montilla M, Soto H, Parra E, Torres M, Carrillo P, Lugo L, et al. Infestación por triatominos en comunidades indígenas de Valledupar, Colombia. Rev Saúde Pública. 2011;45:773-80. https://doi.org/10.1590/S0034-89102011005000037
Cantillo O, Garcés E, Gómez A, Cortés L, Pereira A, Marcet P, et al. Eco-epidemiological study of an endemic Chagas disease region in northern Colombia reveals the importance of Triatoma maculata (Hemiptera: Reduviidae), dogs and Didelphis marsupialis in Trypanosoma cruzi maintenance. Parasit Vectors. 2015;22:482.
https://doi.org/10.1186/s13071-015-1100-2
Instituto Nacional de Salud. Sistema Nacional de Vigilancia en Salud Pública-Sivigila. Fecha de consulta: 14 de noviembre del 2016. Disponible en: http://www.portalsivigila.ins.gov.co/sivigila/reportes/pages/vigilancia.php
Departamento Administrativo Nacional de Estadística (DANE). Atlas estadístico. Fecha de consulta: 16 de noviembre de 2016. Disponible en: http://www. http://sige.dane.gov.co/atlasestadistico/
Sociedad Geográfica de Colombia. Departamento de Córdoba. Fecha de consulta: 14 de noviembre de 2016. Disponible en: http://www.sogeocol.com.co
Freire EE, Perfetti del Corral M. Pobreza Monetaria 2015: Córdoba. Fecha de consulta: 15 de noviembre de 2016. Disponible en: http://www.dane.gov.co/index.php/estadisticas-por-tema/informacion-regional
Salomon OD. Aspectos de prevención de la enfermedad de Chagas con participación de la comunidad. Fecha de consulta: 14 de noviembre de 2016. Disponible en:
http://www.fac.org.ar/fec/chagas2/llave/md1/md103/salomon.htm
Dumonteil E, Ramírez-Sierra MJ, Ferral J, Euan-García M, Chávez-Núñez L. Usefulness of community participation for the fine temporal monitoring of house infestation by nondomiciliated triatomines. J Parasitol. 2009;95:469-71. https://doi.org/10.1645/GE-1712.1
Gómez-Núñez JC. Desarrollo de un nuevo método para evaluar la infestación intradomiciliaria por Rhodnius prolixus. Acta Cien Ven. 1965;16:26-31.
Angulo V, Esteban L. Nueva trampa para la captura de triatominos en hábitats silvestres y peridomésticos. Biomédica. 2011;31:264-68. https://doi.org/10.7705/biomedica.v31i2.301
Lent H, Wygodzinsky P. Revision of the triatominae (Hemiptera, Reduviidae), and their significance as vectors of Chagas’ disease. Bulletin of the American Museum of Natural History. New York: American Museum of Natural History; 1979. p. 389.
Weirauch C, Bérenger JM, Berniker L, Forero D, Forthman M, Frankenberg S, et al. An illustrated identification key to assassin bug subfamilies and tribes (Hemiptera: Reduviidae). Can J Art Iden. 2014;26:1-115.
Wincker P, Britto C, Borges J, Cardoso MA, Oelemann W, Morel CM. Use of a simplified polymerase chain reaction procedure to detect Trypanosoma cruzi in blood samples from chronic chagasic patients in a rural endemic area. Am J Trop Med. Hyg. 1994;51:771-7. https://doi.org/10.4269/ajtmh.1994.51.771
Virreira M, Torrico F, Truyens C, Alonso-Vega C, Solano M, Carlier Y, et al. Comparison of polymerase chain reaction methods for reliable and easy detection of congenital Trypanosoma cruzi infection. Am J Trop Med Hyg. 2003;68:574-82.
Piron M, Fisa R, Casamitjana N, López-Chejade P, Puig L, Vergés M, et al. Development of a real-time PCR assay for Trypanosoma cruzi detection in blood samples. Acta Trop. 2007;103:195-200. https://doi.org/10.1016/j.actatropica.2007.05.019
Souto RP, Fernandes O, Macedo AM, Campbell DA, Zingales B. DNA markers define two major phylogenetic lineages of Trypanosoma cruzi. Mol Biochem Parasitol. 1996;83:141-52. https://doi.org/10.1016/S0166-6851(96)02755-7
Villa LM, Guhl F, Zabala D, Ramírez JD, Urrea DA, Hernández DC, et al. The identification of two Trypanosoma cruzi I genotypes from domestic and sylvatic transmission cycles in Colombia based on a single polymerase chain reaction amplification of the spliced-leader intergenic region. Mem Inst Oswaldo Cruz. 2013;108:932-5. https://doi.org/10.1590/0074-0276130201
Falla A, Herrera C, Fajardo A, Montilla M, Vallejo GA, Guhl F. Haplotype identification within Trypanosoma cruzi I in Colombian isolates from several reservoirs, vectors and humans. Acta Trop. 2009;110:15-21. https://doi.org/10.1016/j.actatropica.2008.12.003
Sawabe K, Isawa H, Hoshino K, Sasaki T, Roychoudhury S, Higa Y, et al. Host-feeding habits of Culex pipiens and Aedes albopictus (Diptera: Culicidae) collected at the urban and suburban residential areas of Japan. J Med Entomol. 2010;47:442-50.
Rangel JO. Colombia Diversidad Biótica XI Patrones de la estructura y de la riqueza de la vegetación en Colombia. Bogotá: Instituto de Ciencias Naturales, Universidad Nacional; 2011. p. 485.
Zumaya-Estrada FA, Messenger LA, López-Ordóñez T, Lewis MD, Flores-López CA, Martínez-Ibarra AJ, et al. North American import? Charting the origins of an enigmatic Trypanosoma cruzi domestic genotype. Parasit Vectors. 2012;5:226. https://doi.org/10.1186/1756-3305-5-226
Duque MC, Ramírez JD, Rendón LM, Guhl F. Evaluación de la variabilidad genética de aislamientos colombianos de Trypanosoma cruzi mediante marcadores microsatélites. Infectio. 2011;15:227-34. https://doi.org/10.1016/S0123-9392(11)70736-6
Ramírez JD, Duque MC, Montilla M, Cucunubá Z, Guhl F. Natural and emergent Trypanosoma cruzi I genotypes revealed by mitochondrial (Cytb) and nuclear (SSU rDNA) genetic markers. Exp Parasitol. 2012;132:487-94. https://doi.org/10.1016/j.exppara.2012.09.017
León C, Hernández C, Montilla M, Ramírez J. Retrospective distribution of Trypanosoma cruzi I genotypes in Colombia. Mem Inst Oswaldo Cruz. 2015;110:387-93. https://doi.org/10.1590/0074-02760140402
Zeledón R, Rabinovich JE. Chagas’ disease: An ecological appraisal with special emphasis on its insect vectors. Annu Rev Entomol. 1981;26:101-33. https://doi.org/10.1146/annurev.en.26.010181.000533
Christensen HA, de Vásquez AM. Host feeding profiles of Rhodnius pallescens (Hemiptera: Reduviidae) in rural villages of Central Panamá. Am J Trop Med Hyg. 1981;30:278-83. https://doi.org/10.4269/ajtmh.1981.30.278
Some similar items:
- Víctor Manuel Angulo, Lyda Esteban, Plutarco Urbano, Eduwin Hincapié, Luis Alberto Núñez, Comparison of methods for the capture of triatomines (Hemiptera: Reduviidae) in Attalea butyracea palms in the Eastern Plains of Colombia , Biomedica: Vol. 33 No. 4 (2013)
- Luis Alberto Corté, Henry Alberto Suárez, Triatomines (Reduviidae: Triatominae) in a Chagas disease focus in Talaigua Nuevo (Bolívar, Colombia). , Biomedica: Vol. 25 No. 4 (2005)
- Luis C. Orozco, Diana Camargo, Myriam C. López, Sofía Duque, Luis E. Gualdrón, Elvia Cáceres, Margarita Ronderos, Maritza Rey, Augusto Corredor, Immunodiagnosis of Trypanosoma cruzi infection in humans using ELISA on blood collected on filter paper , Biomedica: Vol. 19 No. 2 (1999)
- Kevin Escandón-Vargas, Carlos A. Muñoz-Zuluaga, Liliana Salazar, Blood-feeding of Rhodnius prolixus , Biomedica: Vol. 37 No. 3 (2017)
- Víctor Manuel Angulo, Lyda Esteban, Katherine Paola Luna, Attalea butyracea palms adjacent to housing as a source of infestation by Rhodnius prolixus (Hemiptera: Reduviidae) , Biomedica: Vol. 32 No. 2 (2012)
- Andrea Arévalo, Julio César Carranza, Felipe Guhl, Jairo A. Clavijo, Gustavo Adolfo Vallejo, Comparison of feeding and defecation patterns of Rhodnius colombiensis and Rhodnius prolixus (Hemiptera, Reduviidae, Triatominae) under laboratory conditions. , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Gustavo Adolfo Vallejo, Felipe Guhl, Julio César Carranza, Omar Triana, Gerardo Pérez, Paola Andrea Ortiz, Dairo Humberto Marín, Lina Marcela Villa, Jazmín Suárez, Isaura Pilar Sánchez, Ximena Pulido, Ingrid Bibiana Rodríguez, Leyder Elena Lozano, Daniel Alfonso Urrea, Fredy Arvey Rivera, César Cuba-Cuba, Jairo Alfonso Clavijo, Trypanosoma rangeli parasite-vector-vertebrate interactions and their relationship to the systematics and epidemiology of American trypanosomiasis , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Juan Fernando Ríos, Margarita Arboleda, Alba Nelly Montoya, Erika Patricia Alarcón, Gabriel Jaime Parra-Henao, Probable outbreak of oral transmission of Chagas disease in Turbo, Antioquia , Biomedica: Vol. 31 No. 2 (2011)
- Andrea Arévalo, Julio César Carranza, Felipe Guhl, Gustavo Adolfo Vallejo, Electrophoretic patterns of salivary hemeproteins (nitrophorines) of Rhodnius colombiensis and R. prolixus (Hemiptera, Reduviidae, Triatominae) , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Maritza Alarcón , Cesare Colasante, Sonia Araujo , Reinaldo Gutiérrez -Marín, Dalmiro Cazorla-Perfetti , Claudia Magaly Sandoval-Ramírez, Metacyclogenesis of Trypanosoma cruzi in B. ferroae (Reduviidae: Triatominae) and feces infectivity under laboratory conditions , Biomedica: Vol. 41 No. 1 (2021)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























