Primer estudio piloto para el tamizaje neonatal de linfopenias T y B en Colombia
Resumen
Introducción. La detección temprana de linfopenias congénitas facilitaría su tratamiento oportuno y la prevención de complicaciones serias, y optimizaría los recursos del sistema de salud.
Objetivo. Realizar un estudio piloto para el tamizaje neonatal de linfopenias mediante la cuantificación de los círculos de escisión (TREC y KREC) de los receptores de los linfocitos T y B en muestras de sangre periférica de neonatos de Medellín, Colombia.
Materiales y métodos. Se recolectaron muestras de 1.092 neonatos y de seis pacientes con sospecha de linfopenia, mediante pinchazo del talón o del dedo gordo del pie. Se extrajo el ADN y se midieron los niveles de TREC y KREC por qPCR.
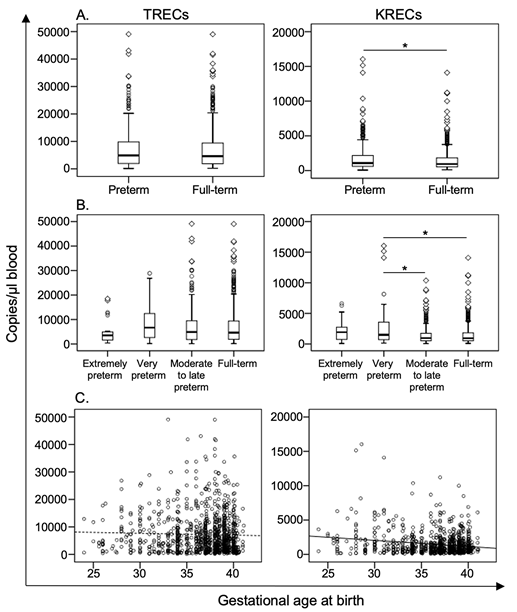
Resultados. En los seis pacientes con sospecha de linfopenia se encontraron niveles muy bajos o indetectables de TREC. Todos los neonatos tamizados presentaron niveles normales de TREC y KREC. Se observó concordancia entre los valores de TREC o de KREC en dos tipos diferentes de papel de filtro. Los niveles de TREC y KREC disminuyeron considerablemente después de 24 semanas de almacenamiento de las muestras de sangre en papel de filtro. Se encontró una asociación positiva entre los niveles de los TREC y el peso al nacer, y una correlación negativa entre los KREC y la edad gestacional. Los niveles de estos marcadores no se asociaron con el tipo de parto.
Conclusiones. Se presenta el primer estudio piloto para el tamizaje temprano de linfopenias congénitas en neonatos en Colombia. Se propone el uso de un límite de detección de 119 y 69 copias/μl de sangre de TREC y KREC, respectivamente, para futuros programas de detección neonatal de linfopenias en el país.
Descargas
Referencias bibliográficas
Bousfiha AA, Jeddane L, Ailal F, Benhsaien I, Mahlaoui N, Casanova JL, et al. Primary immunodeficiency diseases worldwide: More common than generally thought. J Clin Immunol. 2013;33:1-7. https://doi.org/10.1007/s10875-012-9751-7
Quinn J, Modell V, Orange JS, Modell F. Growth in diagnosis and treatment of primary immunodeficiency within the global Jeffrey Modell Centers Network. Allergy Asthma Clin Immunol. 2022;18:19. https://doi.org/10.1186/s13223-022-00662-6
Dorsey MJ, Wright NAM, Chaimowitz NS, Dávila-Saldaña BJ, Miller H, Keller MD, et al. Infections in infants with SCID: Isolation, infection screening, and prophylaxis in PIDTC centers. J Clin Immunol. 2021;41:38-50. https://doi.org/10.1007/s10875-020-00865-9
Barry JC, Crowley TB, Jyonouchi S, Heimall J, Zackai EH, Sullivan KE, et al. Identification of 22q11.2 deletion syndrome via newborn screening for severe combined immunodeficiency. J Clin Immunol. 2017;37:476-85. https://doi.org/10.1007/s10875-017-0403-9
Tangye SG, Al-Herz W, Bousfiha A, Cunningham-Rundles C, Franco JL, Holland SM, et al. Human inborn errors of immunity: 2022 Update on the Classification from the International Union of Immunological Societies Expert Committee. J Clin Immunol. 2022;42:1473-507. https://doi.org/10.1007/s10875-022-01289-3
Lev A, Somech R, Somekh I. Newborn screening for severe combined immunodeficiency and inborn errors of immunity. Curr Opin Pediatr. 2023;35:692-702. https://doi.org/10.1097/MOP.0000000000001291
Eissa H, Thakar MS, Shah AJ, Logan BR, Griffith LM, Dong H, et al. Post-transplantation late complications increase over time for patients with SCID: A Primary Immune Deficiency Treatment Consortium (PIDTC) landmark study. J Allergy Clin Immunol. 2024;153:287-96. https://doi.org/10.1016/j.jaci.2023.09.027
Douek DC, Vescio RA, Betts MR, Brenchley JM, Hill BJ, Zhang L, et al. Assessment of thymic output in adults after haematopoietic stem-cell transplantation and prediction of T-cell reconstitution. Lancet. 2000;355:1875-81. https://doi.org/10.1016/S0140-6736(00)02293-5
Chan K, Puck JM. Development of population-based newborn screening for severe combined immunodeficiency. J Allergy Clin Immunol. 2005;115:391-8. https://doi.org/10.1016/j.jaci.2004.10.012
Routes JM, Grossman WJ, Verbsky J, Laessig RH, Hoffman GL, Brokopp CD, et al. Statewide newborn screening for severe T-cell lymphopenia. JAMA. 2009;302:2465-70. https://doi.org/10.1001/jama.2009.1806
Dorsey MJ, Puck JM. Newborn screening for severe combined immunodeficiency in the United States: Lessons learned. Immunol Allergy Clin North Am. 2019;39:1-11. https://doi.org/10.1016/j.iac.2018.08.002
van Zelm MC, Szczepanski T, van der Burg M, van Dongen JJ. Replication history of B lymphocytes reveals homeostatic proliferation and extensive antigen-induced B cell expansion. J Exp Med. 2007;204:645-55. https://doi.org/10.1084/jem.20060964
Kanegae MPP, Barreiros LA, Sousa JL, Brito MAS, de Oliveira Junior EB, Soares LP, et al. Newborn screening for severe combined immunodeficiencies using TRECS and KRECS: Second pilot study in Brazil. Rev Paul Pedriatr. 2017;35:25-32. https://doi.org/10.1590/1984-0462/;2017;35;1;00013
Tagliaferri L, Kunz JB, Happich M, Esposito S, Bruckner T, Hübschmann D, et al. Newborn screening for severe combined immunodeficiency using a novel and simplified method to measure T-cell excision circles (TREC). Clin Immunol. 2017;175:51-5. https://doi.org/10.1016/j.clim.2016.11.016
Giraldo GA, Suárez-Obando F, Mora L, Sánchez P, Prieto JC. Evaluation of TSH levels in the program of congenital hypothyroidism newborn screening in a pilot study of preterm newborns in Bogotá, Colombia. J Inborn Errors Metab Screen. 2015;3. https://doi.org/10.1177/2326409815597706
Sottini A, Serana F, Bertoli D, Chiarini M, Valotti M, Vaglio-Tessitore M, et al. Simultaneous quantification of T-cell receptor excision circles (TRECs) and K-deleting recombination excision circles (KRECs) by real-time PCR. J Vis Exp. 2014;94:52184. https://doi.org/10.3791/52184
Baker MW, Grossman WJ, Laessig RH, Hoffman GL, Brokopp CD, Kurtycz DF, et al. Development of a routine newborn screening protocol for severe combined immunodeficiency. J Allergy Clin Immunol. 2009;124:522-7. https://doi.org/10.1016/j.jaci.2009.04.007
Kwan A, Hu D, Song M, Gomes H, Brown DR, Bourque T, et al. Successful newborn screening for SCID in the Navajo Nation. Clin Immunol. 2015;158:29-34. https://doi.org/10.1016/j.clim.2015.02.015
Esquerda M, Palau F, Lorenzo D, Cambra FJ, Bofarull M, Cusi V, et al. Ethical questions concerning newborn genetic screening. Clin Genet. 2021;99:93-8. https://doi.org/10.1111/cge.13828
Evans A, Bonhomme N, Goodman A, Terry SF. Newborn screening and health communications. Genet Test Mol Biomarkers. 2018;22:507-8. https://doi.org/10.1089/gtmb.2018.0234
van der Spek J, Groenwold RH, van der Burg M, van Montfrans JM. TREC based newborn screening for severe combined immunodeficiency disease: A systematic review. J Clin Immunol. 2015;35:416-30. https://doi.org/10.1007/s10875-015-0152-6
Kimizu T, Nozaki M, Okada Y, Sawada A, Morisaki M, Fujita H, et al. Multiplex real-time PCR-based newborn screening for severe primary immunodeficiency and spinal muscular atrophy in Osaka, Japan: Our results after 3 years. Genes (Basel). 2024;15:314. https://doi.org/10.3390/genes15030314
Taylor JL, Lee FK, Yazdanpanah GK, Staropoli JF, Liu M, Carulli JP, et al. Newborn blood spot screening test using multiplexed real-time PCR to simultaneously screen for spinal muscular atrophy and severe combined immunodeficiency. Clin Chem. 2015;61:412-9. https://doi.org/10.1373/clinchem.2014.231019
De Felipe B, Olbrich P, Goycochea-Valdivia W, Delgado-Pecellin C, Sánchez-Moreno P, Sánchez B, et al. Newborn screening for primary T- and B-cell immune deficiencies—A prospective study in Andalucía. Int J Neonatal Screen. 2017;3:27. https://doi.org/10.3390/ijns3040027
Rechavi E, Lev A, Simon AJ, Stauber T, Daas S, Saraf-Levy T, et al. First year of Israeli newborn screening for severe combined immunodeficiency – Clinical achievements and insights. Front Immunol. 2017;8:1448. https://doi.org/10.3389/fimmu.2017.01448
Zhao Q, Dai R, Li Y, Wang Y, Chen X, Shu Z, et al. Trends in TREC values according to age and gender in Chinese children and their clinical applications. Eur J Pediatr. 2022;181:529-38. https://doi.org/10.1007/s00431-021-04223-8
Ward CE, Baptist AP. Challenges of newborn severe combined immunodeficiency screening among premature infants. Pediatrics. 2013;131:e1298-302. https://doi.org/10.1542/peds.2012-1921
Audrain M, Thomas C, Mirallie S, Bourgeois N, Sebille V, Rabetrano H, et al. Evaluation of the T-cell receptor excision circle assay performances for severe combined immunodeficiency neonatal screening on Guthrie cards in a French single center study. Clin Immunol. 2014;150:137-9. https://doi.org/10.1016/j.clim.2013.11.012
Gaviglio A, Lasarev M, Sheller R, Singh S, Baker M. Newborn screening for severe combined immunodeficiency: Lessons learned from screening and follow-up of the preterm newborn population. Int J Neonatal Screen. 2023;9:68. https://doi.org/10.3390/ijns9040068
Wirbelauer J, Thomas W, Rieger L, Speer CP. Intrauterine growth retardation in preterm infants ≤ 32 weeks of gestation is associated with low white blood cell counts. Am J Perinatol. 2010;27:819-24. https://doi.org/10.1055/s-0030-1254547
Olearo E, Oberto M, Oggè G, Botta G, Pace C, Gaglioti P, et al. Thymic volume in healthy, small for gestational age and growth-restricted fetuses. Prenat Diagn. 2012;32:662-7. https://doi.org/10.1002/pd.3883
Schlinzig T, Johansson S, Stephansson O, Hammarström L, Zetterström RH, von Döbeln U, et al. Surge of immune cell formation at birth differs by mode of delivery and infant characteristics – A population-based cohort study. PloS ONE. 2017;12:e0184748. https://doi.org/10.1371/journal.pone.0184748
Neu J, Rushing J. Cesarean versus vaginal delivery: Long-term infant outcomes and the hygiene hypothesis. Clin Perinatol. 2011;38:321-31. https://doi.org/10.1016/j.clp.2011.03.008
Kristensen K, Henriksen L. Cesarean section and disease associated with immune function. J Allergy Clin Immunol. 2016;137:587-90. https://doi.org/10.1016/j.jaci.2015.07.040
Barreiros LA, Sousa JL, Geier C, Leiss-Piller A, Kanegae MPP, França TT, et al. SCID and other inborn errors of immunity with low TRECs – The Brazilian Experience. J Clin Immunol. 2022;42:1171-92. https://doi.org/10.1007/s10875-022-01275-9
Kumarasamy G, Khairiz K, Chang WL, Aye TT, Ali A. Paving the way in implementation of SCID newborn screening in developing nations: Feasibility study and strategies to move forward in Malaysia. Front Immunol. 2024;15:1400247. https://doi.org/10.3389/fimmu.2024
Huang J, Shankar A, Hurden I, Thomas R, Hill J, Seth D, et al. Increased mortality in infants with abnormal T-cell receptor excision circles. Pediatr Res. 2024;96:199-207. https://doi.org/10.1038/s41390-024-03121-7
Shih ST, Keller E, Wiley V, Wong M, Farrar MA, Chambers GM. Economic evaluation of newborn screening for severe combined immunodeficiency. Int J Neonatal Screen. 2022;8:44. https://doi.org/1010.3390/ijns8030044
Algunos artículos similares:
- Wilson Mejía-Naranjo, Myriam Sánchez-Gomez, La desnutrición proteica estimula la expresión de receptores de la hormona del crecimiento en linfocitos B esplénicos de rata , Biomédica: Vol. 24 Núm. 4 (2004)
- Adriana Cuéllar, Angela Fonseca, Alberto Gómez, Efecto del lipopolisacárido en cultivos de células dendríticas humanas y su inhibición por la polimixina B. , Biomédica: Vol. 24 Núm. 4 (2004)
- Clelia Rosa Calao, José Luis Marrugo, Efectos genotóxicos asociados a metales pesados en una población humana de la región de La Mojana, Colombia, 2013 , Biomédica: Vol. 35 (2015): Agosto, Suplemento 2, Salud y contaminantes ambientales
- Andrés Felipe Lamos-Duarte, Rafael Parra-Medina, Carlos Santiago Rivadeneira-Chamorro, Juan Pablo Castañeda-González, Alejandro Escobar, Adriana Rojas-Villarraga, Gabriel Santiago Rodríguez-Vargas, Ana María Arredondo, Héctor Cubides, José Fernando Polo , Juan José Capasso, Claudia Ibañez , Jairo Hernán Cajamarca-Barón, Características clínicas, histopatológicas e inmunohistoquímicas de pacientes con síndrome seco y biopsia de glándula salival con puntaje de foco de 1 o más , Biomédica: Vol. 45 Núm. 1 (2025)
Derechos de autor 2024 Biomédica

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























