Grupos de riesgo citogenético de leucemia mieloide aguda pediátrica a partir del análisis de supervivencia en un hospital de referencia para cáncer en Perú
Resumen
Introducción. La leucemia mieloide aguda es una neoplasia heterogénea caracterizada por la proliferación de células mieloides inmaduras. El análisis citogenético ha revelado la presencia de aberraciones cromosómicas de importancia en el pronóstico del paciente.
Objetivo. Determinar los grupos de riesgo citogenético de pacientes pediátricos con leucemia mieloide aguda a partir de la supervivencia global.
Materiales y métodos. Se hizo un estudio observacional de corte transversal. Se incluyeron los registros clínicos de los pacientes pediátricos con diagnóstico de leucemia mieloide aguda de novo admitidos en el Instituto Nacional de Enfermedades Neoplásicas entre el 2001 y el 2011 y sometidos a análisis citogenético de médula ósea. Los grupos de riesgo citogenético se establecieron según los criterios del Medical Research Council. Las curvas de supervivencia global se elaboraron con el método de Kaplan-Meier y se compararon mediante la prueba de Mantel-Cox y una regresión de Cox, utilizando el programa R, versión 3.3.2.
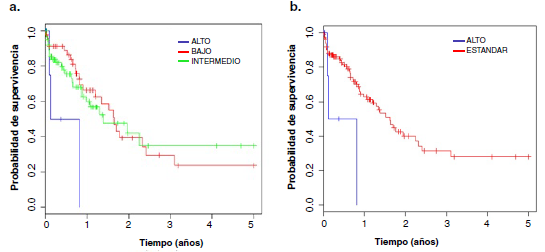
Resultados. Se incluyeron 130 pacientes, 68 varones (52,3 %) y 62 mujeres (47,7 %), mayoritariamente del subtipo M2 (33 %). La edad promedio fue de 7,7 (rango de 0 a 15 años). Se observaron aberraciones cromosómicas en el 60,8 % y la más frecuente fue la traslocación t(8;21). Según el análisis de supervivencia global, se observaron dos grupos de riesgo citogenético: favorable y desfavorable.
Conclusión. Se determinaron dos grupos de riesgo citogenético: alto (o desfavorable) y estándar (o favorable).
Descargas
Referencias bibliográficas
Kömür M, Erbey F, Bayram I, Tanyeli A. Incidence and prognostic importance of molecular genetic defects in children with acute myeloblastic leukemia. Asian Pac J Cancer Prev. 2010;11:1393-5.
De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: A comprehensive review and 2016 update. Blood Cancer J. 2016;6:e441. https://doi.org/10.1038/bcj.2016.50
Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Leukemia - Acute Myeloid Leukemia (AML). USA; 2020. Fecha de consulta: 8 de abril de 2020. Disponible en: https://seer.cancer.gov/statfacts/html/amyl.html
Creutzig U, Heuvel-Eibrink MM, Gibson B, Dworzak MN, Adachi S, de Bont E, et al. Diagnosis and management of acute myeloid leukemia in children and adolescents: Recommendations from an international expert panel. Blood. 2012;120:3187-3205. https://doi.org/10.1182/blood-2012-03-362608
Chen X, Pan J, Wang S, Hong S, Hong S, He S. The epidemiological trend of acute myeloid leukemia in childhood: A population-based analysis. J Cancer. 2019;10:4824-35. https://doi.org/10.7150/jca.32326
Bolouri H, Farrar JE, Triche T Jr, Ries RE, Lim EL, Alonzon TA, et al. The molecular landscape of pediatric acute myeloid leukemia reveals recurrent structural alterations and age-specific mutational interactions. Nat Med. 2018;24:103-12. https://doi.org/10.1038/nm.4439
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR, et al. Proposals for the classification of the acute leukaemias. French-American-British (FAB) co-operative group. Br J Haematol. 1976;33:451-8. https://doi.org/10.1111/j.1365-2141.1976.tb03563.x
Vardiman JW, Harris NL, Brunning RD. The World Health Organization (WHO) classification of the myeloid neoplasms. Blood. 2002;100:2292-302. https://doi.org/10.1182/blood-2002-04-1199
Forestier E, Heim S, Blennow E, Borgström G, Holmgren G, Heinonen K, et al. Cytogenetic abnormalities in childhood acute myeloid leukaemia: A Nordic series comprising all children enrolled in the NOPHO-93-AML trial between 1993 and 2001. Br J Haematol. 2003;121:566-77. https://doi.org/10.1046/j.1365-2141.2003.04349.x
Harrison CJ, Hills RK, Moorman AV, Grimwade DJ, Hann I, Webb DKH, et al. Cytogenetics of childhood acute myeloid leukemia: United Kingdom Medical Research Council Treatment Trials AML 10 and 12. J Clin Oncol. 2010;28:2674-81. https://ascopubs.org/doi/10.1200/JCO.2009.24.8997
Nunes A, Paes C, Murao M, Viana MB, De Oliveira BM. Cytogenetic abnormalities, WHO classification, and evolution of children and adolescents with acute myeloid leukemia. Hematol Transfus Cell Ther. 2019;41:236-43. https://doi.org/10.1016/j.htct.2018.09.007
Braoudaki M, Tzortzatou-Stathopoulou F. Clinical cytogenetics in pediatric acute leukemia: An update. Clin Lymphoma Myeloma Leuk Clinical. 2012;12:230-37. https://doi.org/10.1016/j.clml.2012.04.004
Lazarević V. Cytogenetics abnormalities in acute myeloid leukemia in Sweden. A population based study. Doctoral Dissertation. Lund: Lund University; 2015.
Milan T, Canaj HV, Villeneuve C, Ghosh A, Barabé F, Cellot S, et al. Pediatric leukemia: Moving toward more accurate models. Exp Hematol. 2019;74:1-12.
https://doi.org/10.1016/j.exphem.2019.05.003
Chen J, Glasser CL. New and emerging targeted therapies for pediatric acute myeloid leukemia (AML). Children (Basel) 2020;10:12. https://doi.org/10.3390/children7020012
Masetti R, Vendemini F, Zama D, Biagi C, Pession A, Locatelli F. Acute myeloid leukemia in infants: Biology and treatment. Front Pediatr. 2015;3:37. https://doi.org/10.3389/fped.2015.00037
Ross ME, Mahfouz R, Onciu M, Liu HC, Zhou X, Song G, et al. Gene expression profiling of pediatric acute myelogenous leukemia. Blood. 2004;104:3679-87. https://doi.org/10.1182/blood-2004-03-1154
Oorschot D-v, Kuipers JE, Arentsen-Peters S, Schotte D, de Haas V, Trka J, et al. Differentially expressed miRNAs in cytogenetic and molecular subtypes of pediatric acute myeloid leukemia. Pediatr Blood Cancer. 2012;58:715-21. https://doi.org/10.1002/pbc.23279
Meshinchi S, Ries RE, Trevino LR, Hampton OA, Alonzo T, Farrar JE, et al. Identification of novel somatic mutations, regions of recurrent loss of heterozygosity (LOH) and significant clonal evolution from diagnosis to relapse in childhood AML determined by exome capture sequencing – an NCI/COG Target AML Study. Blood. 2012;120:123. https://doi.org/10.1182/blood.V120.21.123.123
Dawod M, Hanbali A. Prognosis and survival in acute myelogenous leukemia, myeloid leukemia - Clinical diagnosis and treatment. London: IntechOpen; 2012. Peer-reviewed chapter. https://doi.org/10.5772/27092
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457-81. https://doi.org/10.2307/2281868
Stevenson M. Introduction to survival analysis. Palmerston North. 2007. Fecha de consulta: 16 de mayo de 2015. Disponible en: http://www.biecek.pl/statystykaMedyczna/Stevenson_survival_analysis_195.721.pdf
Ahmed ZA, Shaiakh MS, Moatter T, Nasir A. Karyotype complexity and characterization of childhood acute myeloid leukemia (AML) in Pakistan. Ann Oncol. 2018;29(S9):ix88. https://doi.org/10.1093/annonc/mdy437
Davis KL, Marina N, Arber DA, Ma L, Cherry A, Dahl GV, et al. Pediatric acute myeloid leukemia as classified using 2008 WHO criteria: A single-center experience. Am J Clin Pathol. 2013;139:818-25. https://doi.org/10.1309/AJCP59WKRZVNHETN
Balgobind BV, van den Heuvel-Eibrink MM, De Menezes RX, Reinhardt D, Hollink IHIM, Arentsen-Peters STJCM, et al. Evaluation of gene expression signatures predictive of cytogenetic and molecular subtypes of pediatric acute myeloid leukemia. Haematologica. 2011;96:221-30. https://doi.org/10.3324/haematol.2010.029660
Blais S, Boutroux H, Pasquet M, Leblanc T, Fenneteau O, Gandemer V, et al. Is acute myeloblastic leukemia in children under 2 years of age a specific entity? A report from the FRENCH ELAM02 Study Group. HemaSphere. 2019;3:6. https://doi.org/10.1097/HS9.0000000000000316
Junior LCB, Levy IE, Frances LTVM, Wanderley AV, Carneiro RM, Bentes AQ. Frequency of acute myeloid leukemia in children attended in Belém, Pará from August 2005 to May 2009. J Bras Patol Med. 2015;51:72-6. https://doi.org/10.5935/1676-2444.20150013
Rubnitz JE, Razzouk BI, Lensing S, Pounds S, Pui C-H, Ribeiro RC. Prognostic factors and outcome of recurrence in childhood acute myeloid leukemia. Cancer. 2007;109:157-63. https://doi.org/10.1002/cncr.22385
Wells RJ, Arthur DC, Srivastava A, Heerema NA, Le Beau M, Alonzo TA, et al. Prognostic variables in newly diagnosed children and adolescents with acute myeloid leukemia: Children’s Cancer Group Study 213. Leukemia. 2002;16:601-7. https://doi.org/10.1038/sj.leu.2402390
Farah RA, Horkos JG, Bustros YD, Farhat HZ, Abla O. A multicenter experience from Lebanon in childhood and adolescent acute myeloid leukemia: High rate of early death in childhood acute promyelocytic leukemia. Mediterr J Hematol Infect Dis. 2015;7:e2015012. https://doi.org/10.4084/MJHID.2015.012
Pession A, Masetti R, Rizzari C, Putti MC, Casale F, Fagioli F, et al. Results of the AIEOP AML 2002/01 multicenter prospective trial for the treatment of children with acute myeloid leukemia. Blood. 2013;122:170-8. https://doi.org/10.1182/blood-2013-03-491621
Arber DA, Stein AS, Carter NH, Ikle D, Forman SJ, Slovak ML. Prognostic impact of acute myeloid leukemia classification. Importance of detection of recurring cytogenetic abnormalities and multilineage dysplasia on survival. Am J Clin Pathol. 2003;119:672-80. https://doi.org/10.1309/EM7K-CQR4-GLMH-RCX4
Aziz F, Qureshi IZ. Clinical and cytogenetics analysis in Pakistani leukemia patients. Pak J Zool. 2008;40:147-57.
Fröhling S, Schlenk RF, Kayser S, Morhardt M, Benner A, Döhner K, et al. Cytogenetics and age are major determinants of outcome in intensively treated acute myeloid leukemia patients older than 60 years: Results from AMLSG trial AML HD98-B. Blood. 2006;108:3280-8. https://doi.org/10.1182/blood-2006-04-014324
Omer SH, Kordofani AA, Osman IM, Musa OH, Altayb HN, Elamin BK. Prevalence of the different FAB sub type of acute myeloid leukemia related to hematological parameters in Sudanese. J Hematol Blood Disord. 2017;3:1-5. https://doi.org/10.15744/2455-7641.3.102
Lunardon S, dos Santos E, Coriolano M, Ramos N. Acute myeloid leukemia: Survival analysis of patients at a university hospital of Paraná. Rev Bras Hematol Hemoter. 2015;37(1):21-7. https://doi.org/10.1016/j.bjhh.2014.11.008
Harani MS, Adil SN, Shaikh MU, Kakepoto GN, Khurshid M. Frequency of fab FAB subtypes in acute myeloid leukemia patients at Aga Khan University Hospital Karachi. J Ayub Med Coll Abbottabad. 2005;17:26-9.
Kalwinsky DK, Raimondi SC, Schell MJ, Mirro J Jr, Santana VM, Behm F, et al. Prognostic importance of cytogenetic subgroups in de novo pediatric acute nonlymphocytic leukemia. J Clin Oncol. 1990;8:75-83. https://doi.org/10.1200/JCO.1990.8.1.75
Mrózek K, Heinonen K, Bloomfield CD. Prognostic value of cytogenetic findings in adults with acute myeloid leukemia. Int J Hematol. 2000;72:261-71.
Teixeira M. Combined classical and molecular cytogenetic analysis of cancer. Eur J Cancer. 2002;38:1580-84. https://doi.org/10.1016/s0959-8049(02)00117-x
Dolz S, Barragán E, Fuster Ó, Llop M, Cervera J, Such E, et al. Novel real-time polymerase chain reaction assay for simultaneous detection of recurrent fusion genes in acute myeloid leukemia. J Mol Diagn. 2013;15:678-86. https://doi.org/10.1016/j.jmoldx.2013.04.003
Rubnitz JE, Raimondi SC, Halbert AR, Tong X, Srivastava DK, Razzouk BI, et al. Characteristics and outcome of t(8;21)-positive childhood acute myeloid leukemia: A single institution’s experience. Leukemia. 2002;16:2072-7. https://doi.org/10.1038/sj.leu.2402633
Zidanloo SG, Colaga AH. Geographic heterogeneity of the AML1-ETO fusion gene in Iranian patients with acute myeloid leukemia. Rep Biochem Mol Biol. 2014;3:7-13.
Hu S, Gao L, Wang Y, He H, Lu J, Xiao P, et al. Analysis of prognostic factors in Chinese pediatric acute myeloid leukemia. Blood. 2016;128:5258. https://doi.org/10.1182/blood.V128.22.5258.5258
Johansson B, Mertens F, Mitelman F. Geographic heterogeneity of neoplasia-associated chromosome aberrations. Genes Chromosom Cancer. 1991;3:1-7. https://doi.org/10.1002/gcc.2870030102
Kuchenbauer F, Schnittger S, Look T, Gilliland G, Tenen D, Haferlach T, et al. Identification of additional cytogenetic and molecular genetic abnormalities in acute myeloid leukaemia with t(8;21)/AML1-ETO. J Haematol. 2006;134:616-9. https://doi.org/10.1111/j.1365-2141.2006.06229.x
Dong P, Wang Z, Meng F, Luo L, Wei J, Sun H, et al. Clinical characteristics of t(8;21) acute myeloid leukemia with AML1/ETO fusion in a single center in China. Int J Clin Exp Med. 2018;11:9312-22.
Manola KN. Cytogenetics of pediatric acute myeloid leukemia. Eur J Haematol. 2009;83:391-405. https://doi.org/10.1111/j.1600-0609.2009.01308.x
Meshinchi S, Arceci RJ. Prognostic factors and risk-based therapy in pediatric acute myeloid leukemia. Oncologist. 2007;12:341-55. https://doi.org/10.1634/theoncologist.12-3-341
Martin ML, López M, de la Serna J, Ayala R, García L, Barreiro E. Grupos de riesgo citogenético en la leucemia mieloide aguda: comparación de los modelos adoptados por los grupos MRC (Medical Research Council, Reino Unido) y SWOG (South West Oncology Group de EE.UU.). Med Clin (Barc). 2003;121:121-5. https://doi.org/10.1016/S0025-7753(03)73878-0
Hirsch B, Alonzo TA, Gerbing RB, Kahwash S, Heerema-McKenney A, Aplenc R, et al. Abnormalities Of 12p are associated with high-risk acute myeloid leukemia: A Children’s Oncology Group Report. Blood. 2013;122:612. https://doi.org/10.1182/blood.V122.21.612.612
Soekarman D, von Lindern M, Daenen S, de Jong B, Fonatsch C, Heinze B, et al. The translocation (6;9)(p23;q34) shows consistent rearrangement of two genes and defines a myeloproliferative disorder with specific clinical features. Blood. 1992;79:2990-7. https://doi.org/10.1182/blood.V79.11.2990.bloodjournal79112990
Johnston DL, Alonzo TA, Gerbing RB, Hirsch B, Heerema NA, Ravindranath Y, et al. Outcome of pediatric patients with acute myeloid leukemia (AML) and -5/5q- abnormalities from five pediatric AML treatment protocols: A report from the Children’s Oncology Group. Pediatr Blood Cancer. 2013;60:2073-8. https://doi.org/10.1002/pbc.24573
Hasle H, Alonzo TA, Auvrignon A, Behar C, Chang M, Creutzig U, et al. Monosomy 7 and deletion 7q in children and adolescents with acute myeloid leukemia: An international retrospective study. Blood. 2007;109:4641-7. https://doi.org/10.1182/blood-2006-10-051342
Soupir CP, Vergilio J-A, Cin PD, Muzikansky A, Kantarjian H, Jones D, et al. Philadelphia Chromosome–positive acute myeloid leukemia a rare aggressive leukemia with clinicopathologic features distinct from chronic myeloid leukemia in myeloid blast crisis. Am J Clin Pathol. 2007;127:642-50. https://doi.org/10.1309/B4NVER1AJJ84CTUU
Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, et al. Refinement of cytogenetic classification in acute myeloid leukemia: Determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood. 2010;116:354-65. https://doi.org/10.1182/blood-2009-11-254441
Pession A, Masetti R, Rizzari C, Putti MC, Casale F, Fagioli F, et al. Results of the AIEOP AML 2002/01 multicenter prospective trial for the treatment of children with acute myeloid leukemia. Blood. 2013;122:170-8. https://doi.org/10.1182/blood-2013-03-491621
Farrar JE, Schuback HL, Ries RE, Wai D, Hampton OA, Trevino LR, et al. genomic profiling of pediatric acute myeloid leukemia reveals a changing mutational landscape from disease diagnosis to relapse. Cancer Res. 2016;76:2197-205. https://doi.org/10.1158/0008-5472.CAN-15-1015
Algunos artículos similares:
- Ana Carolina Guimaraes, Ruy Lino-Junior, Virlanea Lima, Camila Cavellani, Rosana Rosa Corrêa, Mauricio Llaguno, Marlene Reis, Vicente Teixeira, Análisis epidemiológico de pacientes coinfectados con enfermedad de Chagas y cisticercosis , Biomédica: Vol. 29 Núm. 1 (2009)
- Keila Torres, Francys Avendaño-Rangel, Eliécer Lizano, María Rojas, Claudina Rodríguez-Bonfante, Rafael Bonfante-Cabarcas, Elis Aldana, Evaluación de la estructura espacial de Triatoma maculata del centro-occidente de Venezuela y su viabilidad alimentado con sangre humana en condiciones de laboratorio , Biomédica: Vol. 30 Núm. 1 (2010)
- John F. Arboleda, Luis F. García, Cristiam M. Álvarez, Papel de las células dendríticas tolerogénicas ILT3+/ILT4+ en la respuesta inmunitaria a trasplante de órganos y tejidos , Biomédica: Vol. 31 Núm. 2 (2011)
- Diana Carolina Suárez, Ángela Patricia Rey, Magda Lorena Orduz, Renzo Leonardo Prada, Zorayda Tarazona, Supervivencia de Trypanosoma cruzi en bebidas experimentalmente contaminadas , Biomédica: Vol. 32 Núm. 1 (2012)
- Marcela González, Claudia Patricia González, Alvaro Sanabria, Determinación ecográfica del volumen normal de la glándula tiroides en una población pediátrica de Bogotá, Colombia. , Biomédica: Vol. 26 Núm. 1 (2006)
- Wilson Daza, Silvana Dadan, Michelle Higuera, Perfil de las enfermedades gastrointestinales en un centro de gastroenterología pediátrica en Colombia: 15 años de seguimiento , Biomédica: Vol. 37 Núm. 3 (2017)
- Karime Osorio-Arango, Mauricio Beltrán-Durán, Yazmín Arias-Murillo, Franklyn Prieto, Adriana Robayo, Supervivencia de receptores de trasplante renal en Colombia, 2008-2012 , Biomédica: Vol. 37 Núm. 2 (2017)
- Luisa F. Imbachi, Lina M. Ibañez , Paula Hurtado-Villa, Estado de salud y barreras en la atención de niños con defectos congénitos nacidos entre el 2011 y el 2017 en dos instituciones de salud de Cali , Biomédica: Vol. 40 Núm. 1 (2020)
- Freddy Israel Pantoja, Willinton Robert Ricaurte , Diana Elizabeth Rosero , Relación entre la muerte y el ingreso a cuidados intensivos de pacientes pediátricos con bacteriemia por Staphylococcus aureus adquirido en la comunidad, 2014-2017 , Biomédica: Vol. 41 Núm. 1 (2021)
- Jorge Andrés Puerto, Susana Uribe , Víctor Calvo, Ricardo Cardona, Efecto de la inmunoterapia con alérgenos específicos en pacientes pediátricos con asma atendidos en una institución de salud de Colombia , Biomédica: Vol. 41 Núm. 3 (2021)
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























