Sensibilidad in vitro a benznidazol, nifurtimox y posaconazol de cepas de Trypanosoma cruzi de Paraguay
Resumen
Introducción. Trypanosoma cruzi, agente causal de la enfermedad de Chagas, exhibe una sustancial heterogeneidad fenotípica y genotípica que puede influir en las variaciones epidemiológicas y clínicas de la enfermedad, así como en la sensibilidad a los fármacos utilizados en el tratamiento.
Objetivo. Evaluar la sensibilidad in vitro al benznidazol, el nifurtimox y el posaconazol de 40 cepas clonadas de T. cruzi de Paraguay, con distintos genotipos, huéspedes y localidades de origen.
Materiales y métodos. En su estado epimastigote, los parásitos se incubaron en medio de cultivo LIT (Liver Infusion Tryptose) con diferentes concentraciones de cada fármaco en ensayos por triplicado. El grado de sensibilidad se estimó a partir de las concentraciones inhibitorias del 50 y el 90% (IC50 e IC90) y se obtuvieron los valores promedio y la desviación estándar de cada cepa y fármaco. La significación estadística entre grupos se determinó mediante análisis de varianzas con el test no paramétrico de Wilcoxon/Kruskal-Wallis y valores de p<0,05.
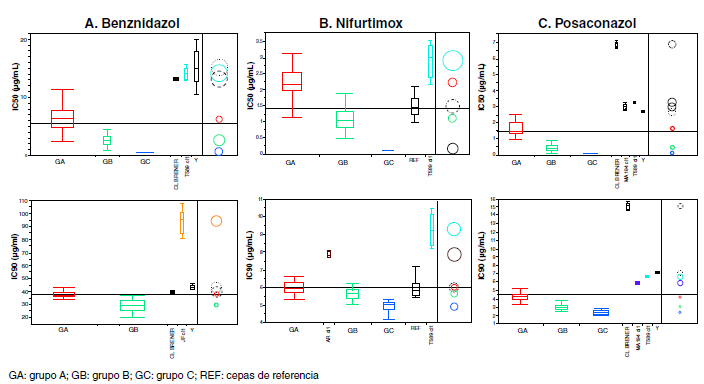
Resultados. Se observó un amplio rango de respuesta a los fármacos. Se identificaron dos grupos de parásitos (A y B) con diferencias significativas en la sensibilidad al benznidazol (p<0,0001), y tres grupos (A, B, C) en cuanto a la sensibilidad al nifurtimox y el posaconazol (p<0,0001).
Conclusiones. En general, las cepas fueron más sensibles al nifurtimox que al benznidazol y el posaconazol. Estas diferencias evidencian la heterogeneidad de las poblaciones de T. cruzi que circulan en Paraguay, lo que debe considerarse en el tratamiento y el seguimiento de las personas afectadas.
Descargas
Referencias bibliográficas
Stanaway JD, Roth G. The burden of Chagas disease: Estimates and challenges. Glob Heart. 2015;10:139-44. https://doi.org/10.1016/j.gheart.2015.06.001
World Health Organization (WHO). Integrating neglected tropical diseases into global health and development. Fourth WHO report on neglected tropical diseases, 2017. Fecha de consulta: 20 de mayo del 2019. Disponible en: https://www.who.int/neglected_diseases/resources/9789241565448/en/
Herricks JR, Hotez PJ, Wanga V, Coffeng LE, Haagsma JA, Basáñez MG, et al. The global burden of disease study 2013: What does it mean for the NTDs? PLoS Negl Trop Dis. 2017;11:e0005424. https://doi.org/10.1371/journal.pntd.0005424
Buccheri R, Kassab MJ, Freitas VL, Silva SC, Bezerra RC, Khoury Z, et al. Chagasic meningoencephalitis in an HIV infected patient with moderate immunosuppression: Prolonged survival and challenges in the HAART era. Rev Inst Med Trop Sao Paulo. 2015;57:531-5. https://doi.org/10.1590/S0036-46652015000600014
Gray EB, La Hoz RM, Green JS, Vikram HR, Benedict T, Rivera H, et al. Reactivation of Chagas disease among heart transplant recipients in the United States, 2012-2016. Transpl Infect Dis. 2018;20:e12996. https://doi.org/10.1111/tid.12996
Kaushal M, Shabani S, Cochran EJ, Samra H, Zwagerman NT, Kaushal M. Cerebral trypanosomiasis in an immunocompromised patient: Case report and review of the literature. World Neurosurg. 2019;pii:S1878-875031537-2. https://doi.org/10.1016/j.wneu.2019.05.260
Pereiro AC. Guidelines for the diagnosis and treatment of Chagas disease. Lancet.2019;393:1486-7. https://doi.org/10.1016/S0140-6736(19)30288-0
Sangenito LS, da Silva Santos V, d’Avila-Levy CM, Branquinha MH, Souza Dos Santos AL, de Oliveira SSC. Leishmaniasis and Chagas disease - neglected tropical diseases: Treatment updates. Curr Top Med Chem. 2019;19:174-7. https://doi.org/10.2174/156802661903190328155136
Filardi LS, Brener Z. Susceptibility and natural resistance of Trypanosoma cruzi strains to drugs used clinically in Chagas disease. Trans R Soc Trop Med Hyg. 1987;81:755-9. https://doi.org/10.1016/0035-9203(87)90020-4
Luna KP, Hernández IP, Rueda CM, Zorro MM, Croft SL, Escobar P. In vitro susceptibility of Trypanosoma cruzi strains from Santander, Colombia, to hexadecylphosphocholine (miltefosine), nifurtimox and benznidazole. Biomédica. 2009;29:448-55. https://doi.org/10.7705/biomedica.v29i3.15
Mejía-Jaramillo AM, Fernández GJ, Montilla M, Nicholls RS, Triana-Chávez O. Trypanosoma cruzi strains resistant to benznidazole occurring in Colombia. Biomédica. 2012;32:196-205. https://doi.org/10.1186/1756-3305-4-169
Dos Santos FM, Caldas S, de Assis Cáu SB, Crepalde GP, de Lana M, Machado-Coelho GL, et al. Trypanosoma cruzi: Induction of benznidazole resistance in vivo and its modulation by in vitro culturing and mice infection. Exp Parasitol. 2008;120:385-90. https://doi.org/10.1016/j.exppara.2008.09.007
Mejía AM, Hall BS, Taylor MC, Gómez-Palacio A, Wilkinson SR, Triana-Chávez O, et al. Benznidazole-resistance in Trypanosoma cruzi is a readily acquired trait that can arise independently in a single population. J Infect Dis. 2012;206:220-8. https://doi.org/10.1093/infdis/jis331
Urbina JA, Payares G, Contreras LM, Liendo A, Sanoja C, Molina J, et al. Antiproliferative effects and mechanism of action of SCH 56592 against Trypanosoma (Schizotrypanum) cruzi: In vitro and in vivo studies. Antimicrob Agents Chemother. 1998;42:1771-7. https://doi.org/10.1128/AAC.42.7.1771
Urbina JA, Payares G, Sanoja C, Lira R, Romanha AJ. In vitro and in vivo activities of ravuconazole on Trypanosoma cruzi, the causative agent of Chagas disease. Int J Antimicrob Agents. 2003;21:27-38. https://doi.org/10.1016/s0924-8579(02)00273-x
Molina I, Gómez i Prat J, Salvador F, Treviño B, Sulleiro E, Serre N, et al. Randomized trial of posaconazole and benznidazole for chronic Chagas’ disease. N Engl J Med. 2014;37020:1899-908. https://doi.org/10.1056/NEJMoa1313122
Morillo CA, Waskin H, Sosa-Estani S, Del Carmen Bangher M, Cuneo C, Milesi R, et al. STOP-CHAGAS Investigators. Benznidazole and posaconazole in eliminating parasites in asymptomatic T. cruzi carriers: The STOP-CHAGAS trial. J Am Coll Cardiol. 2017;69:939-47. https://doi.org/10.1016/j.jacc.2016.12.023
Zingales B, Miles MA, Campbell DA, Tibayrenc M, Macedo AM, Teixeira MM, et al. The revised Trypanosoma cruzisubspecific nomenclature: Rationale, epidemiological relevance and research applications. Infect Genet Evol. 2012;12:240-53. https://doi.org/10.1016/j.meegid.2011.12.009
Burgos JM, Begher S, Silva HM, Bisio M, Duffy T, Levin MJ, et al. Molecular identification of Trypanosoma cruzi I tropism for central nervous system in Chagas reactivation due to AIDS. Am J Trop Med Hyg. 2008;78:294-7.
Llewellyn MS, Rivett-Carnac JB, Fitzpatrick S, Lewis MD, Yeo M, Gaunt MW, et al. Extraordinary Trypanosoma cruzi diversity within single mammalian reservoir hosts implies a mechanism of diversifying selection. Int J Parasitol. 2011;41:609-14. https://doi.org/10.1016/j.ijpara.2010.12.004
Bontempi IA, Bizai ML, Ortiz S, Manattini S, Fabbro D, Solari A, et al. Simple methodology to directly genotype Trypanosoma cruzi discrete typing units in single and mixed infections from human blood samples. Infect Genet Evol. 2016;43:123-9. https://doi.org/10.1016/j.meegid.2016.05.026
Chapman MD, Baggaley RC, Godfrey-Fausset PF, Malpas TJ, White G, Canese J, et al. Trypanosoma cruzi from the Paraguayan Chaco: Isoenzyme profiles of strains isolated at Makthlawaiya. J Protozool. 1984;31:482-6. https://doi.org/10.1111/j.1550-7408.1984.tb02999.x
Cura CI, Mejía-Jaramillo AM, Duffy T, Burgos JM, Rodriguero M, Cardinal MV, et al. Trypanosoma cruzi I genotypes in different geographical regions and transmission cycles based on a microsatellite motif of the intergenic spacer of spliced-leader genes. Int J Parasitol. 2010;40:1599-607. https://doi.org/10.1016/j.ijpara.2010.06.006
del Puerto F, Sánchez Z, Nara E, Meza G, Paredes B, Ferreira E, et al. Trypanosoma cruzi lineages detected in congenitally infected infants and Triatoma infestans from the same disease-endemic region under entomologic surveillance in Paraguay. Am J Trop Med Hyg. 2010;82:386-90. https://doi.org/10.4269/ajtmh.2010.09-0006
Sánchez Z, Russomando G, Chena L, Nara E, Cardozo E, Paredes B, et al. Triatoma sordida, indicadores de adaptación y transmisión de Trypanosoma cruzi en intradomicilio del Chaco Paraguayo. Mem Inst Investig Cienc Salud. 2016;14:96-101. https://doi.org/10.18004/Mem.iics/1812-9528/2016.014(03)96-101
Acosta N, Miret J, López E, Schinini A. First report of Sapajus cay naturally infected by Trypanosoma cruzi in San Pedro Department, Paraguay. Rev Bras Parasitol Vet. 2016;25:327-32. https://doi.org/10.1590/S1984-29612016052
Acosta N, López E, Lewis MD, Llewellyn MS, Gómez A, Román F, et al. Hosts and vectors of Trypanosoma cruzi discrete typing units in the Chagas disease endemic region of the Paraguayan Chaco. Parasitol. 2017;144:884-98. https://doi.org/10.1017/S0031182016002663
Camargo E. Growth and differentiation in Trypanosoma cruzi I. Origen of metacyclic trypanosomes in liquid media. Rev Inst Med Trop São Paulo. 1964;6:93-100.
Yeo M, Lewis MD, Carrasco HJ, Acosta N, Llewellyn M, da Silva Valente SA, et al. Resolution of multiclonal infections of Trypanosoma cruzi from naturally infected triatomine bugs and from experimentally infected mice by direct plating on a sensitive solid medium. Int J Parasitol. 2007;37:111-20. https://doi.org/10.1016/j.ijpara.2006.08.002
Yeo M, Acosta N, Llewellyn M, Sánchez H, Adamson S, Miles GA, et al. Origins of Chagas disease: Didelphis species are natural hosts of Trypanosoma cruzi I and armadillos hosts of Trypanosoma cruzi II, including hybrids. Int J Parasitol. 2005;35:225-33. https://doi.org/10.1016/j.ijpara.2004.10.024
Lewis MD, Ma J, Yeo M, Carrasco HJ, Llewellyn MS, Miles MA. Genotyping of Trypanosoma cruzi: Systematic selection of assays allowing rapid and accurate discrimination of all known lineages. Am J Trop Med Hyg. 2009;81:1041-9. https://doi.org/10.4269/ajtmh.2009.09-0305
Andrade SG, Magalhães JB, Pontes AL. Evaluation of chemotherapy with benznidazole and nifurtimox in mice infected with Trypanosoma cruzi strains of different types. Bull World Health Organ. 1985;63:721-6.
Muñoz-Calderón A, Santaniello A, Pereira A, Yannuzzi J, Díaz-Bello Z, Alarcón de Noya B. Susceptibilidad in vitro a nifurtimox y benznidazol de aislados de Trypanosoma cruzi obtenidos de pacientes venezolanos con enfermedad de Chagas infectados por mecanismos de transmisión oral y vectorial. Rev Ibero-Latinoam Parasitol. 2012;71:14-22.
Wilkinson SR, Taylor MC, Horn D, Kelly JM, Cheeseman I. A mechanism for cross-resistance to nifurtimox and benznidazole in trypanosomes. Proc Natl Acad Sci USA. 2008;105:5022-7. https://doi.org/10.1073/pnas.0711014105
Mejía-Jaramillo AM, Fernández GJ, Palacio L, Triana-Chávez O. Gene expression study using real-time PCR identifies an NTR gene as a major marker of resistance to benzonidazole in Trypanosoma cruzi. Parasit Vectors. 2011;4:169. https://doi.org/10.1186/1756-3305-4-169
Campos MC, Leon LL, Taylor MC, Kelly JM. Benznidazole-resistance in Trypanosoma cruzi: Evidence that distinct mechanisms can act in concert. Mol Biochem Parasitol. 2014;193:17-9. https://doi.org/10.1016/j.molbiopara.2014.01.002
González L, García-Huertas P, Triana-Chávez O, García GA, Murta SMF, Mejía-Jaramillo AM. Aldo-ketoreductase and alcohol dehydrogenase contribute to benznidazole natural resistance in Trypanosoma cruzi. Mol Microbiol. 2017;106:704-18. https://doi.org/10.1111/mmi.13830
García-Huertas P, Mejía-Jaramillo AM, Machado CR, Guimarães AC, Triana-Chávez O. Prostaglandin F2α synthase in Trypanosoma cruzi plays critical roles in oxidative stress and susceptibility to benznidazole. R Soc Open Sci. 2017a;4:170773. https://doi.org/10.1098/rsos.170773
Quebrada-Palacio LP, González MN, Hernandez-Vasquez Y, Perrone AE, Parodi-Talice A, Bua J, et al. Phenotypic diversity and drug susceptibility of Trypanosoma cruzi TcV clinical isolates. PLoS ONE. 2018;13:e020346. https://doi.org/10.1371/journal.pone.0203462
Campos MC, Castro-Pinto DB, Ribeiro GA, Berredo-Pinho MM, Gomes LH, da Silva Bellieny MS, et al. P-glycoprotein efflux pump plays an important role in Trypanosoma cruzi drug resistance. Parasitol Res. 2013;112:2341-51. https://doi.org/10.1007/s00436-013-3398-z
García-Huertas P, Mejía-Jaramillo AM, González L, Triana-Chávez O. Transcriptome and functional genomics reveal the participation of adenine phosphoribosyltransferase in Trypanosoma cruzi resistance to benznidazole. J Cell Biochem. 2017;118:1936-45. https://doi.org/10.1002/jcb.25978
Nozaki T, Engel JC, Dvorak JA. Cellular and molecular biological analyses of nifurtimox resistance in Trypanosoma cruzi. Am J Trop Med Hyg. 1996;55:111-7. https://doi.org/10.4269/ajtmh.1996.55.111
Toledo MJ, Bahia MT, Carneiro CM, Martins-Filho OA, Tibayrenc M, Barnabé C, et al. Chemotherapy with benznidazole and itraconazole for mice infected with different Trypanosoma cruzi clonal genotypes. Antimicrob Agents Chemother. 2003;47:223-30. https://doi.org/10.1128/aac.47.1.223-230.2003
Villarreal D, Barnabè C, Sereno D, Tibayrenc M. Lack of correlation between in vitro susceptibility to benznidazole and phylogenetic diversity of Trypanosoma cruzi, the agent of Chagas disease. Exp Parasitol. 2004;108:24-31. https://doi.org/10.1016/j.exppara.2004.07.001
Teston AP, Monteiro WM, Reis D, BossolaniGleison DP, Gomes ML, de Araújo SM, et al. In vivo susceptibility to benznidazole of Trypanosoma cruzi strains from the western Brazilian Amazon. Trop Med Int Health. 2013;18:85-95. https://doi.org/10.1111/tmi.12014
Gruendling AP, Massago M, Teston AP, Monteiro WM, Kaneshima EN, Araújo SM, et al. Impact of benznidazole on infection course in mice experimentally infected with Trypanosoma cruzi I, II, and IV. Am J Trop Med Hyg. 2015;92:1178-89. https://doi.org/10.4269/ajtmh.13-0690
Wilkinson SR, Bot C, Kelly JM, Hall BS. Trypanocidal activity of nitroaromatic prodrugs: Current treatments and future perspectives. Curr Top Med Chem. 2011;11:2072-84. https://doi.org/10.2174/156802611796575894
Hall BS, Wilkinson SR. Activation of benznidazole by trypanosomal type I nitroreductases results in glyoxal formation. Antimicrob Agents Chemother. 2012;56:115-23. https://doi.org/10.1128/AAC.05135-11
Moraes CB, Giardini MA, Kim H, Franco CH, Araujo-Junior AM, Schenkman S, et al. Nitroheterocyclic compounds are more efficacious than CYP51 inhibitors against Trypanosoma cruzi: Implications for Chagas disease drug discovery and development. Sci Rep. 2014;4:4703. https://doi.org/10.1038/srep04703
Cal M, Ioset JR, Fügi MA, Mäser P, Kaiser M. Assessing anti-T. cruzi candidates in vitro for sterile cidality. Int J Parasitol Drugs Drug Resist. 2016;6:165-70. https://doi.org/10.1016/j.ijpddr.2016.08.003
MacLean LM, Thomas J, Lewis MD, Cotillo I, Gray DW, De Rycker M. Development of Trypanosoma cruzi in vitro assays to identify compounds suitable for progression in Chagas’disease drug discovery. PLoS Negl Trop Dis. 2018;12:e0006612. https://doi.org/10.1371/journal.pntd.0006612
Burgos LG, Ortiz BD, Canese A, Ojeda A, Melo M. Reactivation of Chagas disease y immunosuppressive therapy in a patient with systemic lupus erythematosus: Report of an exceptional case. Am J Dermatopathol. 2012;34:e84-9. https://doi.org/10.1097/DAD.0b013e318257f9e2
Vera de Bilbao N, Samudio M, Schinini A, Acosta N, López E, González N, et al. Evaluación a 24 meses post-tratamiento con benznidazol en niños de 6 a12 años infectados con Trypanosoma cruzi. Rev Patol Trop. 2004;33:301-12.
Bustamante JM, Craft JM, Crowe BD, Ketchie SA, Tarleton RL. New, combined, and reduced dosing treatment protocols cure Trypanosoma cruzi infection in mice. J Infect Dis 2014;209:150-62. https://doi.org/10.1093/infdis/jit420
Diniz L de F, Urbina JA, de Andrade IM, Mazzeti AL, Martins TA, Caldas IS, et al. Benznidazole and posaconazole in experimental Chagas disease: Positive interaction in concomitant and sequential treatments. PLoS Negl Trop Dis. 2013;7:e2367. https://doi.org/10.1371/journal.pntd.0002367
Campos MC, Phelan J, Francisco AF, Taylor MC, Lewis MD, Pain A, et al. Genome-wide mutagenesis and multi-drug resistance in American trypanosomes induced by the front-line drug benznidazole. Sci Rep. 2017;7:14407. https://doi.org/10.1038/s41598-017-14986-6
Diniz LF, Caldas IS, Guedes PMM, Crepalde G, de Lana M, Carneiro CM, et al. Effects of ravuconazole treatment on parasite load and immune response in dogs experimentally infected with Trypanosoma cruzi. Antimicrob Agent Chemother. 2010;54:2979-86. https://doi.org/10.1128/AAC.01742-09
Caldas S, Caldas IS, Cecílio AB, Diniz LF, Talvani A, Ribeiro I, et al. Therapeutic responses to different anti-Trypanosoma cruzi drugs in experimental infection by benznidazole resistant stock. Parasitol. 2014;21:1-10. https://doi.org/10.1017/S0031182014000882
Algunos artículos similares:
- Iveth J. González, Las metacaspasas y su rol en la vida y muerte de los parásitos protozoarios humanos , Biomédica: Vol. 29 Núm. 3 (2009)
- Patricia Escobar, Katherine Paola Luna, Indira Paola Hernández, César Mauricio Rueda, María Magdalena Zorro, Simon L. Croft, Susceptibilidad in vitro a hexadecilfosfocolina (miltefosina), nifurtimox y benznidazole de cepas de Trypanosoma cruzi aisladas en Santander, Colombia , Biomédica: Vol. 29 Núm. 3 (2009)
- María Esther Pedrozo, Sandra Ocampos, Rosa Galeano, Andrea Ojeda, Agueda Cabello, Dalva De Assis, Casos de intoxicación aguda por plaguicidas en la colonia Puerto Pirapó, Itapúa, Paraguay, febrero de 2014 , Biomédica: Vol. 37 Núm. 2 (2017)
- Andrea Arévalo, Julio César Carranza, Felipe Guhl, Jairo A. Clavijo, Gustavo Adolfo Vallejo, Comparación de los patrones de alimentación y defecación de Rhodnius colombiensis y Rhodnius prolixus (Hemiptera, Reduviidae, Triatominae) en condiciones de laboratorio , Biomédica: Vol. 27 Núm. 1esp (2007): Enfermedad de Chagas
- Gustavo Adolfo Vallejo, Felipe Guhl, Julio César Carranza, Omar Triana, Gerardo Pérez, Paola Andrea Ortiz, Dairo Humberto Marín, Lina Marcela Villa, Jazmín Suárez, Isaura Pilar Sánchez, Ximena Pulido, Ingrid Bibiana Rodríguez, Leyder Elena Lozano, Daniel Alfonso Urrea, Fredy Arvey Rivera, César Cuba-Cuba, Jairo Alfonso Clavijo, Interacción tripanosoma-vector-vertebrado y su relación con la sistemática y la epidemiología de la tripanosomiasis americana , Biomédica: Vol. 27 Núm. 1esp (2007): Enfermedad de Chagas
- Myriam C. López, Sofía Duque, Luis C. Orozco, Diana Camargo, Luis E. Gualdrón, Elvia Cáceres, Margarita Ronderos, Maritza Rey, Augusto Corredor, Inmunodiagnóstico de la infección chagásica por ELlSA , Biomédica: Vol. 19 Núm. 2 (1999)
- Luis C. Orozco, Diana Camargo, Myriam C. López, Sofía Duque, Luis E. Gualdrón, Elvia Cáceres, Margarita Ronderos, Maritza Rey, Augusto Corredor, Inmunodiagnóstico de la infección en humanos por Trypanosoma cruzi mediante ELISA utilizando sangre recolectada en papel de filtro , Biomédica: Vol. 19 Núm. 2 (1999)
- Claudia Urueña, Paola Santander, Hugo Díez, Marleny Montilla, Ignacio Zarante, María del Carmen Thomas, Manuel Carlos López, Concepción Puerta, Localización cromosómica de los genes KMP-11 en cepas KP1(+) y KP1 (-) de Trypanosoma rangeli. , Biomédica: Vol. 24 Núm. 2 (2004)
- Mariana Gómez-Camponovo, José Moreno, Álvaro Javier Idrovo, Malvina Páez, Marcel Achkar, Evaluación del sistema de vigilancia epidemiológica del dengue en Paraguay entre 2009 y 2011 mediante la ley de Benford , Biomédica: Vol. 36 Núm. 4 (2016)
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























