Infections by Ehrlichia spp., Anaplasma spp. and Babesia spp. In Puerto Ordaz, Bolívar, Venezuela
Abstract
Introduction. The prevalence of tick-borne infections remains unknown in most Latin American countries, including Venezuela.
Objective. To estimate the prevalence of tick-borne infections in humans and dogs in Puerto Ordaz, Bolívar state, Venezuela.
Materials and methods. An exploratory, descriptive, and prospective study was conducted involving 181 individuals –who voluntarily agreed to participate and provided their written informed consent– and 10 dogs brought in by their owners. A clinical-epidemiological survey was conducted, and venous and capillary blood samples were collected. Buffy coat and blood smear tests were prepared and stained using Giemsa.
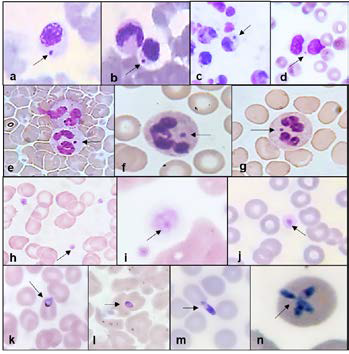
Results. Tick-borne infections were detected in 85.1% (n = 154) of the participants. The most frequent infection was caused by Ehrlichia spp. (n = 153; 84.5%), followed by Babesia spp. (n = 39; 21.5%), and Anaplasma spp. (n = 39; 21.5%). Women were the most affected (n = 117; 64.6%), with ages ranging from 5 to 97 years. Among infected individuals, 29.3% (n = 53) were co-infected with Ehrlichia spp. and Babesia spp., while 6.4% (n = 12) presented co-infection with all three pathogens under study. All the dogs (n = 10) presented tick-borne infections: 10 with Ehrlichia spp., 5 with Anaplasma spp., and 5 with Babesia spp. A statistically significant association was found between the presence of peridomestic ticks –favored by not walking the dog and having sick or elderly individuals– and tick-borne infections (p < 0.05).
Conclusions. A high prevalence of tick-borne diseases is evidenced, both in the studied humans and dogs.
Downloads
References
1. Sánchez S, Tagliafierro T, Coleman J, Benach J, Tokarz R. Polymicrobial nature of tickborne diseases. mBio. 2019;10:2055-19. https://doi.org/10.1128/mBio.02055-19
2. Alcántara-Rodríguez V, Sánchez-Montes S, Contreras H, Colunga-Salas P, Fierro-Flores L, Avalos S, et al. Human monocytic ehrlichiosis, México City, México. Emerg Infect Dis. 2020;26:3016-9. https://doi.org/10.3201/eid2612.200520
3. Madison-Antenucci S, Kramer L, Gebhardt L, Kauffman E. Emerging tick-borne diseases. Clin Microbiol Rev. 2020;33:00083-19. https://doi.org/10.1128/cmr.e00083-18
4. Erickson TA, Mayes B, Murray KO, Gunter SM. The epidemiology of human ehrlichiosis in Texas, 2008-2017. Ticks Tick Borne Dis. 2021;12:101788. https://doi.org/10.1016/j.ttbdis.2021.101788
5. Bamford C, Blumberg LH, Bosman M, Frean J, Hoek KGP, Miles J, et al. Neoehrlichiosis in symptomatic immunocompetent child, South Africa. Emerg Infect Dis. 2023;29:407-10. https://doi.org/10.3201/eid2902.221451
6. Ismail N, McBride JW. Emerging tick-borne infections: Ehrlichiosis and anaplasmosis. Clin Lab Med. 2017;37:317-40. https://doi.org/10.1016/j.cll.2017.01.006
7. Vieira R, Biondo A, Guimarães A, Santos A, Santos R, Dutra L, et al. Ehrlichiosis in Brazil. Rev Bras Parasitol Vet. 2011;20:1-12. https://doi.org/10.1590/S1984-29612011000100002
8. Anaya-Ramírez E, Palacios-Salvatierra R, Mosquera P, Álvarez C, Peralta C, Gonzales R, et al. Prevalence of antibodies to rickettsias and ehrlichias in four border departments of Perú. Rev Peru Med Exp Salud Pública. 2017;34:268-72. https://doi.org/10.17843/rpmesp.2017.342.1812
9. Eraso-Cadena M, Molina-Guzmán L, Cardona X, Cardona-Arias J, Ríos-Osorio L, Gutiérrez-Builes L. Serological evidence of exposure to some zoonotic microorganisms in cattle and humans with occupational exposure to livestock in Antioquia, Colombia. Cad Saúde Pública. 2018;3:e00193617. https://doi.org/10.1590/0102-311X00193617
10. Tamí I, Tamí-Maury I. Identificación morfológica de Ehrlichia spp. en las plaquetas de pacientes con infección por virus de la inmunodeficiencia humana en Venezuela. Rev Panam Salud Pública. 2004;16:345-59. https://doi.org/10.1590/s1020-49892004001100008
11. Tami I, García F, Tami M, Arcía R. Ehrlichiosis en animales y humanos en Venezuela. Acta Cient Soc Venez Bioanalistas Esp. 1994;3:19-24.
12. Pérez M, Bodor M, Zhang C, Xiong Q, Rikihisa Y. Human infection with Ehrlichia canis accompanied by clinical signs in Venezuela. Ann N Y Acad Sci. 2006;1078:110-17. https://doi.org/10.1196/annals.1374.016
13. Matei I, Estrada-Peña A, Cutler S, Vayssier-Taussat M, Varela-Castro L, Potkonjak A, et al. A review on the eco epidemiology and clinical management of human granulocytic anaplasmosis and its agent in Europe. Parasit Vectors. 2019;12:599. https://doi.org/10.1186/s13071-019-3852-6
14. Wang F, Yan M, Liu A, Chen T, Luo L. The seroprevalence of Anaplasma phagocytophilum in global human populations: A systematic review and meta-analysis. Transbound Emerg Dis. 2020;67:2050-64. https://doi.org/10.1111/tbed.13548
15. Arraga-Alvarado C, Palmar M, Parra O, Salas P. Fine structural characterisation of a Rickettsia-like organism in human platelets from patients with symptoms of ehrlichiosis. J Med Microbiol.1999;48:991-7. https://doi.org/10.1099/00222615-48-11-991
16. Maggi R, Mascarelli P, Havenga L, Naidoo V, Breitschwert E. Co-infection with Anaplasma platys, Bartonella henselae, and Candidatus Mycoplasma haematoparvum in a veterinarian. Parasit Vectors. 2013;6:103. https://doi.org/10.1186/1756-3305-6-103
17. Arraga-Alvarado C, Qurollo B, Parra O, Berrueta M, Hegarty B, Breitschwerdt E. Molecular evidence of Anaplasma platys infection in two women from Venezuela. Am J Trop Med Hyg. 2014;91:1161-5. https://doi.org/10.4269/ajtmh.14-0372
18. Krause P. Human babesiosis. Int J Parasitol. 2019;49:165-74. https://doi.org/10.1016/j.ijpara.2018.11.007
19. Ingram D, Crook T. Rise in babesiosis cases, Pennsylvania, USA, 2005-2018. Emerg Infect Dis. 2020;26:1703-9. https://doi.org/10.3201/eid2608.191293
20. Wood RR, Roberts RW, Kerr SM, Wasden M, Hammer TG, McCreadie JW, et al. Tick-borne pathogens associated with medically important ticks in Alabama: A four-year survey. Vector Borne Zoonotic Dis. 2023;23:57-62. https://doi.org/10.1089/vbz.2022.0038
21. Waked R, Krause PJ. Human babesiosis. Infect Dis Clin North Am. 2022;36:655-70. https://doi.org/10.1016/j.idc.2022.02.009
22. Chen Z, Li H, Gao X, Bian A, Yan H, Kong D, et al. Human babesiosis in China: A systematic review. Parasitol Res. 2019;118:1103-12. https://doi.org/10.1007/s00436-019-06250-9
23. Pérez M, Rikihisa Y, Wen B. Ehrlichia canis-like agent isolated from a man in Venezuela: Antigenic and genetic characterization. J Clin Microbiol. 1996;34:2133-9. https://doi.org/10.1128/jcm.34.9.2133-2139.1996
24. Martínez M, Gutiérrez C, Monger F, Ruíz J, Watts A, Mijares V, et al. Ehrlichia chaffeensis in child, Venezuela. Emerg Infect Dis. 2008;14:519-20. https://doi.org/10.3201/eid1403.061304
25. Añez N, Rojas A, Crisante G, Abelló J, Zambrano C, Quiñonez M. Human babesiosis in western Venezuela. Case reports. Cient Med. 2020;1:1-6. https://doi.org/10.47449/CM.2020.1.1.25
26. Gutiérrez N, Martínez A, Triana A. Identificación microscópica y molecular de ehrlichias en perros del estado Aragua – Venezuela. Salus. 2009;12:197-204.
27. González-Ascanio Y, Vásquez-Franco K. Ehrlichiosis monocítica humana y babesiosis en Venezuela. Rev Med Ris. 2018;24:125-32. https://doi.org.10.22517/25395203.16521
28. Instituto Nacional de Estadística República Bolivariana de Venezuela. Censo de población y vivienda 2014. Fecha de consulta: 25 de octubre de 2024. Disponible en: http://www.ine.gob.ve/documentos/Demografia/CensodePoblacionyVivienda/pdf/bolivar.pdf
29. Peña O. División política del municipio Caroní. Fecha de consulta: 20 de agosto de 2022. Disponible en: https://es.scribd.com/document/259069492/Division-Politica-Del-Municipio-Caroní
30. Méndez-Castellano H. Método Graffar modificado para Venezuela: manual de procedimiento del área de familia. Caracas: FUNDACREDESA; 1982.
31. Arraga-Alvarado C, Montero-Ojeda M, Bernardoni A, Anderson B, Parra O. Ehrlichiosis humana: reporte del primer caso en Venezuela. Invest Clin. 1996;37:35-9.
32. Delmoral J, Traviezo-Valles L, Cárdenas E, Kompalic A. Babesiosis humana por Babesia sp., (Piroplasmida: Babesiidae). Reporte de un caso en el estado Lara. Bol Venez Infectol. 2009;2:34-6.
33. Dulcey L, Therán J, Caltagirone R, Aguas M, Pinto L, Gonzales H. Babesiosis. Reporte de caso clínico en Venezuela. Revisión de literatura. Bol Venez Infectol. 2022;33:92-6.
34. Álvarez D, Corona-González B, Rodríguez-Mallón A, Rodríguez González I, Alfonso P, et al. Ticks and tick-borne diseases in Cuba, half a century of scientific research. Pathogens. 2020;9:616. https://doi.org/10.3390/pathogens9080616
35. Bouchard C, Dibernardo A, Koffi J, Wood H, Leighton P, Lindsay L. Increased risk of tickborne diseases with climate change. Can Commun Dis Rep. 2019;45:83-9. https://doi.org/10.14745/ccdr.v45i04a02
36. Dumic I, Jevtic D, Veselinovic M, Nordstrom C, Jovanovic M, Mogulla V, et al. Human granulocytic anaplasmosis – A systematic review of published cases. Microorganisms. 2022;10:1433. https://doi.org/10.3390/microorganisms10071433
37. Khan R, Bansal A, Astiz M. Co-infection with Babesia and Ehrlichia in New York City. Crit Care Med. 2006;34:A166.
38. Montes-Farah J, De la Vega-Del Risco F, Bello-Espinosa A, Fortich-Salvador A. Coinfección de babesiosis y ehrlichiosis: un caso en Cartagena de Indias, Colombia. Rev Cienc Bioméd. 2020;3:339-45. https://doi.org/10.32997/rcb-2012-3132
39. Yoshinari N, Abrão M, Bonoldi V, Soares C, Madruga C, Scofield A, et al. Coexistence of antibodies to tick-borne agents of babesiosis and Lyme borreliosis in patients from Cotia county, State of São Paulo, Brazil. Mem Inst Oswaldo Cruz. 2003;98:311-8. https://doi.org/10.1590/s0074-02762003000300004
40. Oteo J. Espectro de las enfermedades transmitidas por garrapatas. Rev Pediatr Aten Primaria. 2016;18(Supl. 25):47-51.
41. Ortíz J, Millhouse P, Morillo A, Campoverde L, Kaur A, Wirth M, et al. Babesiosis: Appreciating the pathophysiology and diverse sequela of the infection. Cureus. 2020;12:e11085. https://doi.org/10.7759/cureus.11085
42. Kandhi S, Ghazanfar H, Qureshi Z, Kalangi H, Jyala A, Argüello E. An atypical presentation of a severe case of Anaplasma phagocytophilum. Cureus. 2022;14:e23224. https://doi.org/10.7759/cureus.23224
43. Leikauskas J, Read J, Ketso P, Nichols K, Armstrong P, Kwit N. Anaplasmosis-related fatality in Vermont: A case report. Vector Borne Zoonotic Dis. 2022;22:188-90. https://doi.org/10.1089/vbz.2021.0095
44. Kuriakose K, Pettit A, Schmitz J, Moncayo A, Bloch K. Assessment of risk factors and outcomes of severe ehrlichiosis infection. JAMA Netw Open. 2020;3:e2025577. https://doi.org/10.1001/jamanetworkopen.2020.25577
45. Dantas-Torres F. Biology and ecology of the brown dog tick, Rhipicephalus sanguineus. Parasit Vectors. 2010;3:26. https://doi.org/10.1186/1756-3305-3-26
46. Escárcega A, Luna B, De la Mora A, Jiménez F. Análisis exploratorio de enfermedades rickettsiales transmitidas por garrapatas en perros de Ciudad Juárez, Chihuahua, México. Acta Univ. 2018;28:72-8. https://doi.org/10.15174/au.2018.1678
47. Buczek A, Buczek W, Buczek A, Bartosik K. The potential role of migratory birds in the rapid spread of ticks and tick-borne pathogens in the changing climatic and environmental conditions in Europe. Int J Environ Res Public Health. 2020;17:2117. https://doi.org/10.3390/ijerph17062117
48. Russell A, Prusinski M, Sommer J, O’Connor C, White J, Falco R, et al. Epidemiology and spatial emergence of anaplasmosis, New York, USA, 2010-2018. Emerg Infect Dis. 2021;27:2154-62. https://doi.org/10.3201/eid2708.210133
49. Li H, Zhou Y, Wang W, Guo D, Huang S, Jie S. The clinical characteristics and outcomes of patients with human granulocytic anaplasmosis in China. Int J Infect Dis. 2011;15:859-66. https://doi.org/10.1016/j.ijid.2011.09.008
50. Folkema AM, Holman RC, Dahlgren FS, Cheek JE, McQuiston JH. Epidemiology of ehrlichiosis and anaplasmosis among American Indians in the United States, 2000-2007. Am J Trop Med Hyg. 2012;87:529-37. https://doi.org/10.4269/ajtmh.2012.12-0060
51. Harkirat M, Singh N, Singh N, Singh C, Rath S. Molecular prevalence and risk factors for the occurrence of canine monocytic ehrlichiosis. Vet Med (Praha). 2014;50:129-36. https://doi.org/10.17221/7380-VETMED
52. Huerto-Medina E, Dámaso-Mata B. Factores asociados a la infección por Ehrlichia canis en perros infestados con garrapatas en la ciudad de Huánuco. Rev Peru Med Exp Salud Pública. 2015;32:756-60. https://doi.org/10.17843/rpmesp.2015.324.1769
53. Pérez-Macchi S, Pedrozo R, Bittencourt P, Müller A. Prevalence, molecular characterization, and risk factor analysis of Ehrlichia canis and Anaplasma platys in domestic dogs from Paraguay. Comp Immunol Microbiol Infect Dis. 2019;62:31-9. https://doi.org/10.1016/j.cimid.2018.11.015
54. Harvey N. How old is my dog? Identification of rational groupings in pet dogs based upon normative age-linked processes. Front Vet Sci. 2021;8:643085. https://doi.org/10.3389/fvets.2021.643085
55. Yuasa Y, Tsai Y, Chang C, Hsu T, Chou C. The prevalence of Anaplasma platys and a potential novel Anaplasma species exceed that of Ehrlichia canis in asymptomatic dogs and Rhipicephalus sanguineus in Taiwan. J Vet Med Sci. 2017;79:1494-502. https://doi.org/10.1292/jvms.17-0224
56. Selim A, Almohammed H, Abdelhady A, Alouffi A, Ayed F. Molecular detection and risk factors for Anaplasma platys infections in dogs from Egypt. Parasit Vectors. 2021;14:429. https://doi.org/10.1186/s13071-021-04943-8
57. Kovačević M, Beletić A, Ilić A, Milanović Z, Tyrrell P, Bush J, et al. Molecular and serological prevalence of Anaplasma phagocytophilum, A. platys, Ehrlichia canis, E. chaffeenses, E. ewingii, Borrelia burgdorferi, Babesia canis, B. gibsoni, and B. vogeli among clinically healthy outdoor dogs in Serbia. Vet Parasitol Reg Stud Reports. 2018;14:117-22. https://doi.org/10.1016/j.vprsr.2018.10.001
58. Angelou A, Gelasakis A, Verde N, Pantchev N, Schaper R, Chandrashekar R, et al. Prevalence and risk factors for selected canine vector-borne diseases in Greece. Parasit Vectors. 2019;12:283. https://doi.org/10.1186/s13071-019-3543-3
59. Saleh M, Allen K, Lineberry M, Little S, Reichard M. Ticks infesting in dogs and cats in North America: Biology, geographic distribution, and pathogen transmission. Vet Parasitol. 2021;294:109392. https://doi.org/10.1016/j.vetpar.2021.109392
60. Toepp A, Willardson K, Larson M, Scott B, Johannes A, Senesac R, et al. Frequent exposure to many hunting dogs significantly increases tick exposure. Vector Borne Zoonotic Dis. 2018;18:519-23. https://doi.org/10.1089/vbz.2017.2238
61. Ortíz D, Piche-Ovares M, Romero-Vega L, Wagman J, Troyo A. The impact of deforestation, urbanization, and changing land use patterns on the ecology of mosquito and tick-borne diseases in Central America. Insects. 2022;13:20. https://doi.org/10.3390/insects13010020
62. Bayles B, Allan B. Social-ecological factors determine spatial variation in human incidence of tick-borne ehrlichiosis. Epidemiol Infect. 2014;142:1911-24. https://doi.org/10.1017/S0950268813002951
63. Acosta-Jammet G, Weitzel T, López J, Alvarado D, Abarca K. Prevalence and risk factors of antibodies to Anaplasma spp., in Chile: A household-based cross-sectional study in healthy adults and domestic dogs. Vector Borne Zoonotic Dis. 2020;20:572-79. https://doi.org/10.1089/vbz.2019.2587
64. Cermeño J, Espinoza R, Penna S. Paracoccidioides spp. en excretas de golondrinas migratorias (Tyrannus savana y Progne tapera fusca), procedentes de Sur América. Rev Cient Fac Vet Univ Zulia. 2021;32:1-7. https://doi.org/10.52973/rcfcv-e32083
65. Bogdanka A. Diagnostic evaluation of Ehrlichia canis human infections. Open J Med Microbiol. 2014;4:132-9. https://doi.org/10.4236/ojmm.2014.42015
66. Movilla R, García C, Siebert S, Roura X. Countrywide serological evaluation of canine prevalence for Anaplasma spp., Borrelia burgdorferi (sensu lato), Dirofilaria immitis, and Ehrlichia canis in Mexico. Parasit Vectors. 2016;78:421. https://doi.org/10.1186/s13071-016-1686-z
67. Merino-Charrez O, Badillo-Moreno V, Loredo-Osti J, Barrios-García H, Carvajal-de-la-Fuente V. Molecular detection of Ehrlichia canis and Anaplasma phagocytophilum and hematological changes of infected dogs. Abanico Vet. 2021;11:e119. https://doi.org/10.21929/abavet2021.29
68. Bader J, Nascimento R, Otranto D, Dantas-Torres F. Vector-borne pathogens in dogs from Guatemala, Central América. Vet Parasitol Reg Stud Reports. 2020;22:100468. https://doi.org/10.1016/j.vprsr.2020.100468
69. Alhassan A, Hove P, Sharma B, Matthew-Belmar V, Karasek I, Lanza-Perea M, et al. Molecular detection and characterization of Anaplasma platys and Ehrlichia canis in dogs from the Caribbean. Ticks Tick Borne Dis. 2021;12:101727. https://doi.org/10.1016/j.ttbdis.2021.101727
70. Lee S, Lee H, Park J, Yoon S, Seo H, Noh J, et al. Prevalence of antibodies against Anaplasma spp., Borrelia burgdorferi (sensu lato), Babesia gibsoni, and Ehrlichia spp. in dogs in the Republic of Korea. Ticks Tick Borne Dis. 2020;11:101412. https://doi.org/10.1016/j.ttbdis.2020.101412
71. Ramos R, Ramos C, Araújo F, Oliveira R, Souza I, Pimentel D, et al. Molecular survey and genetic characterization of tick-borne pathogens in dogs in metropolitan Recife (northeastern Brazil). Parasitol Res. 2010;107:1115-20. https://doi.org/10.1007/s00436-010-1979-7
Some similar items:
- Guillermo Terán-Angel, Henk Schallig, Olga Zerpa, Vestalia Rodríguez, Marian Ulrich, Maira Cabrera, The direct agglutination test as an alternative method for the diagnosis of canine and human visceral leishmaniasis , Biomedica: Vol. 27 No. 3 (2007)
- Jorge Miranda, Verónica Contreras, Yesica Negrete, Marcelo B. Labruna, Salim Mattar, Surveillance of Rickettsia sp. infection in capybaras (Hydrochoerus hydrochaeris) a potential model of epidemiological alert in endemic areas , Biomedica: Vol. 31 No. 2 (2011)
- Mariolga Berrizbeitia, Juan Luis Concepcion, Valentina Carzola, Jessicca Rodríguez, Ana Cáceres, Wilfredo Quiñones, Seroprevalence of T. cruzi infection in Canis familiaris, Sucre state, Venezuela , Biomedica: Vol. 33 No. 2 (2013)
- Margaret Paternina-Gómez, Yirys Díaz-Olmos, Luis Enrique Paternina, Eduar Elías Bejarano, High prevalence of infection with Leishmania (Kinetoplastea: Trypanosomatidae) in dogs in northern Colombia , Biomedica: Vol. 33 No. 3 (2013)
- María I. Alvarez, Luz D. Caicedo, Dermatophytes in dogs from Cali, Colombia , Biomedica: Vol. 21 No. 2 (2001)
- Ana Lucía Rodríguez, Beatriz Eugenia Ferro, María Ximena Varona, Mauricio Santafé, Exposure to Leptospira in stray dogs in the city of Cali. , Biomedica: Vol. 24 No. 3 (2004)
- Victoria Rodríguez, Oneida Espinosa, Julio César Carranza, Sofía Duque, Adriana Arévalo, Jairo Alfonso Clavijo, Daniel Alfonso Urrea, Gustavo Adolfo Vallejo, Giardia duodenalis genotypes found in the Instituto Colombiano de Bienestar Familiar day care centers and dogs in Ibagué, Colombia , Biomedica: Vol. 34 No. 2 (2014)
- Jorge Sánchez, Susana Diez, Ricardo Cardona, Pet avoidance in allergy cases: Is it possible to implement it? , Biomedica: Vol. 35 No. 3 (2015)
- Daly Martínez-Ortiz, Marco Torres-Castro, Edgar Koyoc-Cardeña, Karina López, Alonso Panti-May, Iván Rodríguez-Vivas, Adriano Puc, Karla Dzul, Jorge Zavala-Castro, Anuar Medina-Barreiro, Juan Chablé-Santos, Pablo Manrique-Saide, Molecular evidence of Rickettsia typhi infection in dogs from a rural community in Yucatán, México , Biomedica: Vol. 36 (2016): Suplemento 1, Microbiología médica
- Stefany Monsalve, Santiago Rucinque, Luis Polo, Gina Polo, Assessment of the spatial accessibility to the rabies vaccination campaign in Bogotá, Colombia , Biomedica: Vol. 36 No. 3 (2016)
Copyright (c) 2025 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























