Clinical and genetic description of patients with chronic granulomatous disease in a pediatric hospital
Abstract
Introduction. Chronic granulomatous disease is a congenital immune disorder characterized by increased susceptibility to fungal and bacterial infections and dysregulated inflammation. It is caused by defects in the NADPH oxidase and EROS protein.
Objective. To characterize clinically and genetically four patients with chronic granulomatous disease at the Hospital Infantil de México Federico Gómez.
Materials and methods. Patients diagnosed with chronic granulomatous disease by the dihydrorhodamine oxidase technique were molecularly and genetically characterized by measuring NADPH oxidase subunit expression and exome sequencing and analysis. The different clinical variables were obtained from clinical files, and each case was described.
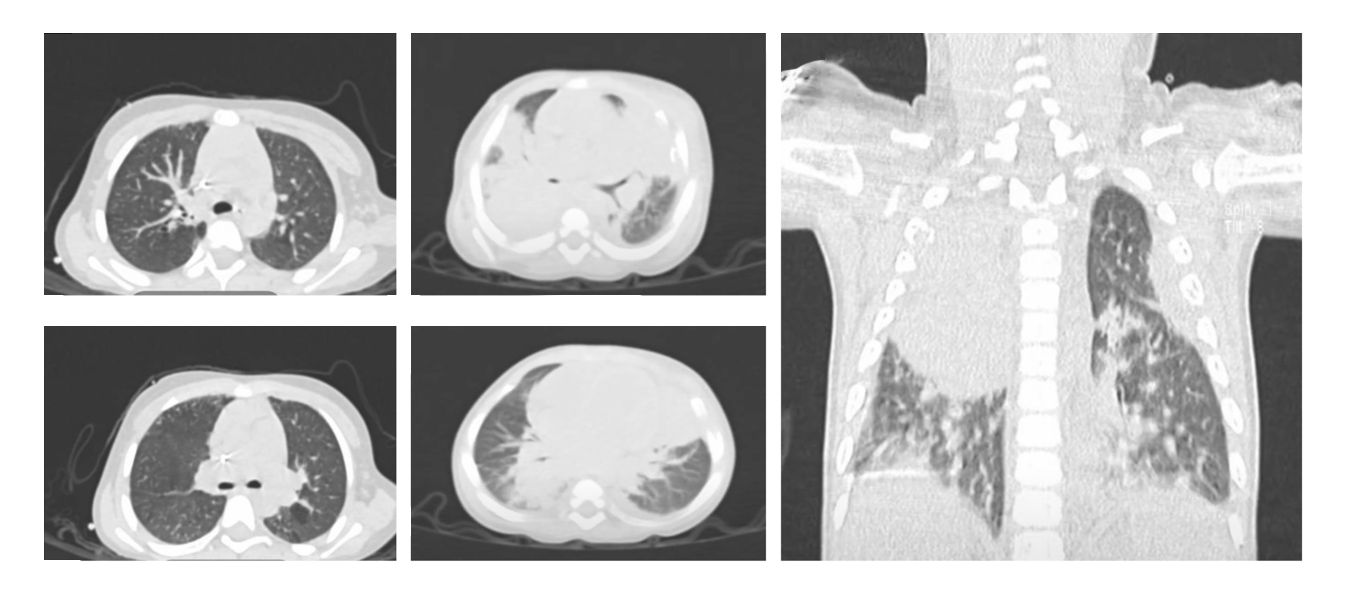
Results. We described four male patients with chronic granulomatous disease: two with pathogenic variants in CYBB, one with CYBB and adjacent genes deleted, and one without p47phox expression. Mothers of the three patients with mutated CYBB were carriers. All three cases with CYBB had severe and recurrent infections in addition to Calmette-Guérin bacillus infection as the initial manifestation. The autosomal recessive case of p47phox deficiency had the mildest clinical presentation. Deleting CYBB and several contiguous genes was associated with a poor prognosis. None of the patients received hematopoietic stem cell transplantation.
Conclusions. Chronic granulomatous disease, secondary to pathogenic variants in CYBB was the most common in these Mexican patients. The carrier mothers should be
followed clinically because of the potential risk of inflammatory, autoimmune, and infectious manifestations. One of the first manifestations was Calmette-Guérin bacillus infection, and in countries such as Mexico, where this vaccine is administered, cases with any type of adverse reaction should be evaluated to rule out chronic granulomatous disease.
Downloads
References
Mortimer PM, Mc Intyre SA, Thomas DC. Beyond the extra respiration of phagocytosis: NADPH oxidase 2 in adaptive immunity and inflammation. Front Immunol. 2021;12. https://doi.org/10.3389/fimmu.2021.733918
Sun B, Zhu Z, Hui X, Sun J, Wang W, Ying W, et al. Variant type X91+ chronic granulomatous disease: Clinical and molecular characterization in a Chinese cohort. J Clin Immunol. 2022;42:1564-79. https://doi.org/10.1007/s10875-022-01324-3
León-Lara X, Rodríguez-D’Cid R, Rioja-Valencia R, Ayala-Alvirde A, Aliaga-Taipe IL, Espinosa-Padilla S, et al. Alteraciones inflamatorias clínicas y moleculares en enfermedad granulomatosa crónica. Rev Alerg Mex. 2021;67. http://doi.org/10.29262/ram.v67i4.784
Thomas DC. How the phagocyte NADPH oxidase regulates innate immunity. Free Radic Biol Med. 2018;125:44-52. https://doi.org/10.1016/j.freeradbiomed.2018.06.011
Li T, Zhou X, Ling Y, Jiang N, Ai J, Wu J, et al. Genetic and clinical profiles of disseminated Bacillus Calmette-Guérin disease and chronic granulomatous disease in China. Front Immunol. 2019;10. https://doi.org/10.3389/fimmu.2019.00073
Abd Elaziz D, EL Hawary R, Meshaal S, Alkady R, Lotfy S, Eldash A, et al. Chronic granulomatous disease: A cohort of 173 patients—10-years single center experience from Egypt. J Clin Immunol. 2023;43:1799-811. https://doi.org/10.1007/s10875-023-01541-4
Yanagimachi M, Kato K, Iguchi A, Sasaki K, Kiyotani C, Koh K, et al. Hematopoietic cell transplantation for chronic granulomatous disease in Japan. Front Immunol. 2020;11. https://doi.org/10.3389/fimmu.2020.01617
Blancas-Galicia L, Santos-Chávez E, Deswarte C, Mignac Q, Medina-Vera I, León-Lara X, et al. Genetic, immunological, and clinical features of the first Mexican cohort of patients with chronic granulomatous disease. J Clin Immunol. 2020;40:475-93. https://doi.org/10.1007/s10875-020-00750-5
González-Valdez MC. Enfermedad granulomatosa crónica (EGC). Experiencia del Hospital Infantil de México en los últimos 25 años (tesis). México, D.F.: Universidad Autónoma de México; 1995.
Gérard B, El Benna J, Alcain F, Gougerot-Pocidalo MA, Grandchamp B, Chollet-Martin S. Characterization of 11 novel mutations in the X-linked chronic granulomatous disease (CYBB gene): Mutations in brief. Hum Mutat. 2001;18:163. https://doi.org/10.1002/humu.1166
Heyworth PG, Curnutte JT, Rae J, Noack D, Roos D, van Koppen E, et al. Hematologically important mutations: X-linked chronic granulomatous disease (second update). Blood Cells Mol Dis. 2001;27:16-26. https://doi.org/10.1006/bcmd.2000.0347
Bourdeaut F, Quartier P, Alkaer G, Fischer A, Casanova J, Blanche S. Propionibacterium acnes chest infections in patients with chronic granulomatous disease: Case reports. Clin Infect Dis. 2002;34:853-4. https://doi.org/10.1086/338875
von Goessel H, Hossle JP, Seger R, Gungor T. Characterization of 17 new cases of X-linked chronic granulomatous disease with seven novel mutations in the CYBB gene. Exp Hematol. 2006;34:528-35. https://doi.org/10.1016/j.exphem.2006.01.005
Kannengiesser C, Gérard B, El Benna J, Henri D, Kroviarski Y, Chollet-Martin S, et al. Molecular epidemiology of chronic granulomatous disease in a series of 80 kindreds: Identification of 31 novel mutations. Hum Mutat. 2008;29:e132-49. https://doi.org/10.1002/humu.20820
Wolach B, Gavrieli R, de Boer M, Gottesman G, Ben-Ari J, Rottem M, et al. Chronic granulomatous disease in Israel: Clinical, functional and molecular studies of 38 patients. Clin Immunol. 2008;129:103-14. https://doi.org/10.1016/j.clim.2008.06.012
Roos D, Kuhns DB, Maddalena A, Roesler J, López JA, Ariga T, et al. Hematologically important mutations: X-linked chronic granulomatous disease (third update). Blood Cells Mol Dis. 2010;45:246-65. https://doi.org/10.1016/j.bcmd.2010.07.012
Bustamante J, Boisson-Dupuis S, Abel L, Casanova J-L. Mendelian susceptibility to mycobacterial disease: Genetic, immunological, and clinical features of inborn errors of IFN-γ immunity. Semin Immunol. 2014;26:454-70. https://doi.org/10.1016/j.smim.2014.09.008
de Oliveira-Junior EB, Prando C, López JA, Arango JC, Buzolin M, Rehder J, et al. Highperformance liquid chromatography under partially denaturing conditions (dHPLC) is a fast and cost-effective method for screening molecular defects: Four novel mutations found in X-linked chronic granulomatous disease. Scand J Immunol. 2012;76:158-66. https://doi.org/10.1111/j.1365-3083.2012.02714.x
Jakobsen MA, Katzenstein TL, Valerius NH, Roos D, Fisker N, Mogensen TH, et al. Genetical analysis of all Danish patients diagnosed with chronic granulomatous disease. Scand J Immunol. 2012;76:505-11. https://doi.org/10.1111/j.1365-3083.2012.02771.x
de Oliveira-Junior EB, Zurro NB, Prando C, Cabral-Marques O, Pereira PVS, Schimke L-F, et al. Clinical and genotypic spectrum of chronic granulomatous disease in 71 Latin American patients: First report from the LASID registry: Chronic granulomatous disease in Latin America. Pediatr Blood Cancer. 2015;62:2101-7. https://doi.org/10.1002/pbc.25674
Conti F, Lugo-Reyes SO, Blancas Galicia L, He J, Aksu G, Borges de Oliveira E Jr, et al. Mycobacterial disease in patients with chronic granulomatous disease: A retrospective analysis of 71 cases. J Allergy Clin Immunol. 2016;138:241-8.e3. https://doi.org/10.1016/j.jaci.2015.11.041
Wu J, Wang W-F, Zhang Y-D, Chen T-X. Clinical features and genetic analysis of 48 patients with chronic granulomatous disease in a single center study from Shanghai, China (2005-2015): New studies and a literature review. J Immunol Res. 2017;2017:1-17. https://doi.org/10.1155/2017/8745254
Boonyawat B, Suksawat Y, Pacharn P, Suwanpakdee P, Traivaree C. X-linked chronic granulomatous disease: Initial presentation with intracranial hemorrhage from vitamin K deficiency in infant. Case Rep Pediatr. 2018;2018:1-6. https://doi.org/10.1155/2018/7041204
Kutluğ Ş, Şensoy G, Birinci A, Saraymen B, Köker Y, Yιldιran A. Seven chronic granulomatous disease cases in a single-center experience and a review of the literature. Asian Pac J Allergy Immunol. 2018;36:35-41. https://doi.org/10.12932/ap0859
Ariga T, Sakiyama Y, Furuta H, Matsumoto S. Molecular genetic studies of two families with X-linked chronic granulomatous disease: Mutation analysis and definitive determination of carrier status in patients’ sisters. Eur J Haematol. 1994;52:99-102. https://doi.org/10.1111/j.1600-0609.1994.tb01293.x
Patiño PJ, Pérez JE, López JA, Condino-Neto A, Grumach AS, Botero JH, et al. Molecular analysis of chronic granulomatous disease caused by defects in gp91-phox. Hum Mutat. 1999;1:29-37. https://doi.org/10.1002/(sici)1098-1004(1999)13:1<29::aid-humu3>3.0.co;2-x
Agudelo-Flórez P, López JA, Redher J, Carneiro-Sampaio MMS, Costa-Carvalho BT, Grumach AS, et al. The use of reverse transcription-PCR for the diagnosis of X-linked chronic granulomatous disease. Braz J Med Biol Res. 2004;37:625-34. https://doi.org/10.1590/s0100-879x2004000500001
Kuhns DB, Alvord WG, Heller T, Feld JJ, Pike KM, Marciano BE, et al. Residual NADPH oxidase and survival in chronic granulomatous disease. N Engl J Med. 2010;363:2600-10. https://doi.org/10.1056/nejmoa1007097
Labrosse R, Abou-Diab J, Blincoe A, Cros G, Luu TM, Deslandres C, et al. Very early-onset inflammatory manifestations of X-linked chronic granulomatous disease. Front Immunol 2017;8. https://doi.org/10.3389/fimmu.2017.01167
Kohn DB, Booth C, Kang EM, Pai S-Y, Shaw KL, Santilli G, et al. Lentiviral gene therapy for X-linked chronic granulomatous disease. Nat Med. 2020;26:200-6. https://doi.org/10.1038/s41591-019-0735-5
Chiu TL-H, Leung D, Chan K-W, Yeung HM, Wong C-Y, Mao H, et al. Phenomic analysis of chronic granulomatous disease reveals more severe integumentary infections in X-linked compared with autosomal recessive chronic granulomatous disease. Front Immunol. 2022;12. https://doi.org/10.3389/fimmu.2021.803763
Henrickson SE, Jongco AM, Thomsen KF, Garabedian EK, Thomsen IP. Noninfectious manifestations and complications of chronic granulomatous disease. J Pediatric Infect Dis Soc. 2018;7(Suppl.1):S18-24. https://doi.org/10.1093/jpids/piy014
Lhomme F, Peyrard T, Babinet J, Abou-Chahla W, Durieu I, Moshous D, et al. Chronic granulomatous disease with the McLeod phenotype: A French national retrospective case series. J Clin Immunol. 2020;40:752-62. https://doi.org/10.1007/s10875-020-00791-w
Scheffler-Mendoza SC, Yamazaki-Nakashimada MA, Olaya-Vargas A, Morin-Contreras A, Juárez-Echenique JC, Alcántara-Ortigoza MA, et al. Successful stem cell transplantation in a child with chronic granulomatous disease associated with contiguous gene deletion syndrome and complicated by macrophage activation syndrome. Clin Immunol. 2014;154:112-5. https://doi.org/10.1016/j.clim.2014.07.004
Some similar items:
- Oscar G. Gómez, Attenuated Salmonella vaccine as a vector for heterologous antigens , Biomedica: Vol. 20 No. 2 (2000)
- Ingrid García, Fernando De la Hoz, Yolima Reyes, Pablo Montoya, Martha Inírida Guerrero, Clara Inés León, Respiratory syntomatic prevalence, infection and tuberculosis disease and associated factors: population-based study. , Biomedica: Vol. 24 (2004): Suplemento 1
- Blanca I. Restrepo, New tools for detection of latent tuberculosis. , Biomedica: Vol. 24 (2004): Suplemento 1
Copyright (c) 2024 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























