In vitro evaluation of the pathogenicity of fungi isolated from the Urabá region (Antioquia, Colombia) against Aedes aegypti larvae
Abstract
Introduction. Aedes aegypti is an important vector of arboviral diseases like dengue among others. Traditional control strategies, such as the use of insecticides, have lost effectiveness due to the emergence of resistance in mosquito populations. Biological control and fungi applied for biocontrol are presented as viable and ecological alternatives.
Objective. To evaluate in vitro pathogenicity of Trichoderma sp. isolates obtained from Urabá (Antioquia) on larvae of Ae. aegypti, and to determine the mean lethal concentration and mean lethal time of the most pathogenic isolate.
Materials and methods. Using the sentinel larvae method of Ae. aegypti, fungi were isolated from water bodies in the Urabá region (Antioquia). The isolates were characterized morphologically and molecularly to determine their taxonomic identity. Pathogenicity tests were performed in vitro on Ae. aegypti larvae in the L2/L3 stages. Subsequently, a strain was selected to establish its mean lethal concentration and mean lethal time.
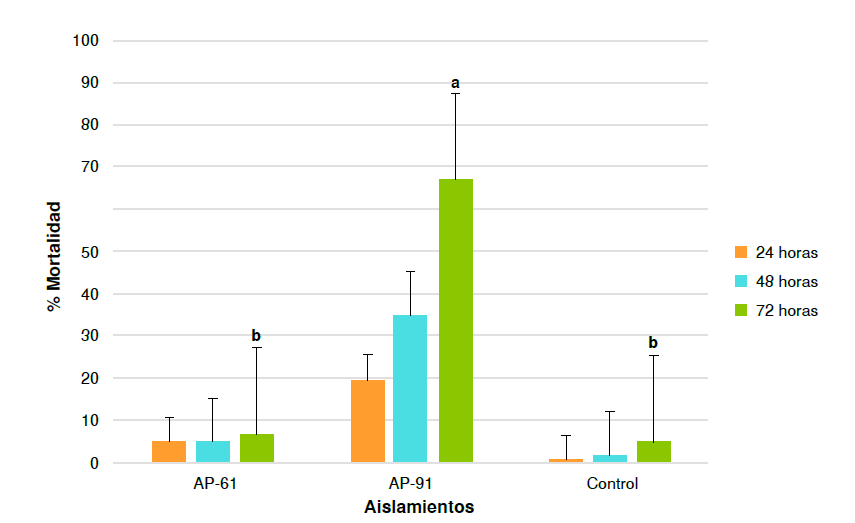
Results. Trichoderma sp. strain AP-91 caused high mortality in larval populations of Ae. aegypti. We estimated a mean lethal concentration of 1.8 × 107 conidia/ml and a mean lethal time of 20.67 hours.
Conclusion. The strain AP-91 showed potential for its use as biological control of Ae. aegypti, making it a suitable candidate for scale-up cultures applied to integrated vector management. This research suggests exploring compounds and enzymes produced by the AP-91 strain to understand better its pathogenicity.
Downloads
References
Powell JR, Tabachnick WJ. History of domestication and spread of Aedes aegypti -- a review. Mem Inst Oswaldo Cruz. 2013;108 (Supl. 1):S11-7. https://doi.org/10.1590/0074-0276130395
Padilla JC, Lizarazo FE, Murillo OL, Mendigaña FA, Pachón E, Vera MJ. Epidemiología de las principales enfermedades transmitidas por vectores en Colombia, 1990-2016. Biomedica. 2017;37(Supl. 2):S27-40. https://doi.org/10.7705/biomedica.v37i0.3769
Bliman PA. A feedback control perspective on biological control of dengue vectors by Wolbachia infection. Eur J Control. 2021;59:188-206.
Rodrigues-Alves ML, Melo-Júnior OA de O, Silveira P, Mariano RM da S, Leite JC, Santos TAP, et al. Historical perspective and biotechnological trends to block arboviruses transmission by controlling Aedes aegypti mosquitos using different approaches. Front Med (Lausanne). 2020:7:275. https://doi.org/10.3389/fmed.2020.00275
Maestre-Serrano R, Gomez-Camargo D, Ponce-Garcia G, Flores AE. Susceptibility to insecticides and resistance mechanisms in Aedes aegypti from the Colombian Caribbean Region. Pestic Biochem Physiol. 2014;116:63-73. https://doi.org/10.1016/j.pestbp.2014.09.014
Conde M, Orjuela LI, Castellanos CA, Herrera-Varela M, Licastro S, Quiñones ML. Evaluación de la susceptibilidad a insecticidas en poblaciones de Aedes aegypti (Diptera: Culicidae) del departamento de Caldas, Colombia, en los años 2007 y 2011. Biomedica. 2015;35:43-52. https://doi.org/10.7705/biomedica.v35i1.2367
Grisales N, Poupardin R, Gomez S, Fonseca-Gonzalez I, Ranson H, Lenhart A. Temephos resistance in Aedes aegypti in Colombia compromises dengue vector control. PLoS Negl Trop Dis. 2013;7:e2438. https://doi.org/10.1371/journal.pntd.0002438
Benelli G, Jeffries CL, Walker T. Biological control of mosquito vectors: Past, present, and future. Insects. 2016;7:52. https://doi.org/10.3390/insects7040052
Rueda ME, Tavares I, López CC, García J. Leptolegnia chapmanii como alternativa biológica para el control de Aedes aegypti. Biomédica. 2019;39:798-810. https://doi.org/10.7705/biomedica.4598
Evans HC, Elliot SL, Barreto RW. Entomopathogenic fungi and their potential for the management of Aedes aegypti (Diptera: Culicidae) in the Americas. Mem Inst Oswaldo Cruz. 2018;113:206-14. https://doi.org/10.1590/0074-02760170369
Gutiérrez AC, Rueda Páramo ME, Falvo ML, López Lastra CC, García JJ. Leptolegnia chapmanii (Straminipila: Peronosporomycetes) as a future biorational tool for the control of Aedes aegypti (L.). Acta Trop. 2017;169:112-8. https://doi.org/10.1016/j.actatropica.2017.01.021
Tawidian P, Rhodes VL, Michel K. Mosquito-fungus interactions and antifungal immunity. Insect Biochem Mol Biol. 2019;111:103182. https://doi.org/10.1016/j.ibmb.2019.103182
Shen D, Nyawira KT, Xia A. New discoveries and applications of mosquito fungal pathogens. Curr Opin Insect Sci. 2020;40:111-6. https://doi.org/10.1016/j.cois.2020.05.003
Zuharah WF, Rohaiyu MR, Azmi WA, Nagao H. Pathogenicity of entomopathogenic fungus, Metarhizium anisopliae MET-GRA4 isolate on dengue vectors, Aedes albopictus and Aedes aegypti mosquito larvae (Diptera: Culicidae). J Asia Pac Entomol. 2021;24:24-9. https://doi.org/10.1016/j.aspen.2021.04.008
da Silveira AA, Andrade JSP, Guissoni ACP, da Costa AC, de Carvalho e Silva A, da Silva HG, et al. Larvicidal potential of cell wall degrading enzymes from Trichoderma asperellum against Aedes aegypti (Diptera: Culicidae). Biotechnol Prog. 2021;37:e3182. https://doi.org/doi/10.1002/btpr.3182
Montalva C, dos Santos K, Collier K, Rocha LFN, Fernandes ÉKK, Castrillo LA, et al. First report of Leptolegnia chapmanii (Peronosporomycetes: Saprolegniales) affecting mosquitoes in central Brazil. J Invertebr Pathol. 2016;136:109-16. https://doi.org/10.1016/j.jip.2016.03.012
Rueda Páramo ME. Aporte al conocimiento de la biología básica y aplicada de Leptolegnia chapmanii (Straminipila, Saprolegniales) como agente de control biológico de Aedes aegypti (Diptera, Culicidae). [Tesis doctoral]. La Plata: Universidad Nacional de La Plata; 2015.
Domsch KH, Gams W, Anderson TH. Compendium of Soil Fungi. Vols. 1-2. London: Academic Press; 1980.
Barnett HL, Hunter BB. Illustrated Genera of Imperfect Fungi. 4th Edition. London: Collier Macmillan Publishers; 1987.
Miranda E, Sandoval L. Desarrollo de un método rápido y sencillo para el aislamiento de DNA de especies fúngicas que afectan el arroz y el tabaco. Fitosanidad. 2000;4:107-8.
White TJ, Bruns T, Lee S, Taylor J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. En: Innis MA, Gelfand DH, Sninsky JJ, White TJ, editores. PCR Protocols: A Guide to Methods and Applications. San Diego (CA): Academic Press, Inc.; 1990. p. 315-22. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
Abarenkov K, Henrik Nilsson R, l-Henrik Larsson K, Taylor AF, May TW, Guldberg Frøslev T, et al. The UNITE database for molecular identification and taxonomic communication of fungi and other eukaryotes: sequences, taxa and classifications reconsidered. Nucleic Acids Res. 2024;52:791-7. https://doi.org/10.1093/nar/gkad1039
Vélez APE, Posada FFJ, Marín MP, González GMT, Osorio VE, Bustillo PAE. Técnicas para el control de calidad de formulaciones de hongos entomopatógenos. Manizales: CENICAFE, 1997. p. 37.
CENAPRECE. Guía para la Instalación y Mantenimiento del Insectario de Aedes aegypti. México D. F.: Secretaría de Salud - Gobierno de México; 2015. p. 31.
Organización Mundial de la Salud. Manual for monitoring insecticide resistance in mosquito vectors and selecting appropriate interventions. Ginebra: OMS; 2022. Fecha de consulta: 18 de octubre de 2023. Disponible en: https://iris.who.int/bitstream/handle/10665/356964/9789240051089-eng.pdf?sequence=1
De Mendiburu F. Agricolae. CRAN R; 2019. Fecha de consulta: 18 de octubre de 2023. Disponible en: https://cran.r-project.org/web/packages/agricolae/index.html
Wickham H, Francois R, Henry L, Müller K, Vaughan D. Dplyr: A grammar of data manipulation. 2023. Fecha de consulta: 16 de octubre de 2023. Disponible en: https://CRAN.R-project.org/package=dplyr
Wickham H. ggplot2: Elegant graphics for data analysis. New York: Springer-Verlag; 2016. Fecha de consulta: 30 de septiembre de 2023. Disponible en: https://ggplot2.tidyverse.org
Hlina BL, Birceanu O, Robinson CS, Dhiyebi H, Wilkie MP. The relationship between thermal physiology and lampricide sensitivity in larval sea lamprey (Petromyzon marinus). J Great Lakes Res. 2021;47 (Suppl. 1): S272-84.
Raja HA, Miller AN, Pearce CJ, Oberlies NH. Fungal identification using molecular tools: A primer for the natural products research community. J Nat Prod. 2017;80:756-70. https://doi.org/10.1021/acs.jnatprod.6b01085
Rueda-Páramo ME, López-Lastra C. Unmasking lethal potential: investigating Leptolegnia chapmanii’s virulence against Aedes aegypti larvae. Biocontrol Sci Technol. 2023;33:1210-4. https://doi.org/10.1080/09583157.2023.2294218
Chen CM, Hsieh HJ, Hu BY, Fu CH. Mosquito-killing water molds isolated from soil samples collected in Taiwan. Pedobiologia (Jena). 2005;49:585-9.
Govindarajan M, Jebanesan A, Reetha D. Larvicidal effect of extracellular secondary metabolites of different fungi against the mosquito, Culex quinquefasciatus Say. Trop Biomed. 2005;22:1-3.
Pereira E da S, Sarquis MI de M, Ferreira-Keppler RL, Hamada N, Alencar YB. Filamentous fungi associated with mosquito larvae (Diptera: Culicidae) in municipalities of the Brazilian Amazon. Neotrop Entomol. 2009;38:352-9. https://doi.org/10.1590/S1519-566X2009000300009
Vázquez-Martínez MG, Rodríguez-Meneses A, Rodríguez AD, Rodríguez MH. Lethal effects of Gliocladium virens, Beauveria bassiana and Metarhizium anisopliae on the malaria vector Anopheles albimanus (Diptera: Culicidae). Biocontrol Sci Technol. 2013;23:1098-109. https://doi.org/10.1080/09583157.2013.822470
Sundaravadivelan C, Padmanabhan MN. Effect of mycosynthesized silver nanoparticles from filtrate of Trichoderma harzianum against larvae and pupa of dengue vector Aedes aegypti L. Environ Sci Pollut Res Int. 2014;21:4624-33. https://doi.org/10.1007/s11356-013-2358-6
Podder D, Ghosh SKr. A new application of Trichoderma asperellum as an anopheline larvicide for ecofriendly management in medical science. Sci Rep. 2019;9:1108. https://doi.org/10.1038/s41598-018-37108-2
Singh G, Prakash S. Virulency of novel nanolarvicide from Trichoderma atroviride against Aedes aegypti (Linn.): a CLSM analysis. Environ Sci Pollut Res Int. 2015;22:12559-65. https://doi.org/10.1007/s11356-015-4531-6
Cisneros-Vázquez LA, Penilla-Navarro RP, Rodríguez AD, Ordóñez-González JG, Valdez-Delgado KM, Danis-Lozano R, et al. Entomopathogenic fungi for the control of larvae and adults of Aedes aegypti in Mexico. Salud Publica Mex. 2023;65:144-50.
Alcalde Mosqueira JG, Roldán Rodríguez JE, Saravia Cueva V del P, Collantes Silva L. Efecto biocida de diferentes concentraciones de Metarhizium anisopliae CCB-LE302 y Beauveria Bassiana CCB-LE265 sobre larvas III de Aedes aegypti. UCV-Scientia. 2014;6:33-41.
Falvo ML. Selección de aislamientos nativos de Metarhizium anisopliae como potenciales agentes de control biológico de Aedes aegypti (Diptera, Culicidae) [Tesis doctoral]. La plata: Universidad Nacional de La Plata; 2019.
Argüelles Palomino D. Efecto de Beauveria bassiana (Hypocreales: Cordycipitaceae) sobre Aedes aegypti (Díptera: Culicidae) en condiciones de laboratorio y otras especies de culícidos en condiciones de campo [Tesis de licenciatura]. Puebla de Zaragoza: Benemérita Universidad Autónoma de Puebla; 2016.
Restrepo B, Lopera T. Estudio seroepidemiologíco de dengue en la región del Urabá Antioqueño-Colombia. Infectio. 2004;8:255-62.
SIVIGILA. Boletín Epidemiológico Semanal BES - Semana Epidemiológica 11 - 10 al 16 de marzo de 2024. Bogotá, D. C.: Instituto Nacional de Salud; 2024.
Some similar items:
- Jorge R. Rey, Philip Lounibos, Ecology of Aedes aegypti and Aedes albopictus in the Americas and disease transmission , Biomedica: Vol. 35 No. 2 (2015)
- Marcela Conde, Lorena I. Orjuela, Cesar Augusto Castellanos, Manuela Herrera-Varela, Susana Licastro, Martha L. Quiñones, Insecticide susceptibility evaluation in Aedes aegypti populations of Caldas, Colombia, in 2007 and 2011 , Biomedica: Vol. 35 No. 1 (2015)
- Clara Beatriz Ocampo, Gloria I. Giraldo Calderon, Mauricio Perez, Carlos A. Morales, Evaluation of the triflumuron and the mixture of Bacillus thuringiensis plus Bacillus sphaericus for control of the immature stages of Aedes aegypti and Culex quinquefasciatus (Diptera: Culicidae) in catch basins , Biomedica: Vol. 28 No. 2 (2008)
- María Elena Cuéllar-Jiménez, Olga Lucía Velásquez-Escobar, Ranulfo González-Obando, Carlos Andrés Morales-Reichmann, Detection of Aedes albopictus (Skuse) (Diptera: Culicidae) in the city of Cali, Valle del Cauca, Colombia , Biomedica: Vol. 27 No. 2 (2007)
- Celeny Ortiz, Guillermo L. Rúa-Uribe, Carlos A. Rojas, Knowledge, practices and entomological aspects of dengue in Medellín, Colombia: A comparative study of neighborhoods with high and low incidence , Biomedica: Vol. 38 No. Sup. 2 (2018): Suplemento 2, Medicina tropical
- César A. Rodriguez, Diana C. Gallego, José A. González, Paola A. Luna, Mitzilene Navarro, Germán A. Ramírez, Mosquitoe breeding sites and fauna ln Armenia's urban area aiier the January 25, 1999, earthquake , Biomedica: Vol. 21 No. 2 (2001)
- Wilber Gómez-Vargas, Kelly Valencia-Jiménez, Guillermo Correa-Londoño, Faiber Jaramillo-Yepes, Novel larvicide tablets of Bacillus thuringiensis var. israelensis: Assessment of larvicidal effect on Aedes aegypti (Diptera: Culicidae) in Colombia , Biomedica: Vol. 38 No. Sup. 2 (2018): Suplemento 2, Medicina tropical
- José Joaquín Carvajal, Nildimar Alves Honorio, Silvia Patricia Díaz, Edinso Rafael Ruiz, Jimmy Asprilla, Susanne Ardila, Gabriel Parra-Henao, Detection of Aedes albopictus (Skuse) (Diptera: Culicidae) in the municipality of Istmina, Chocó, Colombia , Biomedica: Vol. 36 No. 3 (2016)
- Mabel Carabalí, Clara Beatriz Ocampo, María Eugenia Toledo, Lyda Osorio, Mass communication of dengue surveillance data: effect of an intervention in Guadalajara de Buga, Colombia , Biomedica: Vol. 33 (2013): Suplemento 1, Fiebres hemorrágicas
- Sandy Milena Caldera, María Cristina Jaramillo, Suljey Cochero, Alveiro Pérez-Doria, Eduar Elías Bejarano, Genetic differences between populations of Aedes aegypti from municipalities in northern Colombia, with high and low dengue incidence , Biomedica: Vol. 33 (2013): Suplemento 1, Fiebres hemorrágicas
Copyright (c) 2024 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























