Strong antibody reactivity to HIV-1 synthetic peptides in seropositive indigenous Warao people
Abstract
Introduction. Previous studies have described an epidemic of HIV-1 in the indigenous Warao population living in the Orinoco Delta, Venezuela. The Warao face extraordinary challenges amid of their ongoing HIV-1 epidemic, the highest reported HIV-1 prevalence in indigenous groups (9.6%) in South America.
Objective. To investigate the antibody reactivity to HIV-1 synthetic peptides in seropositive individuals, with a particular focus on the indigenous Warao population from Venezuela.
Materials and methods. The HIV-1 Pol region from infected patients’ isolates was amplified, sequenced, and analyzed using phylogenetic tools. Custom-designed synthetic peptides were derived from conserved regions of HIV-1 glycoproteins 41 and 120, based on reference sequences. Multiple antigen blot assays were used to evaluate the presence of antibodies against synthetic peptides.
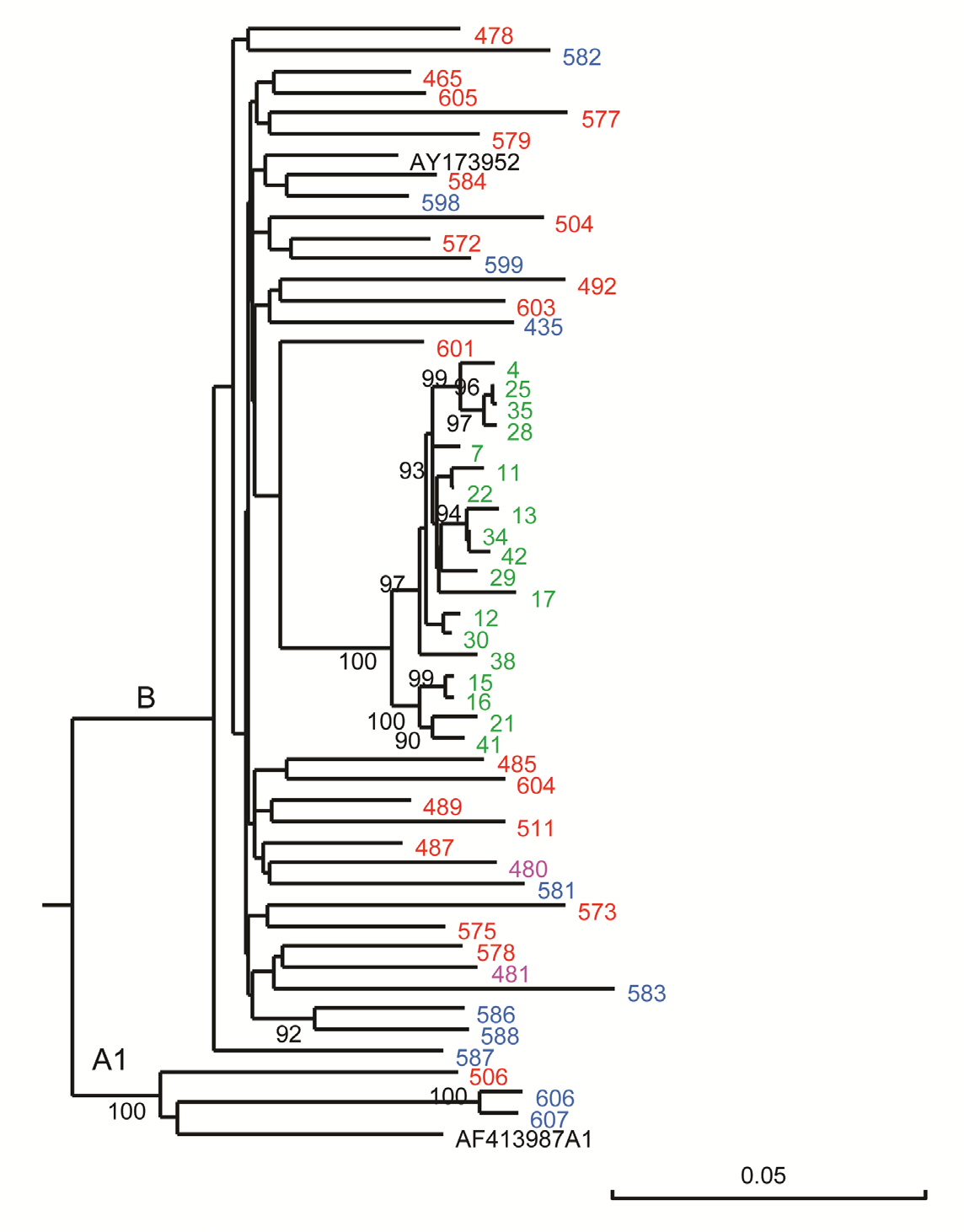
Results. The most frequent HIV-1 subtype was B, the most common in Venezuela, although some individuals were infected with subtype A1. Distinct patterns of reactivity to synthetic peptides were observed between the sera of the general population and the Warao population; the sera of the latter exhibited a high intensity of peptide recognition.
Conclusions. The use of synthetic peptides, coupled with the robust performance of multiple antigen blot assays, enriches our understanding of antibody responses in different HIV-1-infected populations.
Downloads
References
1. World Health Organization. Global health sector strategy on HIV 2016-2021 towards ending aids. Geneva: WHO; 2016. Accessed on: May 1st, 2024. Available at: http://apps.who.int/iris/bitstream/10665/246178/1/WHO-HIV-2016.05-eng.pdf
2. Russell NK, Nazar K, Del Pino S, Alonso Gonzalez M, Díaz Bermúdez XP, Ravasi G. HIV, syphilis, and viral hepatitis among Latin American indigenous peoples and Afro A systematic review. Rev Panam Salud Pública. 2019;43:e17. https://doi.org/10.26633/RPSP.2019.17
3. Villalba J, Bello G, Maes M, Sulbaran Y, Garzaro D, Loureiro CL, et al. HIV-1 epidemic in Warao Amerindians from Venezuela. AIDS. 2013;27:1783-91. https://doi.org/10.1089/AID.2015.0155
4. Maes M, Verhagen LM, Ortega D, Sánchez GL, Segovia Y, del Nogal B, et al. Influence of bacille Calmette-Guérin on tuberculin skin testing in Venezuelan Amerindians in high tuberculosis burden areas. J Infect Dev Ctries. 2014;8:176-83. https://doi.org/10.3855/jidc.3297
5. Rangel HR, Bello G, Villalba JA, Sulbaran YF, Garzaro D, Maes M, et al. The evolving HIV-1 epidemic in Warao Amerindians is dominated by an extremely high frequency of CXCR4-utilizing strains. AIDS Res Hum Retroviruses. 2015;31:1265-8. https://doi.org/10.1089/AID.2015.0155
6. Yandrapally S, Mohareer K, Arekuti G, Vadankula GR, Banerjee S. HIV co-receptortropism: Cellular and molecular events behind the enigmatic co-receptor switching. Crit Rev Microbiol. 2021;47:499-516. https://doi.org/10.1080/1040841X.2021.1902941
7. Cretich M, Gori A, D’Annessa I, Chiari M, Colombo G. Peptides for infectious diseases: From probe design to diagnostic microarrays. Antibodies (Basel). 2019;8:23. https://doi.org/10.3390/antib8010023
8. Chico M, Sandoval C, Guevara A, Calvopiña M, Cooper PJ, Reed SG, et al. Chagas disease in Ecuador: Evidence for disease transmission in an indigenous population in the Amazon region. Mem Inst Oswaldo Cruz. 1997;92:317-20. https://doi.org/10.1590/s0074-02761997000300002
9. Gabbai AA, Bordin JO, Vieira-Filho JP, Kuroda A, Oliveira AS, Cruz MV, et al. Selectivity of human T lymphotropic virus type-1 (HTLV-1) and HTLV-2 infection among different populations in Brazil. Am J Trop Med Hyg. 1993;49:664-71. https://doi.org/10.4269/ajtmh.1993.49.664
10. Araújo Z, de Waard JH, Camargo M, López-Ramos JE, de Larrea CF, Vanegas M, et al. Diagnostic potential of the serological response to synthetic peptides from Mycobacterium tuberculosis antigens for discrimination between active and latent tuberculosis infections. Int J Pept Res Ther. 2022;28:98. https://doi.org/10.1007/s10989-022-10392-3
11. Alcaro MC, Peroni E, Rovero P, Papini AM. Synthetic peptides in the diagnosis of HIV infection. Curr Protein Pept Sci. 2003;4:285-90. https://doi.org/10.2174/1389203033487117
12. Borras-Cuesta F, Fedon Y, Petit-Camurdan A. Enhancement of peptide immunogenicity by linear polymerization. Eur J Immunol. 1988;18:199-202. https://doi.org/10.1002/eji.1830180203
13. Rangel HR, Garzaro D, Gutiérrez CR, Vásquez L, Guillen G, Torres JR, Pujol FH. HIV diversity in Venezuela: Predominance of HIV type 1 subtype B and genomic characterization of non-B variants. AIDS Res Hum Retroviruses. 2009;25:347-50. https://doi.org/10.1089/aid.2008.0241
14. Noya O, Alarcon de Noya B. The multiple antigen blot assay (MABA): A simple immunoenzymatic technique for simultaneous screening of multiple antigens. Immunol. Letters. 1998;63:53-6. https://doi.org/10.1016/s0165-2478(98)00055-8
15. Gauna A, Losada S, Lorenzo M, Toledo M, Bermúdez H, D’Angelo P, et al. Use of synthetic peptides and multiple antigen blot assay in the immunodiagnosis of hepatitis C virus infection. Viral Immunol. 2018;31:568-74. https://doi.org/10.1089/vim.2018.0023
16. Dean AG, Arner TG, Sunki GG, Friedman R, Lantinga M, Sangam S, et al. Epi Info™, a database and statistics program for public health professionals. Atlanta: CDC; 2011.
17. Liang Y, Lin H, Dzakah EE, Tang S. Influence of combination antiretroviral therapy on HIV-1 serological responses and their implications: A systematic review and meta-analysis. Front Immunol. 2022;13:844023. https://doi.org/10.3389/fimmu.2022.844023
18. Rangel HR, Garzaro D, Rodríguez AK, Ramírez AH, Ameli G, Del Rosario Gutiérrez C, et al. Deletion, insertion, and stop codon mutations in Vif genes of HIV-1 infecting slow progressor patients. J Infect Dev Ctries. 2009;3:531-8. https://doi.org/10.3855/jidc.471 19. Wang B. Viral factors in non-progression. Front Immunol. 2013;4:355. https://doi.org/10.3389/fimmu.2013.00355
20. Zaunders J, van Bockel D. Innate and adaptive immunity in long-term non-progression in HIV disease. Front Immunol. 2013;4:95. https://doi.org/10.3389/fimmu.2013.00095
21. Araújo Z, Palacios A, Biomon R, Rivas-Santiago B, Serrano CJ, Enciso-Moreno L, et al. Concordance between IFN-γ gene +874 A/T polymorphism and interferon-γ expression in a TB-endemic indigenous setting. Rev Soc Bras Med Trop. 2017;50:199-207. https://doi.org/10.1590/0037-8682-0398-2016
22. Araújo Z, Camargo M, Moreno Pérez DA, Wide A, Pacheco D, Díaz Arévalo D, et al. Differential NRAMP1 gene’s D543N genotype frequency: Increased risk of contracting tuberculosis among Venezuelan populations. Hum Immunol. 2023;84:484-91. https://doi.org/10.1016/j.humimm.2023.06.003
23. Verhagen LM, Hermsen M, Rivera-Olivero I, Sisco MC, Pinelli E, Hermans PW, et al. Stunting correlates with high salivary and serum antibody levels after 13-valent pneumococcal conjugate vaccination of Venezuelan Amerindian children. Vaccine. 2016;34:2312-20. https://doi.org/10.1016/j.vaccine.2016.03.066
Some similar items:
- Elsa Nieves, Néstor Villarreal, Maritza Rondón, Mireya Sánchez, José Carrero, Evaluation of knowledge and practice on tegumentary leishmaniasis in an endemic area of Venezuela , Biomedica: Vol. 28 No. 3 (2008)
- Olga Serrano, Florencio Mendoza, Benny Suárez, Ana Soto, Seroepidemiology of Chagas disease in two rural populations in the municipality of Costa de Oro, at Aragua State, northern Venezuela , Biomedica: Vol. 28 No. 1 (2008)
- Guillermo Terán-Ángel, Vestalia Rodríguez, Rosilved Silva, Olga Zerpa, Henk Schallig, Marian Ulrich, Maira Cabrera, Herramientas no invasivas en Venezuela: comparación entre las pruebas inmunoserológicas DAT, rK26 y rK39 en el diagnóstico de leishmaniasis visceral , Biomedica: Vol. 30 No. 1 (2010)
- Guillermo Terán-Angel, Henk Schallig, Olga Zerpa, Vestalia Rodríguez, Marian Ulrich, Maira Cabrera, The direct agglutination test as an alternative method for the diagnosis of canine and human visceral leishmaniasis , Biomedica: Vol. 27 No. 3 (2007)
- Elsa Nieves, Neudo Buelvas, Maritza Rondón, Néstor González, The salivary glands of two sand fly vectors of Leishmania: Lutzomyia migonei (França) and Lutzomyia ovallesi (Ortiz) (Diptera: Psychodidae) , Biomedica: Vol. 30 No. 3 (2010)
- Mariolga Berrizbeitia, Juan Luis Concepcion, Valentina Carzola, Jessicca Rodríguez, Ana Cáceres, Wilfredo Quiñones, Seroprevalence of T. cruzi infection in Canis familiaris, Sucre state, Venezuela , Biomedica: Vol. 33 No. 2 (2013)
- Héctor de Lima, José Carrero, Armando Rodríguez, Zoraya de Guglielmo, Noris Rodríguez, Trypanosomatidae of public health importance occurring in wild and synanthropic animals of rural Venezuela. , Biomedica: Vol. 26 No. 1 (2006)
- Greta Rodríguez-Arroyo, Irene Paradisi, Merlyn Vívenes-Lugo, Dinorah Castro-Guerra, Álvaro Rodríguez-Larralde, LEP, LDLR and APOA4 gene polymorphisms and their relationship with the risk of overweight, obesity and chronic diseases in adults of the State of Sucre, Venezuela , Biomedica: Vol. 36 No. 1 (2016)
- Manuel Toquero, Antonio Morocoima, Elizabeth Ferrer, Seroprevalence and risk factors of cysticercosis in two rural communities in Anzoátegui state, Venezuela , Biomedica: Vol. 37 No. Sup.1 (2017): Suplemento 1, Alteraciones del sistema nervioso
- Noris García-Jordán, Mariolga Berrizbeitia, Juan Luis Concepción, Elis Aldana, Ana Cáceres, Wilfredo Quiñones, Entomological study of Trypanosoma cruzi vectors in the rural communities of Sucre state, Venezuela , Biomedica: Vol. 35 No. 2 (2015)
Copyright (c) 2025 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























