Recommendations on vaccination in children and adolescents with inborn errors of immunity according to the expanded Colombian immunization program
Abstract
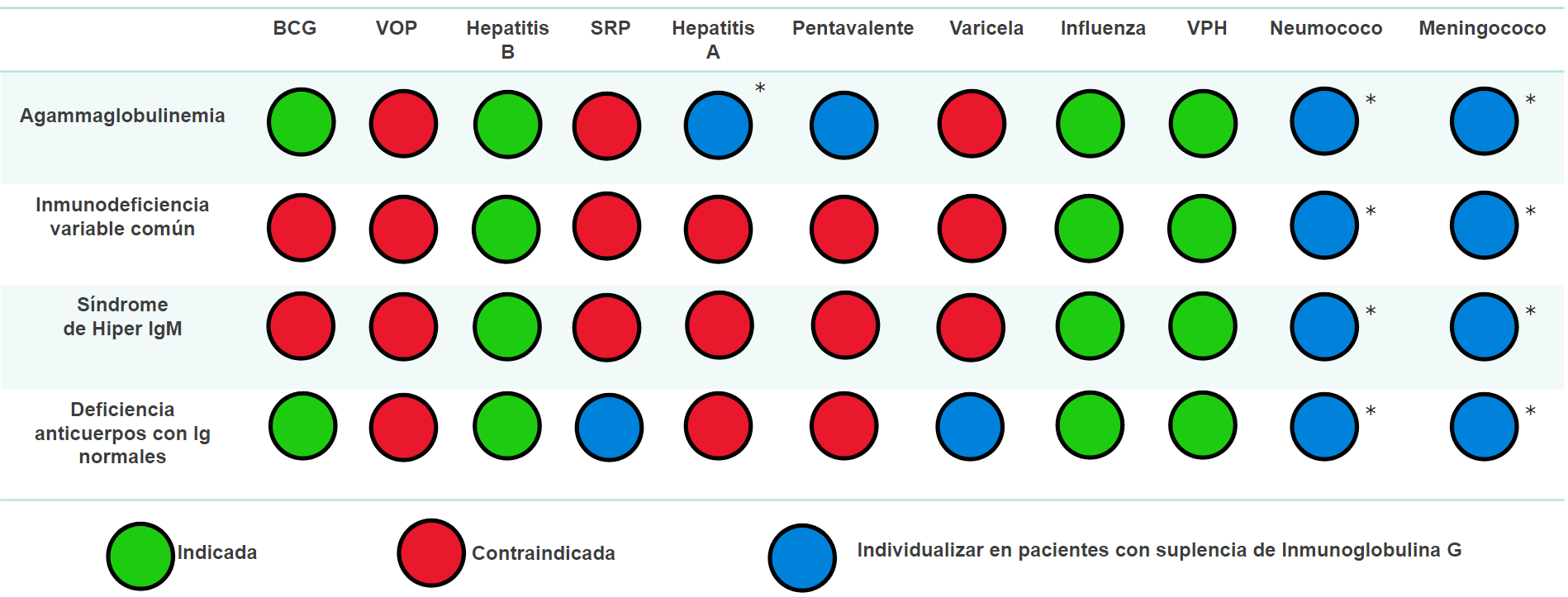
In this manuscript, we carried out an exhaustive analysis of the global recommendations for immunization in inborn errors of immunity patients. We examined the mechanisms of action and types of vaccines, and we described the vaccines included in the Colombian immunization program together with the specific guidelines for immunization in patients with the most frequent inborn errors of immunity in Colombia.
These recommendations were adjusted according to the severity and subclassifications of each immunodeficiency, considering variations in the immune response to offer evidencebased recommendations for vaccination in children with these conditions. We included the most common inborn errors of immunity worldwide and considered the vaccines included in the Colombian immunization program to avoid delays in vaccination schedules.
This work was achieved through a narrative, non-systematic review of articles indexed in Spanish and English, using MeSH terms such as: “inborn errors of immunity”, “primary immunodeficiencies”, “vaccination in inborn errors of immunity, “types of vaccines”, “mechanism of action of vaccines”, and “live vaccines in inborn errors of immunity”.
We used search engines such as: PubMed, Medline, ScienceDirect, and websites of recognized institutions such as the Centers for Disease Control and Prevention (CDC).
Downloads
References
Bonilla FA. Vaccines in patients with primary immune deficiency. Immunol Allergy Clin North Am. 2020;40:421-35. https://doi.org/10.1016/j.iac.2020.03.004
Vetter V, Denizer G, Friedland LR, Krishnan J, Shapiro M. Understanding modern-day vaccines: What you need to know. Ann Med. 2018;50:110-20. https://doi.org/10.1080/07853890.2017.1407035
Olivieri B, Betterle C, Zanoni G. Vaccinations and autoimmune diseases. Vaccines (Basel). 2021;9:815. https://doi.org/10.3390/vaccines9080815
Bühler S, Hatz C. Vaccinations in patients with autoimmune diseases. Ther Umsch. 2016;73:275-80. https://doi.org/10.1024/0040-5930/a000792
Barreto IC, Barreto BA, Cavalcante E, Condino A. Immunological deficiencies: More frequent than they seem to be. J Pediatr (Rio J). 2021;97(Suppl.1):S49-58. https://doi.org/10.1016/j.jped.2020.10.009
Soler P. Diagnóstico de las inmunodeficiencias primarias en Pediatría. Congreso de Actualización en Pediatría 2023. Madrid: Lúa Ediciones 3.0; 2023. p. 167-76.
Pedraza A, Vargas-Rumilla MI, Ramírez-Roa JL. Registro de inmunodeficiencias primarias en niños en un hospital de cuarto nivel. Bogotá, 2010-2016. Rev Alerg Méx. 2018;65:341-8. https://doi.org/10.29262/ram.v65i4.338
Comité Asesor de Vacunas (CAV-AEP). Generalidades de las inmunizaciones. Manual de inmunizaciones en línea de la AEP. Fecha de consulta: 13 de septiembre de 2023. Disponible en: http://vacunasaep.org/documentos/manual/cap-1
Iwasaki A, Omer SB. Why and how vaccines work. Cell. 2020;183:290-5. https://doi.org/10.1016/j.cell.2020.09.040
Lagos M, Díaz C, Hernández P. Respuesta inmune y alergia a vacunas. Rev Méd Clín Las Condes (Internet). 2020;31:256-69. https://doi.org/10.1016/j.rmclc.2020.04.003
Richter D, Anca I, André FE, Bakir M, Chlibek R, Čižman M, et al. Immunization of high-risk paediatric populations: Central European Vaccination Awareness Group recommendations. Expert Rev Vaccines. 2014;13:801-15. https://doi.org/10.1586/14760584.2014.897615
Sobh A, Bonilla FA. Vaccination in primary immunodeficiency disorders. J Allergy Clin Immunol Pract. 2016;4:1066-75. https://doi.org/10.1016/j.jaip.2016.09.012
Gumede N, Muthambi V, Schoub BD. Immunodeficiency-associated vaccine-derived poliovirus type 3 in infant, South Africa, 2011. Emerg Infect Dis. 2012;18:992-4. https://doi.org/10.3201/eid1806.120037
Shaghaghi M, Parvaneh N, Ostad-Rahimi P, Fathi SM, Shahmahmoodi S, Abolhassani H, et al. Combined immunodeficiency presenting with vaccine-associated paralytic poliomyelitis: A case report and narrative review of literature. Immunol Invest. 2014;43:292-8. https://doi.org/10.3109/08820139.2013.859156
Patel NC, Hertel PM, Estes MK, de la Morena M, Petru AM, Noroski LM, et al. Vaccine-acquired rotavirus in infants with severe combined immunodeficiency. N Engl J Med. 2010;362:314-9. https://doi.org/10.1056/NEJMoa0904485
Fontana MC, Bosch P, Yarza M, Rueda ML, Laffargue JA, Cao G, et al. Disseminated bacillus Calmette-Guérin infection in a young girl leading to a diagnosis of immunodeficiency. Actas Dermosifiliogr. 2021;113(Supl.1):S2-4. https://doi.org/10.1016/j.ad.2021.11.006
Bayer DK, Martínez CA, Sorte HS, Forbes LR, Demmler-Harrison GJ, Hanson IC, et al. Vaccine-associated varicella and rubella infections in severe combined immunodeficiency with isolated CD4 lymphocytopenia and mutations in IL7R detected by tandem whole exome sequencing and chromosomal microarray. Clin Exp Immunol. 2014;178:459-69. https://doi.org/10.1111/cei.12421
Galicchio M. Guías de manejo: vacunas en pacientes con inmunodeficiencias primarias. Arch Argent Pediatr. 2010;108:454-64.
Khalili A. Vaccination in patients with primary immunodeficiency disorders. Immunol Genet J. 2020;3:171-9. https://doi.org/10.18502/igj.v3i4.7453
Rubin L, Levin MJ, Ljungman P, Davies EG, Avery R, Tomblyn M, et al. Clinical practice guideline for vaccination of the immunocompromised host. Clin Infect Dis. 2014;58:e44-100. https://doi.org/10.1093/cid/cit684
Guo J, Bolívar-Wagers S, Srinivas N, Holubar M, Maldonado Y. Immunodeficiency-related vaccine-derived poliovirus (iVDPV) cases: A systematic review and implications for polio eradication. Vaccine. 2015;33:1235-42. https://doi.org/10.1016/j.vaccine.2015.01.018
Parvaneh N, Casanova JL, Notarangelo LD, Conley ME. Primary immunodeficiencies: A rapidly evolving story. J Allergy Clin Immunol. 2013;131:314-23. https://doi.org/10.1016/j.jaci.2012.11.051
Sociedad Latinoamericana de Infectología Pediátrica. Manual de vacunas de Latinoamérica. Fecha de consulta: 23 de mayo de 2024. Disponible en: http://slipe.org/web/manualdevacunas2021/
Sociedad Latinoamericana de Infectología Pediátrica. Enfermedad meningocócica en la adolescencia. Fecha de consulta: 23 de mayo de 2024. Disponible en: http://slipe.org/web/manualdevacunas2021/
Hofstetter AM, Jakob K, Klein NP, Dekker CL, Edwards KM, Halsey NA, et al. Live vaccine use and safety in DiGeorge syndrome. Pediatrics. 2014;133:e946-54. https://doi.org/10.1542/peds.2013-0831
Moylett EH, Wasan AN, Noroski LM, Shearer WT. Live viral vaccines in patients with partial DiGeorge syndrome: Clinical experience and cellular immunity. Clin Immunol. 2004;112:106-12. https://doi.org/10.1016/j.clim.2004.02.008
Al-Sukaiti N, Reid B, Lavi S, Al-Zaharani D, Atkinson A, Roifman CM, et al. Safety and efficacy of measles, mumps, and rubella vaccine in patients with DiGeorge syndrome. J Allergy Clin Immunol. 2010;126:868-9. https://doi.org/10.1016/j.jaci.2010.07.018
Bakare N, Menschik D, Tiernan R, Hua W, Martin D. Severe combined immunodeficiency (SCID) and rotavirus vaccination: Reports to the Vaccine Adverse Events Reporting System (VAERS). Vaccine. 2010;28:6609-12. https://doi.org/10.1016/j.vaccine.2010.07.039
Werther RL, Crawford NW, Boniface K, Kirkwood CD, Smart JM. Rotavirus vaccine induced diarrhea in a child with severe combined immune deficiency. J Allergy Clin Immunol 2009;124:600. https://doi.org/10.1016/j.jaci.2009.07.005
Galal N M, Meshaal S, ElHawary R, Nasr E, Bassiouni L, Ashghar H, et al. Poliovirus excretion following vaccination with live poliovirus vaccine in patients with primary immunodeficiency disorders: Clinicians’ perspectives in the endgame plan for polio eradication. BMC Res Notes.2018;11:1-5. https://doi.org/10.1186/s13104-018-3822-7
Mohanty MC, Madkaikar MR, Desai M, Taur P, Nalavade UP, Sharma DK, et al. Poliovirus excretion in children with primary immunodeficiency disorders, India. Emerg Infect Dis. 2017;23:1664-70. https://doi.org/10.3201/eid2310.170724
Giri S, Kumar N, Dhanapal P, Venkatesan J, Kasirajan A, Iturriza-Gomara M, et al. Quantity of vaccine poliovirus shed determines the titer of the serum neutralizing antibody response in Indian children who received oral vaccine. J Infect Dis. 2018;217:1395-8. https://doi.org/10.1093/infdis/jix687
Aghamohammadi A, Abolhassani H, Kutukculer N, Wassilak SG, Pallansch MA, Kluglein S, et al. Patients with primary immunodeficiencies are a reservoir of poliovirus and a risk to polio eradication. Front Immunol. 2017;8:685. https://doi.org/10.3389/fimmu.2017.00685
Marciano BE, Huang CY, Joshi G, Rezaei N, Carvalho BC, Allwood Z, et al. BCG vaccination in patients with severe combined immunodeficiency: Complications, risks, and vaccination policies. J Allergy Clin Immunol. 2014;133:1134-41. https://doi.org/10.1016/j.jaci.2014.02.028
Pöyhönen L, Bustamante J, Casanova JL, Jouanguy E, Zhang Q. Life-threatening infections due to live-attenuated vaccines: Early manifestations of inborn errors of immunity. J Clin Immunol. 2019;39:376-90. https://doi.org/10.1007/s10875-019-00642-3
Zhou Q, Hui X, Ying W, Hou J, Wang W, Liu D, et al. A cohort of 169 chronic granulomatous disease patients exposed to BCG vaccination: A retrospective study from a single center in Shanghai, China (2004-2017). J Clin Immunol. 2018;38:260-72. https://doi.org/10.1007/s10875-018-0486-y
van den Berg JM, van Koppen E, Ahlin A, Belohradsky BH, Bernatowska E, Corbeel L, et al. Chronic granulomatous disease: The European experience. PLoS ONE 2009;4:e5234. https://doi.org/10.1371/journal.pone.0005234
Conti F, Lugo-Reyes SO, Blancas-Galicia L, He J, Aksu G, Borges de Oliveira Jr E, Deswarte C, et al. Mycobacterial disease in patients with chronic granulomatous disease: A retrospective analysis of 71 cases. J Allergy Clin Immunol. 2016;138:2418.e3. https://doi.org/10.1016/j.jaci.2015.11.041
Bustamante J, Aksu G, Vogt G, de Beaucoudrey L, Genel F, Chapgier A, et al. BCG-osis and tuberculosis in a child with chronic granulomatous disease. J Allergy Clin Immunol 2007;120:32-8. https://doi.org/10.1016/j.jaci.2007.04.034
de Oliveira-Junior EB, Zurro NB, Prando C, Cabral-Marques O, Pereira PV, Schimke LF, et al. Clinical and genotypic spectrum of chronic granulomatous disease in 71 Latin American patients: First report from the LASID registry. Pediatr Blood Cancer. 2015;62:2101-7. https://doi.org/10.1002/pbc.25674
Kawashima H, Hasegawa D, Nakamura M, Mochizuki S, Kashiwagi Y, Nishimata S, et al. Hazards of early BCG vaccination: BCGitis in a patient with chronic granulomatous disease. Pediatr Int. 2007;49:418-9. https://doi.org/10.1111/j.1442-200X.2007.02383.x
Ram S, Lewis LA, Rice PA. Infections of people with complement deficiencies and patients who have undergone splenectomy. Clin Microbiol Rev. 2010;23:740-80. https://doi.org/10.1128/CMR.00048-09
Eibl MM, Wolf HM. Vaccination in patients with primary immune deficiency, secondary immune deficiency and autoimmunity with immune regulatory abnormalities. Immunotherapy. 2015;7:1273-92. https://doi.org/10.2217/IMT.15.74
Meyts I, Bucciol G, Quinti I, Neven B, Fischer A, Seoane E, et al. Coronavirus disease 2019 in patients with inborn errors of immunity: An international study. J Allergy Clin Immunol. 2021;147:520-31. https://doi.org/10.1016/j.jaci.2020.09.010
Shields AM, Burns SO, Savic S, Richter AG. COVID-19 in 524 patients with primary and secondary immunodeficiency: The United Kingdom experience. J Allergy Clin Immunol. 2021;147:870-5.e1. https://doi.org/10.1016/j.jaci.2020.12.620
Hagin D, Freund T, Navon M, Halperin T, Adir D, Marom R, et al. Immunogenicity of Pfizer-BioNTech COVID-19 vaccine in patients with inborn errors of immunity. J Allergy Clin Immunol. 2021;148:739-49. https://doi.org/10.1016/j.jaci.2021.05.029
Durkee-Shock JR, Keller MD. Immunizing the imperfect immune system: Coronavirus disease 2019 vaccination in patients with inborn errors of immunity. Ann Allergy Asthma Immunol. 2022;129:562-71.e1. https://doi.org/10.1016/j.anai.2022.06.009
Delmonte OM, Bergerson JRE, Burbelo PD, Durkee-Shock JR, Dobbs K, Bosticardo M, et al. Antibody responses to the SARS-CoV-2 vaccine in individuals with various inborn errors of immunity. J Allergy Clin Immunol. 2021;148:1192-7. https://doi.org/10.1016/j.jaci.2021.08.016
Parker EPK, Desai S, Marti M, Nohynek H, Kaslow DC, Kochhar S et al. Response to additional COVID-19 vaccine doses in people who are immunocompromised: A rapid review. Lancet Glob Health. 2022;10:e326-8. https://doi.org/10.1016/S2214-109X(21)00593-3
Centers for Disease Control and Prevention. Interim clinical considerations for use of COVID-19 vaccines. Fecha de consulta: 13 de septiembre de 2023. Disponible en: https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html
van den Broek B, van Els C, Kuipers B, van Aerde K, Henriet S, de Groot R, et al. MenB-4C vaccine induces effective opsonophagocytic killing in children with a complement deficiency. Clin Exp Immunol. 2019;198:381-9. https://doi.org/10.1111/cei.13368
Sociedad Argentina de Pediatría - Subcomisiones, Comités y Grupos de Trabajo. Consenso sobre actualización de inmunizaciones en pacientes con inmunodeficiencias primarias. Arch Argent Pediatr. 2018;116(Supl.2):S20-33.
NHS Lothian Travel Clinics. JCVI Recommendations on dengue vaccine published. Fecha de consulta: 24 de mayo de 2024. Disponible en: https://services.nhslothian.scot/travelclinics/2024/03/22/jcvi-recommendations-on-dengue-vaccine-published/
AHFS Patient Medication Information. Pneumococcal conjugate vaccine. Fecha de consulta: 24 de mayo de 2024. Disponible en: https://medlineplus.gov/druginfo/meds/a610017.html
Some similar items:
- Nelsy Loango, Martha Lucía Gallego, Beatriz Restrepo, Patricia Landázuri, Gender, age and plasma lipids differences associated with apolipoprotein E polymorphism in school children , Biomedica: Vol. 29 No. 3 (2009)
- María Isabel García, Camila Céspedes, Paola Durán , Catalina Forero, Mauricio Coll , Evaluation of the quality of life in children and adolescents with type 1 diabetes in two health institutions, Bogotá, D. C., Colombia , Biomedica: Vol. 43 No. 1 (2023)
- Carlos Alberto Velasco-Benítez, Claudia Jimena Ortiz-Rivera, Post-infectious functional gastrointestinal disorders in children after a non-severe dengue episode without warning signs , Biomedica: Vol. 39 No. Supl. 2 (2019): Enfermedades transmisibles en el trópico, agosto
- Elpidia Poveda, Ney E. Callas, César M. Baracaldo, Carlina Castillo, Patricia Hernández, Leptin levels in school age children associated with anthropometric measurements and lipid profiles , Biomedica: Vol. 27 No. 4 (2007)
- Ney Callas, Elpidia Poveda, César Baracaldo, Patricia Hernández, Carlina Castillo, Martha Guerra, Genetic polymorphism of the E apolipoprotein in school age children: comparison with levels of plasma lipids and apolipoproteins , Biomedica: Vol. 27 No. 4 (2007)
- Judith Sofía García , María Paula Sarmiento , Jesús David Bello , Nora Alejandra Zuluaga, Adriana Carolina Forero , Laura Fernanda Niño , Hyperthyroidism in children and adolescents: Experience in a university hospital in Colombia , Biomedica: Vol. 42 No. 2 (2022)
- Elpidia Poveda, Ney Callas, César Baracaldo, Carlina Castillo, Patricia Hernández, Martha Guerra, Lipid and apoprotein A-I and B-100 levels in school children school from five central-eastern provinces of Colombia , Biomedica: Vol. 27 No. 3 (2007)
- José Guillermo Ortega-Ávila, Mildrey Mosquera, Isabella Echeverry-Jiménez, Lorena Jiménez-Bastidas, Milton Fabián Suárez-Ortegón, Alberto Pradilla, Luis Eduardo Bravo, Cecilia Aguilar-de Plata, Early atherosclerotic lesions and post-mortem serum cholesterol level in a group of Colombian children , Biomedica: Vol. 33 No. 3 (2013)
- Ana Cecilia Aguilar, Alberto Pradilla, Mildrey Mosquera, Ana Beatriz Gracia, José Guillermo Ortega, Jaime Humberto Leiva, Robinson Ramírez-Vélez, Percentile values for physical condition for Cali, Colombian children and adolescents , Biomedica: Vol. 31 No. 2 (2011)
- Rosa Magdalena Uscátegui, Adriana M. Correa, Jaime Carmona-Fonseca, Changes in retinol, hemoglobin and ferritin concentrations in Colombian children with malaria , Biomedica: Vol. 29 No. 2 (2009)
Copyright (c) 2024 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























