Sticky traps for Aedes aegypti surveillance and targeted vector control in Sincelejo, Colombia
Abstract
Introduction. Entomological surveillance of adult Aedes aegypti mosquitoes provides better risk indicators than in immature stages.
Objective. To determine the usefulness of MosquiTRAP™ traps for Ae. aegypti surveillance, targeted vector control, and the design of dengue prevention measures in Sincelejo, Colombia.
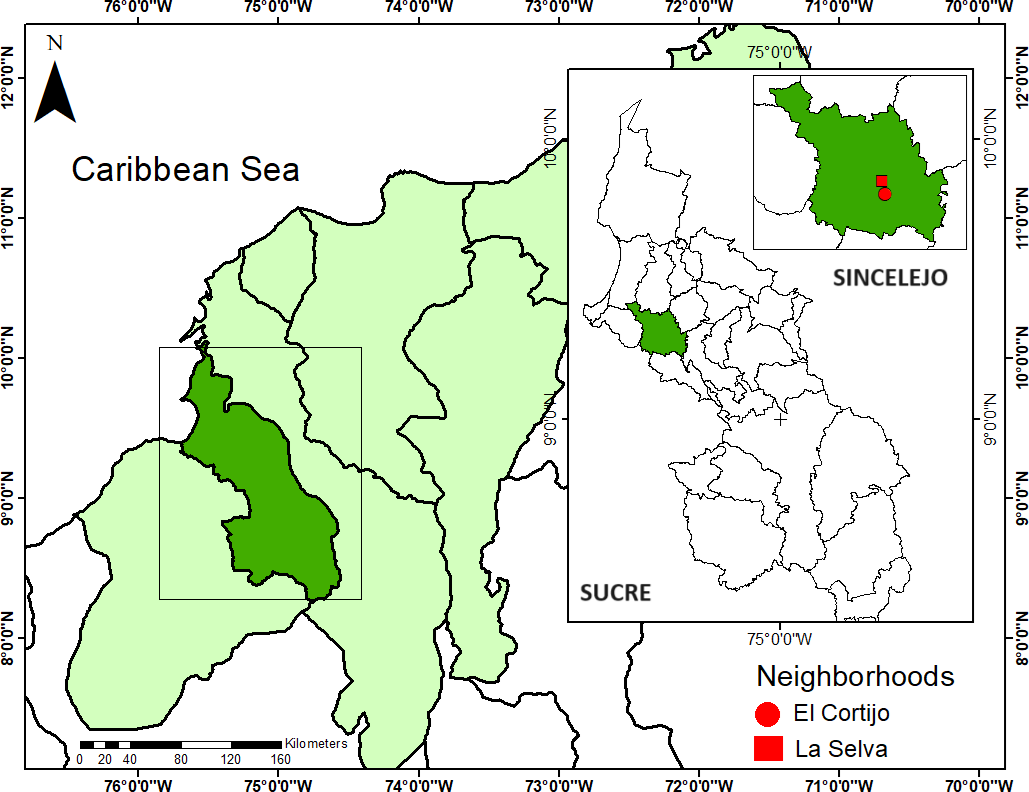
Materials and methods. Forty-nine MosquiTRAP™ traps were deployed over six months to capture gravid Ae. aegypti females in two neighborhoods with historical reports of dengue cases. Entomological indices were calculated to monitor mosquito population dynamics, and the infection frequency of the captured mosquitoes with dengue, zika, and chikungunya virus were assessed. The rates of trap approval and adherence were evaluated, and risk maps were developed based on mosquito abundance. These maps facilitated the identification of specific areas for targeted vector control interventions.
Results. A total of 1,475 mosquitoes were captured, of which 99.1% were identified as A. aegypti. The trap positivity index ranged from 85.7 to 42.9% per inspection, with a mean female Aedes index of two to three mosquitoes per house. Evidence of Ae. aegypti infestation was observed in both neighborhoods, although specific hotspots of high mosquito abundance were identified. No viral infection was detected in the captured mosquitoes.
Conclusions. MosquiTRAP™ traps are useful for Ae. aegypti surveillance as a potential tool to guide vector control and prevention measures for diseases transmitted by this mosquito species.
Downloads
References
1. World Health Organization. About vector-borne diseases. Accessed on: March 3, 2022. Available from: https://www.who.int/news-room/fact-sheets/detail/vector-bornediseases#:~:text=Vector%2Dborne%20diseases%20are%20human,that%20are%20transmitted%20by%20vectors
2. Lwande O, Obanda V, Lindström A, Ahlm C, Evander M, Näslund J, et al. Globe-trotting Aedes aegypti and Aedes albopictus: Risk factors for arbovirus pandemics. Vector Borne Zoonotic Dis. 2020;20:71-81. https://doi.org/10.1089/vbz.2019.2486
3. Ferreira-de-Lima V, Lima-Camara, T. Natural vertical transmission of dengue virus in Aedes aegypti and Aedes albopictus: A systematic review. Parasit Vectors. 2018;11:1-8. https://doi.org/10.1186/s13071-018-2643-9
4. Pan American Health Organization. Epidemiological update dengue. Accessed on: November 7, 2021. Available from: https://www.paho.org/en/documents/epidemiologicalupdate-dengue-23-march-2020
5. Ruiz-López F, González-Mazo A, Vélez-Mira A, Gómez GF, Zuleta L, Uribe S, et al. Presence of Aedes (Stegomyia) aegypti (Linnaeus, 1762) and its natural infection with dengue virus at unrecorded heights in Colombia. Biomédica. 2016;36:303-8. https://doi.org/10.7705/biomedica.v36i2.3301
6. Pan American Health Organization. Métodos de vigilancia entomológica y control de los principales vectores en las Américas. Licencia: CC BY-NC-SA 3.0 IGO; 2021. Accessed on: March 6, 2022. Available from: https://doi.org/10.37774/9789275323953
7. Gama R, Silva I, Resende M, Eiras A. Evaluation of the sticky MosquiTRAP™ for monitoring Aedes aegypti (Diptera: Culicidae) in the district of Itapoã, Belo Horizonte, Minas Gerais, Brazil. Neotrop Entomol. 2007;36:294-302. https://doi.org/10.1590/s1519-566x2007000200018
8. Codeço CT, Lima AW, Araújo SC, Lima JBP, Maciel-de-Freitas R, Honório NA, et al. Surveillance of Aedes aegypti: Comparison of house index with four alternative traps. PLoS Negl Trop Dis. 2015;9:e0003475. https://doi.org/10.1371/journal.pntd.0003475
9. Honório NA, Codeço CT, Alves FdC, Magalhães MdA, Lourenço-De-Oliveira R. Temporal distribution of Aedes aegypti in different districts of Rio de Janeiro, Brazil, measured by two types of traps. J Med Entomol. 2009;46:1001-14. https://doi.org/10.1603/033.046.0505
10. Maciel-de-Freitas R, Eiras ÁE, Lourenço-de-Oliveira R. Calculating the survival rate and estimated population density of gravid Aedes aegypti (Diptera, Culicidae) in Rio de Janeiro, Brazil. Cad Saude Publica. 2008;24:2747-54. https://doi.org/10.1590/s0102-311x2008001200003
11. Resende MC, Silva IM, Eiras ÁE. Avaliação da operacionalidade da armadilha MosquiTRAP™ no monitoramento de Aedes aegypti. Epidemiol Serv Saude. 2010;19:329-38. https://doi.org/10.5123/S1679-49742010000400003
12. Manjarres M, Martínez J. Dinámica poblacional y búsqueda de infección natural con virus dengue en poblaciones de Aedes aegypti en el municipio de Sincelejo: dos herramientas para la estimación del riesgo epidemiológico (tesis). Sincelejo: Universidad de Sucre; 2015.
13. Aguilera-Díaz M. La economía del departamento de Sucre: ganadería y sector público. Cartagena: Banco de la República; 2005.
14. Departamento Administrativo Nacional de Estadística - DANE. Proyecciones y retroproyecciones de población municipal para el periodo 1985-2017 y 2018-2035 con base en el CNPV 2018. Accessed on: November 7, 2021. Available from: https://www.dane.gov.co/index.php/estadisticas-por-tema/demografia-y-poblacion/proyecciones-de-poblacion
15. Rueda LM. Pictorial keys for the identification of mosquitoes (Diptera: Culicidae) associated with dengue virus transmission. Zootaxa. 2004;589:1-60. https://doi.org/10.11646/zootaxa.589.1.1
16. Lane J. Neotropical Culicidae. São Paulo: University of São Paulo; 1953.
17. Roehrig JT, Butrapet S, Liss NM, Bennett SL, Luy BE, Childers T, et al. Mutation of the dengue virus type 2 envelope protein heparan sulfate binding sites or the domain III lateral ridge blocks replication in Vero cells prior to membrane fusion. Virology. 2013;441:114-25. https://doi.org/10.1016/j.virol.2013.03.011
18. Lanciotti RS, Kosoy OL, Laven JJ, Velez JO, Lambert AJ, Johnson AJ, et al. Genetic and serologic properties of Zika virus associated with an epidemic, Yap State, Micronesia, 2007. Emerg Infect Dis. 2008;14:1232-9. https://doi.org/10.3201/eid1408.080287
19. Waggoner JJ, Ballesteros G, Gresh L, Mohamed-Hadley A, Tellez Y, Sahoo MK, et al. Clinical evaluation of a single-reaction real-time RT-PCR for pan-dengue and chikungunya virus detection. J Clin Virol. 2016;78:57-61. https://doi.org/10.1016/j.jcv.2016.01.007
20. Wu P, Sun P, Nie K, Zhu Y, Shi M, Xiao C, et al. A gut commensal bacterium promotes mosquito permissiveness to arboviruses. Cell Host Microbe.2019;25:101-12. e5. https://doi.org/10.1016/j.chom.2018.11.004
21. Resende MC, Ázara TM, Costa IO, Heringer LC, Andrade MR, Acebal JL, et al. Field optimisation of MosquiTRAP™ sampling for monitoring Aedes aegypti Linnaeus (Diptera:Culicidae). Mem Inst Oswaldo Cruz. 2012;107:294-302. https://doi.org/10.1590/s0074-02762012000300002
22. Lana RM, Morais MM, Lima TFMd, Carneiro TGdS, Stolerman LM, dos Santos JP, et al. Assessment of a trap based Aedes aegypti surveillance program using mathematical modeling. PLoS One. 2018;13:e0190673. https://doi.org/10.1371/journal.pone.0190673
23. Ritchie SA, Long S, Smith G, Pyke A, Knox TB. Entomological investigations in a focus of dengue transmission in Cairns, Queensland, Australia, by using the sticky ovitraps. J Med Entomol. 2004;41:1-4. https://doi.org/10.1603/0022-2585-41.1.1
24. Winskill P, Carvalho DO, Capurro ML, Alphey L, Donnelly CA, McKemey AR. Dispersal of engineered male Aedes aegypti mosquitoes. PLoS Negl Trop Dis. 2015;9:e0004156. https://doi.org/10.1371/journal.pntd.0004156
25. Fávaro EA, Dibo MR, Mondini A, Ferreira AC, Barbosa AA, Eiras ÁE, et al. Physiological state of Aedes (Stegomyia) aegypti mosquitoes captured with MosquiTRAPsTM in Mirassol, São Paulo, Brazil. J Vector Ecol. 2006;31:285-91. https://doi.org/10.3376/1081-1710(2006)31[285:psoasa]2.0.co;2
26. Quimbayo M, Rúa-Uribe G, Parra-Henao G, Torres C. Evaluation of lethal ovitraps as a strategy for Aedes aegypti control. Biomédica. 2014;34:473-82. https://doi.org/10.7705/biomedica.v34i3.2146
27. Fávaro EA, Mondini A, Dibo MR, Barbosa AA, Eiras ÁE, Neto FC. Assessment of entomological indicators of Aedes aegypti (L.) from adult and egg collections in São Paulo, Brazil. J Vector Ecol. 2008;33:8-16. https://doi.org/10.3376/1081-1710(2008)33[8:aoeioa]2.0.co;2
28. Eiras ÁE, Resende MC. Preliminary evaluation of the “Dengue-MI” technology for Aedes aegypti monitoring and control. Cad Saude Publica. 2009;25(Suppl.1):S45-58. https://doi.org/10.1590/s0102-311x2009001300005
29. Pessoa A. Monitoramento do dengue virus circulante em larvas e mosquitos adultos de Aedes aegypti (thesis). Belo Horizonte: Universidade Federal de Minas Gerais; 2007.
30. Cabarca S, Pérez C, Blanco-Tuirán P, Castellar A, Camacho-Burgos E. Infestación por Aedes aegypti en una localidad del municipio de Sincelejo, departamento de Sucre. Rev Investig Med Trop. 2015;1:14-9.
31. Eiras AE, Resende MC, Acebal JL, Paixão KS. New cost-benefit of Brazilian technology for vector surveillance using trapping system. In: Manguin S, editor. Malaria. London: IntechOpen; 2018. p. 1-24. https://doi.org/10.5772/intechopen.78781
32. Dos Santos TP, Cruz OG, da Silva KAB, de Castro MG, de Brito AF, Maspero RC, et al. Dengue serotype circulation in natural populations of Aedes aegypti. Acta Tropica. 2017;176:140-3. https://doi.org/10.1016/j.actatropica.2017.07.014
33. Instituto Nacional de Salud. Estadísticas de vigilancia rutinaria. Accesseed on: November 7, 2021. Available from: http://portalsivigila.ins.gov.co/Paginas/Vigilancia-Rutinaria.aspx
34. de Melo DPO, Scherrer LR, Eiras ÁE. Dengue fever occurrence and vector detection by larval survey, ovitrap and MosquiTRAP™: A space-time clusters analysis. PLoS One. 2012;7:e42125. https://doi.org/10.1371/journal.pone.0042125
35. Louis VR, Phalkey R, Horstick O, Ratanawong P, Wilder-Smith A, Tozan Y, et al. Modeling tools for dengue risk mapping – A systematic review. Int J Health Geogr. 2014;13:50. https://doi.org/10.1186/1476-072X-13-50
36. Haufe W. The effects of atmospheric pressure on the flight responses of Aedes aegypti (L.). Bull Entomol Res. 1954;45:507-26. https://doi.org/10.1017/S000748530002959X
37. Galun R, Fraenkel G. The effect of low atmospheric pressure on adult Aedes aegypti and on housefly pupae. J Insect Physiol. 1961;7:161-76. https://doi.org/10.1016/0022-1910(61)90069-5
Some similar items:
- Mikel A. González , María Altagracia Rodríguez-Sosa , Yohan Enmanuel Vásquez-Bautista , Elizabeth del Carmen Rosario , Jesús Confesor Durán-Tiburcio , Pedro María Alarcón-Elbal, A survey of tire-breeding mosquitoes (Diptera: Culicidae) in the Dominican Republic: Considerations about a pressing issue , Biomedica: Vol. 40 No. 3 (2020)
- Patricio Mora , Paúl Quinatoa , Diego Morales, Update of the spatiotemporal distribution of Aedes albopictus in Ecuador , Biomedica: Vol. 45 No. 3 (2025)
- Manuel Muñoz, Juan Carlos Navarro, Mayaro: A re-emerging Arbovirus in Venezuela and Latin America , Biomedica: Vol. 32 No. 2 (2012)
- Cristian Camilo Canizales, Julio Cesar Carranza , Gustavo Adolfo Vallejo, Daniel Alfonso Urrea , Aedes albopictus distribution in Ibagué, Colombia: Potential risk of arboviral outbreaks , Biomedica: Vol. 43 No. 4 (2023)
- Celeny Ortiz, Guillermo L. Rúa-Uribe, Carlos A. Rojas, Knowledge, practices and entomological aspects of dengue in Medellín, Colombia: A comparative study of neighborhoods with high and low incidence , Biomedica: Vol. 38 No. Sup. 2 (2018): Suplemento 2, Medicina tropical
- Gabriel Parra-Henao, Laura Suárez, Mosquitoes (Diptera: Culiciadae) as potential vectors of arbovirused in the Urabá region, Northwest of Colombia , Biomedica: Vol. 32 No. 2 (2012)
- Jorge R. Rey, Philip Lounibos, Ecology of Aedes aegypti and Aedes albopictus in the Americas and disease transmission , Biomedica: Vol. 35 No. 2 (2015)
- Luz Elena Velásquez, Catalina Gómez, Erika Valencia, Laura Salazar, Eudoro Casas, Paragonimosis in the peri-urban zone of Medellín, Antioquia , Biomedica: Vol. 28 No. 3 (2008)
- Clara Beatriz Ocampo, Gloria I. Giraldo Calderon, Mauricio Perez, Carlos A. Morales, Evaluation of the triflumuron and the mixture of Bacillus thuringiensis plus Bacillus sphaericus for control of the immature stages of Aedes aegypti and Culex quinquefasciatus (Diptera: Culicidae) in catch basins , Biomedica: Vol. 28 No. 2 (2008)
- María Elena Cuéllar-Jiménez, Olga Lucía Velásquez-Escobar, Ranulfo González-Obando, Carlos Andrés Morales-Reichmann, Detection of Aedes albopictus (Skuse) (Diptera: Culicidae) in the city of Cali, Valle del Cauca, Colombia , Biomedica: Vol. 27 No. 2 (2007)
Copyright (c) 2025 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























