Treatment for multidrug-resistant tuberculosis: A comparative analysis of programmatic outcome indicators between Buenaventura and other municipalities of Valle del Cauca, Colombia
Abstract
Introduction. Proper management of multidrug-resistant tuberculosis is a prioritized strategy for tuberculosis control worldwide.
Objective. To evaluate differences concerning demographic and clinical characteristics and programmatic indicators of Buenaventura patient cohort with confirmed diagnosis of multidrug-resistant tuberculosis, compared to those of the other municipalities from Valle del Cauca, Colombia, 2013-2016.
Materials and methods. We conducted an analytical cohort study to compare records of patients older than 15 years with multidrug-resistant tuberculosis included in the Programa de Tuberculosis de Buenaventura (with para-aminosalicylic acid) versus the other municipalities of Valle del Cauca (without para-aminosalicylic).
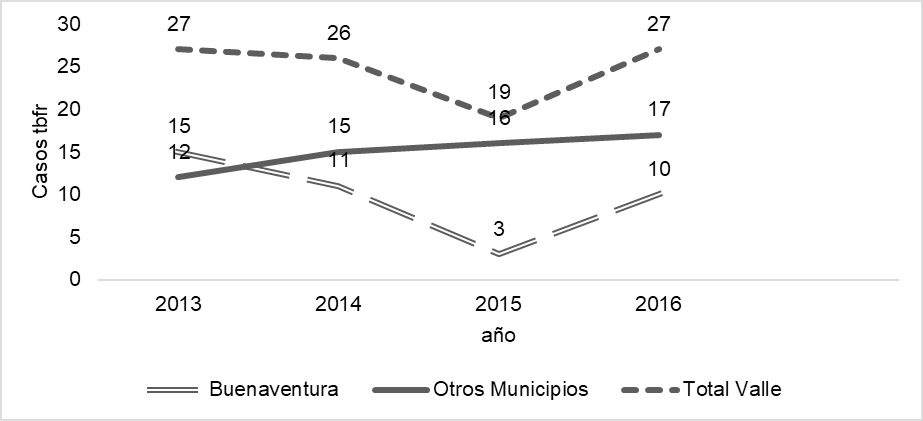
Results. Ninety-nine cases were recorded with a median age of 40 years (IQR = 26 - 53); in Buenaventura, 56% of the patients were women, while in the other municipalities, men predominated with 67%; 95% had health insurance. The most common comorbidity was diabetes (14%). Adverse reactions to antituberculosis medications in Buenaventura were 1.3 times more frequent than in the other municipalities (OR = 2.3; 95% CI = 0.993 - 5.568; p = 0.04). In Buenaventura, the mortality rate was 5% compared to the 15% reported in the other municipalities. Treatment failures were not reported in Buenaventura, but 35% did not continue with the follow-up. Treatment success was higher in Buenaventura (56 %).
Conclusion. A strengthened program in Buenaventura presented better programmatic results than those from the other municipalities of Valle del Cauca. Access to molecular tests, availability of shortened treatments, and continuous monitoring to identify adverse reactions to antituberculosis medications are routes for all other control programs.
Downloads
References
World Health Organization. Implementing the end TB strategy: The essentials. Geneva: WHO; 2015. Fecha de consulta: 20 de octubre de 2022. Disponible en: https://www.who.int/publications/i/item/implementing-the-end-tb-strategy
World Health Organization. Global Tuberculosis Report 2021. Geneva: WHO; 2021. Fecha de consulta: 23 de enero de 2023. Disponible en: https://www.who.int/publications/i/item/implementing-the-end-tb-strategyhttps://www.who.int/publications/i/item/9789240037021
World Health Organization. Tuberculosis country, regional and global profiles 2021. Fecha de consulta: 23 de enero de 2023. Disponible en: https://worldhealthorg.shinyapps.io/tb_profiles/?_inputs_&lan=%22EN%22&entity_type=%22group%22&group_code=%22global%22
Ministerio de Salud y Protección Social de Colombia. Informe de evento Tuberculosis 2021. Bogotá.: Minsalud; 2021.
Villegas SL, Ferro BE, Pérez-Vélez CM, Moreira CA, Forero L, Martínez E, et al. High initial multidrug-resistant tuberculosis rate in Buenaventura, Colombia: A public-private initiative. Eur Respir J. 2012;40:1569-72. https://doi.org/10.18046/EUI/ee.1.2021
Ministerio de Salud y Protección Social de Colombia. Plan estratégico. Colombia libre de tuberculosis 2010-2015. Para la expansión y fortalecimiento de la estrategia alto a la tuberculosis. Bogotá: Minsalud; 2009.
Secretaría Departamental de Salud del Valle. Acta Comité de Evaluación Regional de Casos Especiales de Tuberculosis (CERCET). Cali: Alcaldía de Cali; 2011.
Tobón A, Rueda J, Cáceres DH, Mejía GI, Zapata EM, Montes F, et al. Adverse treatment outcomes in multidrug resistant tuberculosis go beyond the microbe-drug interaction: Results of a multiple correspondence analysis. Biomédica. 2020;40:616-25. https://doi.org/10.7705/biomedica.5072
Chaves-Torres NM, Quijano-Rodríguez JJ, Porras-Andrade PS, Arriaga MB, Netto EM. Factors predictive of the success of tuberculosis treatment: A systematic review with metaanalysis. PLoS ONE. 2019;14:e0226507. https://doi.org/10.1371/journal.pone.0226507
Chaves-Torres NM, Fadul S, Patiño J, Netto E. Factors associated with unfavorable treatment outcomes in patients with rifampicin-resistant tuberculosis in Colombia 2013-2015: A retrospective cohort study. PLoS ONE. 2021;16:e0249565. https://doi.org/10.1371/journal.pone.0249565
Puerto-Castro GM, Montes-Zuluaga FN, Alcalde-Rabanal JE, Pérez F. Patient- and providerrelated factors in the success of multidrug-resistant tuberculosis treatment in Colombia. Rev Panam Salud Pública. 2021;45:e74. https://doi.org/10.26633/RPSP.2021.74
World Health Organization. Automated real-time nucleic acid amplification technology for rapid and simultaneous detection of tuberculosis and rifampicin resistance: Xpert Mtuberculosis/RIF system: Policy statement 2011. Fecha de consulta: 17 de octubre de 2022. Disponible en: https://apps.who.int/iris/handle/10665/44586
Ministerio de Salud y Protección Social de Colombia. Resolución 227 de 2020. Bogotá, D.C.: Minsalud; 2020.
Agredo F, Osorio L. Coverage and fidelity of the Xpert MTB/RIF implementation in a highburden area for pulmonary tuberculosis in Colombia. Biomédica. 2020;40:626-40. https://doi.org/10.7705/biomedica.5272
Palmero D, Cruz V, Museli T, Pavlovsky H, Fernández J, Waisman J. Adverse drug reactions in multidrug-resistant tuberculosis. Medicina (B. Aires). 2010;70:427-33.
Lan Z, Ahmad N, Baghaei P, Barkane L, Benedetti A, Brode SK, et al. Drug-associated adverse events in the treatment of multidrug-resistant tuberculosis: An individual patient data meta-analysis. Lancet Respir Med. 2020;8:383-94. https://doi.org/10.1016/S2213-2600(20)30047-3
Walker IF, Shi O, Hicks JP, Elsey H, Wei X, Menzies D, et al. Analysis of loss to follow-up in 4,099 multidrug-resistant pulmonary tuberculosis patients. Eur Respir J. 2019;54. https://doi.org/10.1183/13993003.00353-2018
Bhering M, Duarte R, Kritski A. Predictive factors for unfavourable treatment in MDR-TB and XDR-TB patients in Rio de Janeiro State, Brazil, 2000-2016. PLoS ONE. 2019;14:e0218299. https://doi.org/10.1371/journal.pone.0218299
Mateus-Solarte JC, Carvajal-Barona R. Factors predictive of adherence to tuberculosis treatment, Valle del Cauca, Colombia. Int J Tuberc Lung Dis. 2008;12:520-6.
PAHO. Tuberculosis in the Americas, 2018. Washington, D.C.: PAHO; 2018. Fecha de consulta: 17 de octubre de 2022. Disponible en: http://iris.paho.org/xmlui/handle/10665.2/49510
Haraka F, Kakolwa M, Schumacher SG, Nathavitharana RR, Denkinger CM, Gagneux S, et al. Impact of the diagnostic test Xpert MTB/RIF on patient outcomes for tuberculosis. Cochrane Database Syst Rev. 2021;5:CD012972. https://doi.org/10.1002/14651858.CD012972.pub2
Schumacher SG, Sohn H, Qin ZZ, Gore G, Davis JL, Denkinger CM, et al. Impact of molecular diagnostics for tuberculosis on patient-important outcomes: A systematic review of study methodologies. PLoS ONE. 2016;11:e0151073. https://doi.org/10.1371/journal.pone.0151073
Moreira CA, Hernández HL, Arias NL, Castaño MC, Ferro BE, Jaramillos E. Resistencia inicial a drogas antituberculosas en Buenaventura, Colombia. Biomédica. 2004;24(Supl.1):73-9. https://doi.org/10.7705/biomedica.v24iSupp1.1305
Collaborative Group for the Meta-Analysis of Individual Patient Data in MDRTBt, Ahmad N, Ahuja SD, Akkerman OW, Alffenaar JC, Anderson LF, et al. Treatment correlates of successful outcomes in pulmonary multidrug-resistant tuberculosis: An individual patient data meta-analysis. Lancet. 2018;392:821-34. https://doi.org/10.1016/S0140-6736(18)31644-1
Ministerio de Salud y Protección Social de Colombia. Plan Estratégico “Hacia el fin de la Tuberculosis” Colombia 2016-2025. Bogotá, D.C.: Minsalud; 2016.
Borisov S, Danila E, Maryandyshev A, Dalcolmo M, Miliauskas S, Kuksa L, et al. Surveillance of adverse events in the treatment of drug-resistant tuberculosis: First global report. Eur Respir J. 2019;54. https://doi.org/10.1183/13993003.01522-2019
World Health Organization. Rapid communication: Key changes to the treatment of drugresistant tuberculosis. Fecha de consulta: 20 de octubre de 2022. Disponible en: https://www.who.int/publications/i/item/WHO-UCN-tuberculosis-2022-2
Some similar items:
- Elizabeth Borrero, Gabriel Carrasquilla, Neal Alexander, Decentralization and health system reform: What is their impact on malaria incidence in Colombian municipalities? , Biomedica: Vol. 32 (2012): Suplemento 1, Malaria
- Claudia Llerena, Angélica Valbuena, Angie Paola Zabaleta, Mycobacterioses identified in the National Reference Laboratory of Colombia from 2012 to 2016 , Biomedica: Vol. 38 No. Sup. 2 (2018): Suplemento 2, Medicina tropical
- John-Leonardo Torres-Castiblanco, Jorge Alberto Carrillo, Daniel Hincapié-Urrego, Adriana Rojas-Villarraga, Tuberculosis in the era of anti-TNF-alpha therapy: Why does the risk still exist? , Biomedica: Vol. 38 No. 1 (2018)
- Alvaro Javier Idrovo, Historical, social and epidemiological roots of tuberculosis in Bogotá, Colombia. , Biomedica: Vol. 24 No. 4 (2004)
- Héctor Serrano-Coll, Olinto Mieles, Calixto Escorcia, Amparo Díaz, Camilo Beltrán, Nora Cardona-Castro, A case series of pure neural leprosy in patients diagnosed in a specialized center for the control of Hansen’s disease in Colombia , Biomedica: Vol. 38 No. 2 (2018)
- Claudia Lucía Colorado, Guillermo Sánchez, Martha Inírida Guerrero, Clara Inés León, Reliability and agreement of two smear reading scales for classification and monitoring of multidrug therapy in leprosy patients , Biomedica: Vol. 31 No. 3 (2011)
- Laura Plata-Casas, Óscar Gutiérrez-Lesmes, Óscar Herrán-Falla, Tuberculosis and human immunodeficiency virus coinfection: Epidemiological situation in the department of Meta, 2010- 2015 , Biomedica: Vol. 38 No. Sup. 2 (2018): Suplemento 2, Medicina tropical
- Carlos A. Torres-Duque, Claudia Díaz, Leslie Vargas, Elsa María Serpa, Walter Mosquera, María Consuelo Garzón, Graciela Mejía, Luz Mary García, Liliana Andrea González, Claudia Marcela Castro, Wellman Ribón, Disseminated mycobacteriosis affecting a prosthetic aortic valve: first case of Mycobacterium peregrinum type III reported , Biomedica: Vol. 30 No. 3 (2010)
- Adriana Rojas-Villarraga, Carlos Andrés Agudelo, Ricardo Pineda-Tamayo, Alvaro Porras, Gustavo Matute, Juan Manuel Anaya, Tuberculosis in patientes treated with tumor necrosis factor alpha antagonists living in an endemic area. Is the risk worthwhile? , Biomedica: Vol. 27 No. 2 (2007)
- Juan Gabriel Bueno-Sánchez, Jairo René Martínez-Morales, Elena E. Stashenko, Wellman Ribón, Anti-tubercular activity of eleven aromatic and medicinal plants occurring in Colombia , Biomedica: Vol. 29 No. 1 (2009)
Copyright (c) 2024 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























