Molecular characterization of the Plasmodium falciparum k13 gene helix domain in samples from native communities of Condorcanqui, Amazonas, Perú
Abstract
Introduction. Resistance of Plasmodium falciparum to different antimalarial drugs is an obstacle to disease elimination. The artemisinin-resistant genotype of P. falciparum can be assessed by examining polymorphisms in the helix domain of the Pfk13 gene. The World Health Organization recommends these mutations as molecular markers to detect artemisinin-resistant in countries where P. falciparum malaria is endemic.
Objective. To identify artemisinin resistance-related mutations present in the helix domain of the P. falciparum k13 gene.
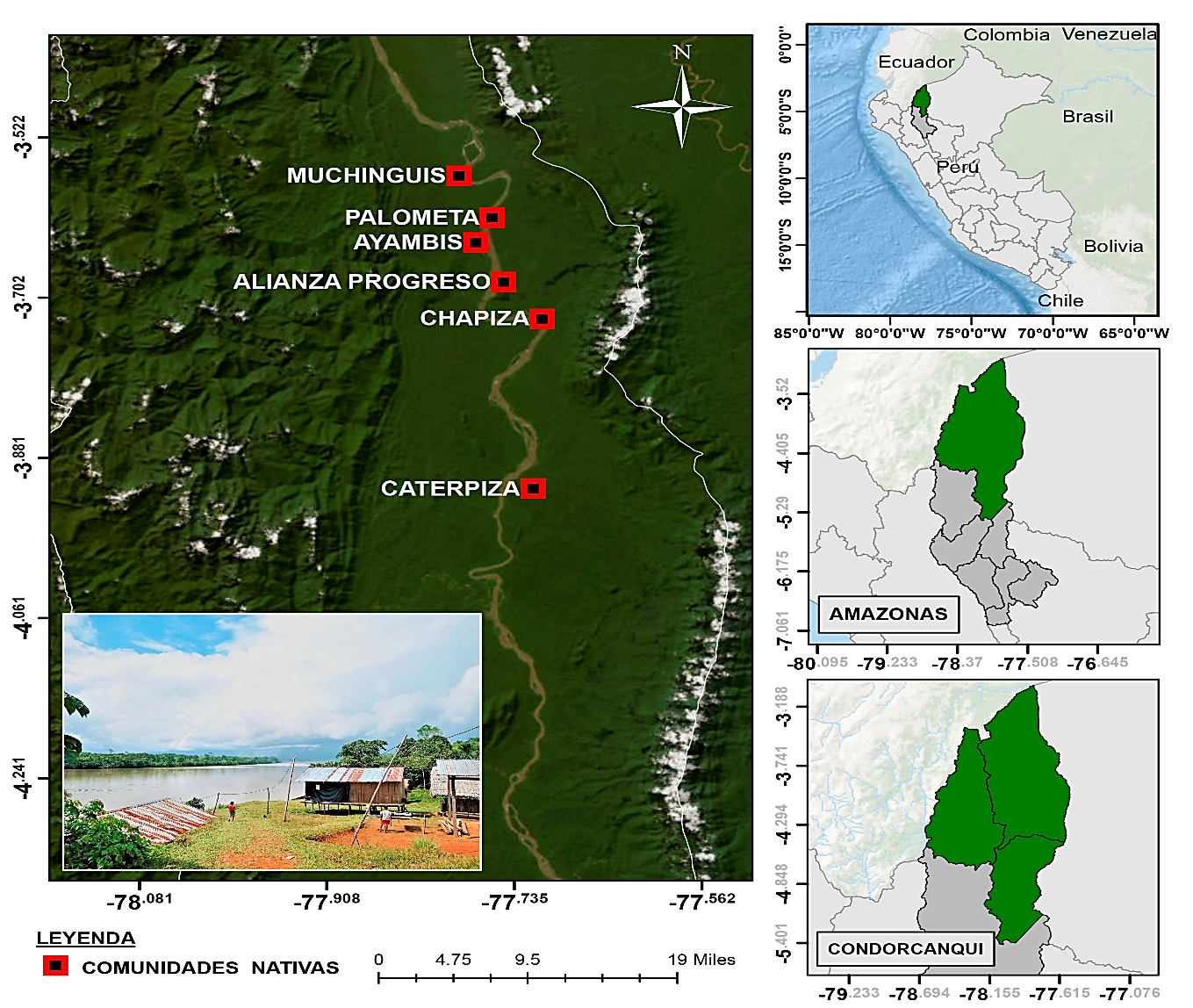
Materials and methods. We collected a total of 51 samples through passive case detection, positive for Plasmodium by microscopy, from six communities in the district of Río Santiago in Condorcanqui, Amazonas. Molecular species confirmation was performed by real-time PCR and Pfk13 helix domain was amplified and sequenced by capillary electrophoresis. The obtained sequences were compared with the wild type 3D7 reference strain.
Results. A total of 51 positive samples were confirmed for P. falciparum from the communities of Ayambis, Chapiza, Palometa, Muchinguis, Alianza Progreso and Caterpiza. DNA sequences alignment showed the absence of resistance-associated mutations in the k13 gene of the collected samples.
Discussion. The obtained results are consistent with similar studies conducted in other South American countries, including Perú, so these data provide a baseline for artemisinin-resistance molecular surveillance in the Amazon region and reinforce the efficacy of artemisinin-based combination therapy in this area.
Downloads
References
Chapadense F, Machado RL, Ventura AMR da S, Áreas A, Machado RB, Viana GR, et al. Plasmodium falciparum malarial parasites from Brazil lack artemisinin resistance-associated mutations in the kelch13 gene. Soc Bras Med Trop. 2019;52:e20180225. https://doi.org/10.1590/0037-8682-0225-2018
Valenzuela G, Castro LE, Valencia-Zamora J, Vera-Arias CA, Rohrbach P, Sáenz FE. Genotypes and phenotypes of resistance in Ecuadorian Plasmodium falciparum. Malar J. 2019;18:415. https://doi.org/10.1186/s12936-019-3044-z
Mathieu LC, Cox H, Early AM, Mok S, Lazrek Y, Paquet J, et al. Local emergence in Amazonia of Plasmodium falciparum K13 C580Y mutants associated with in vitro artemisinin resistance. eLife. 2020;9:e51015. https://doi.org/10.7554/eLife.51015
Boullé M, Witkowski B, Duru V, Sriprawat K, Nair SK, McDew-White M, et al. Artemisininresistant Plasmodium falciparum K13 mutant alleles, Thailand–Myanmar border. Emerg Infect Dis. 2016;22:1503-5. https://doi.org/10.3201/eid2208.160004
Gomes LR, Lavigne A, Peterka CL, Brasil P, Ménard D, Daniel-Ribeiro CT, et al. Absence of K13 polymorphism in Plasmodium falciparum from Brazilian areas where the parasite is endemic. Antimicrob Agents Chemother. 2018;62:e00354-18. https://doi.org/10.1128/AAC.00354-18
Ghanchi NK, Qurashi B, Raees H, Beg MA. Molecular surveillance of drug resistance: Plasmodium falciparum artemisinin resistance single nucleotide polymorphisms in Kelch protein propeller (K13) domain from Southern Pakistan. Malar J. 2021;20:176. https://doi.org/10.1186/s12936-021-03715-0
Kamau E, Campino S, Amenga-Etego L, Drury E, Ishengoma D, Johnson K, et al. K13-propeller polymorphisms in Plasmodium falciparum parasites from sub-Saharan Africa. J Infect Dis. 2015;211:1352-5. https://doi.org/10.1093/infdis/jiu608
Chenet SM, Akinyi-Okoth S, Huber CS, Chandrabose J, Lucchi NW, Talundzic E, et al. Independent emergence of the Plasmodium falciparum Kelch propeller domain mutant allele C580Y in Guyana. J Infect Dis. 2016;213:1472-5. https://doi.org/10.1093/infdis/jiv752
Arróspide N, Hijar-Guerra G, De Mora D, Díaz-Cortéz CE, Veloz-Pérez R, Gutiérrez S, et al. Alelos mutantes asociados a la resistencia a cloroquina y sulfadoxina-pirimetamina en Plasmodium falciparum de las fronteras Ecuador-Perú y Ecuador-Colombia. Rev Peru Med Exp Salud Publica. 2014;31:282-7.
Williams HA, Vincent-Mark A, Herrera Y, Chang OJ. A retrospective analysis of the change in anti-malarial treatment policy: Peru. Malar J. 2009;8:85. https://doi.org/10.1186/1475-2875-8-85
Ministerio de Salud del Perú. Norma técnica de salud para la atención de la malaria y malaria grave en el Perú. Lima: Ministerio de Salud; 2015.
World Health Organization. Good manufacturing practices for sterile pharmaceutical products. Ginebra: WHO; 2004. Fecha de consulta: 5 de mayo de 2023. Disponible en: https://www.who.int/es/publications/i/item/HTM-GMP-MM-SOP-10
Montenegro CC, Bustamante-Chauca TP, Pajuelo-Reyes C, Bernal M, Gonzales L, Tapia-Limonchi R, et al. Plasmodium falciparum outbreak in native communities of Condorcanqui, Amazonas, Perú. Malar J. 2021;20:88. https://doi.org/10.1186/s12936-021-03608-2
Rougemont M, van Saanen M, Sahli R, Hinrikson HP, Bille J, Jaton K. Detection of four Plasmodium species in blood from humans by 18S rRNA gene subunit-based and speciesspecific real-time PCR assays. J Clin Microbiol. 2004;42:5636-43. https://doi.org/10.1128/JCM.42.12.5636-5643.2004
Yasri S, Wiwanitkit V. Artemisinin resistance: An important emerging clinical problem in tropical medicine. Int J Physiol Pathophysiol Pharmacol. 2021;13:152-7.
Montenegro LM, de Las Salas B, Neal AT, Tobon-Castaño A, Fairhurst RM, Lopera-Mesa TM. State of artemisinin and partner drug susceptibility in Plasmodium falciparum clinical isolates from Colombia. Am J Trop Med Hyg. 2021;104:263-70. https://doi.org/10.4269/ajtmh.20-0148
Inoue J, Jovel I, Morris U, Aydin-Schmidt B, Islam A, Segurado AC, et al. Absence of Plasmodium falciparum K13 propeller domain polymorphisms among field isolates collected from the Brazilian Amazon Basin between 1984 and 2011. Am J Trop Med Hyg. 2018;99:1504-7. https://doi.org/10.4269/ajtmh.18-05541504
Gomes LR, Lavigne A, Peterka CL, Brasil P, Ménard D, Daniel-Ribeiro CT, et al. Absence of K13 polymorphism in Plasmodium falciparum from Brazilian areas where the parasite is endemic. Antimicrob Agents Chemother. 2018;62:e00354-18. https://doi.org/10.1128/AAC.00354-18
Villena FE, Sánchez JF, Nolasco O, Braga G, Ricopa L, Barazorda K, et al. Drug resistance and population structure of Plasmodium falciparum and Plasmodium vivax in the Peruvian Amazon. Sci Rep. 2022;12:16474. https://doi.org/10.1038/s41598-022-21028-3
Kattenberg JH, Fernández-Miñope C, van Dijk NJ, Llacsahuanga-Allcca L, Guetens P, Valdivia HO, et al. Malaria molecular surveillance in the Peruvian Amazon with a novel highly multiplexed Plasmodium falciparum AmpliSeq assay. Microbiol Spectr. 2023;11:e0096022. https://doi.org/10.1128/spectrum.00960-22
Rawat M, Kanyal A, Choubey D, Deshmukh B, Malhotra R, Mamatharani DV, et al. Identification of co-existing mutations and gene expression trends associated with K13-mediated artemisinin resistance in Plasmodium falciparum. Front Genet. 2022;13:824483. https://doi.org/10.3389/fgene.2022.824483
Oberstaller J, Zoungrana L, Bannerman CD, Jahangiri S, Dwivedi A, Silva JC, et al. Integration of population and functional genomics to understand mechanisms of artemisinin resistance in Plasmodium falciparum. Int J Parasitol Drugs Drug Resist. 2021;16:119-28. https://doi.org/10.1016/j.ijpddr.2021.05.006
Ministerio de Salud del Perú. Plan hacia la eliminación de la Malaria en el Perú 2022-2030. Lima: Ministerio de Salud del Perú; 2022.
Some similar items:
- Eliana P. Calvo, María O. Rojas, Jacqueline Chaparro, Moisés Wasserman, Expresion of the multiple drug resistance associated gene (pfMDR 1) in Colombian strains of Plasmodium falciparum , Biomedica: Vol. 19 No. 3 (1999)
- Iveth J. González, Metacaspases and their role in the life cycle of human protozoan parasites , Biomedica: Vol. 29 No. 3 (2009)
- Nancy Arróspide, Hernán Sanabria, William J. Araujo-Banchon, Evaluation of the effectiveness of the OptiMAL-IT™ rapid test in the follow up of patients diagnosed with malaria in the Peruvian Amazon , Biomedica: Vol. 42 No. 1 (2022)
- Ana María Vásquez, Felipe Sanín, Luis Gonzalo Álvarez, Alberto Tobón, Alexandra Ríos, Silvia Blair, Therapeutic efficacy of a regimen of artesunate-mefloquine-primaquine treatment for Plasmodium falciparum malaria and treatment effects on gametocytic development , Biomedica: Vol. 29 No. 2 (2009)
- César Segura, Silvia Blair, Mitochondria in the Plasmodium genera. , Biomedica: Vol. 23 No. 3 (2003)
- Andrés Fernando Rodríguez-Gutiérrez, Isabel Cristina Ramírez-Sánchez, Malaria after liver transplantation: Report of two cases and a review of published cases , Biomedica: Vol. 45 No. 2 (2025)
- Jaime Carmona-Fonseca, Eliana Arango, Silvia Blair, Gametocytemia in falciparum malaria treated with amodiaquine or artesunate , Biomedica: Vol. 28 No. 2 (2008)
- Carolina Montoya, Priscila Bascuñán, Julián Rodríguez-Zabala, Margarita M. Correa, Abundance, composition and natural infection of Anopheles mosquitoes from two malaria-endemic regions of Colombia , Biomedica: Vol. 37 No. Sup. 2 (2017): Suplemento 2, Entomología médica, 2017
- Pablo Chaparro, Edison Soto, Julio Padilla, Daniel Vargas, Estimation of the underreporting of malaria measurement in ten municipalities of the Pacific coast of Nariño during 2009 , Biomedica: Vol. 32 (2012): Suplemento 1, Malaria
- Juan Gabriel Piñeros, Margarita Arboleda, Juan Camilo Jaramillo, Silvia Blair, Report of five cases of severe neonatal Plasmodium vivax malaria in Urabá, Colombia , Biomedica: Vol. 28 No. 4 (2008)
Copyright (c) 2023 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























