Sodium caseinate increases the number of B lymphocytes in mouse
Abstract
Introduction: Sodium caseinate, a casein salt, is a proinflammatory agent in mice, and it is able to induce granulopoiesis in vivo and to increase the production of cytokines, which is key for this biological process.
Objective: To assess whether sodium caseinate is able to induce a biological effect on cells from lymphoid origin and the production of cytokines involved in this lineage in vivo.
Materials and methods: We used female BALB /c mice from 8 to 12 weeks old. The animals were injected intraperitoneally (IP) with 1 ml of sodium caseinate (10% PBS w/v) four times every 48 hours. The B cell populations and the incorporation of BrdU were analyzed by flow cytometry. Detection of interleukin-7 was assessed by ELISA (Enzyme-Linked ImmunoSorbent Assay).
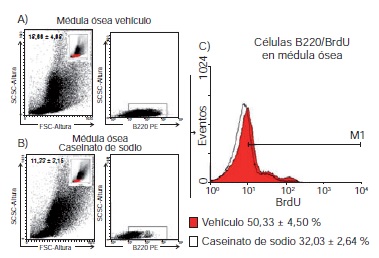
Results: We established that after intraperitoneal injection, the number of B lymphocytes 220+ from the spleen of mice treated with sodium caseinate increased compared to those that only received the vehicle (89.01±1.03 vs 75.66 ± 2.08), and the same was observed with the incorporation of BrdU in B220 + cells (38.59±4.48 vs 11.82±1.04 respectively). We also established that the concentration of interleukin-7 (IL-7) in the serum of mice treated with sodium caseinate increased compared to those that only received the vehicle (62.1 ± 17.5 vs 26.9 ± 4.4 pg/ml).
Conclusion: Sodium caseinate was able to increase the number of B lymphocytes in the spleen; it also induced IL-7 production, a cytokine that is key for the B cell lymphopoiesis.
Downloads
References
Lotem J, Sachs L. Control of in vivo differentiation of myeloid leukemic cells. Leukemia. 1988;2:24s-37s.
Lotem J, Sachs L. Independent regulation of myeloid cell growth and differentiation inducing proteins: In vivo regulation by compounds that induce inflammation. Int J Cancer. 1985;35:93-100.
Santiago-Osorio E, Mora L, Bautista M, Montesinos JJ, Martínez I, Ramos-Mandujano G, et al. Sodium caseinate induces secretion of macrophage colony-stimulating factor from neutrophils. Immunobiology. 2010;215:332-9. https://doi.org/10.1016/j.imbio.2009.03.003
Córdova-Galaviz Y, Ledesma-Martínez E, Aguíñiga-Sánchez I, Soldevila-Melgarejo G, Soto-Cruz I, Weiss-Steider B, et al. Sodium caseinate induces increased survival in leukaemic mouse J774 model. In Vivo. 2014;28: 819-25.
Santiago-Osorio E, Ledesma-Martínez E, Aguiñiga-Sánchez I, Poblano-Pérez I, Weiss-Steider B, Montesinos-Montesinos JJ, et al. Sodium caseinate (CasNa) induces mobilization of hematopoietic stem cells in a BALB/c mouse model. Med Sci Monit Basic Res. 2015;21:206-12. https://doi.org/10.12659/MSMBR.895442
Domínguez-Meléndez V, Silvestre-Santana O, Moreno-Fierros L, Aguiñiga-Sánchez I, Martínez L, Marroquín-Segura R, et al. Sodium caseinate induces mouse granulopoiesis. Inflamm Res. 2012;61:367-73. https://doi.org/10.1007/s00011-011-0421-7
Metcalf D, Robb L, Dunn AR, Mifsud S, Di Rago L. Role of granulocyte-macrophage colony-stimulating factor and granulocyte colony stimulating factor in the development of an acute neutrophil inflammatory response in mice. Blood. 1996;88:3755-64.
Wong CW, Seow HF, Liu AH, Husband AJ, Smithers GW, Watson DL. Modulation of immune responses by bovine beta-casein. Immunol Cell Biol. 1996;74:323-9. https://doi.org/10.1038/icb.1996.58
Tobita K, Kawahara T, Otani H. Bovine beta-casein (1-28), a casein phosphopeptide, enhances proliferation and IL-6 expression of mouse CD19+ cells via Toll-like receptor 4. J Agric Food Chem. 2006;54:8013-7. https://doi.org/10.1021/jf0610864
Ma A, Koka R, Burkett P. Diverse functions of IL-2, IL-15, and IL-7 in lymphoid homeostasis. Annu Rev Immunol. 2006;24:657-79. https://doi.org/10.1146/annurev.immunol.24.021605.090727
Sitnicka E, Bryder D, Theilgaard-Mönch K, Buza-Vidas N, Adolfsson J, Jacobsen SE. Key role of flt3 ligand in regulation of the common lymphoid progenitor but not in maintenance of the hematopoietic stem cell pool. Immunity. 2002;17:463-72. https://doi.org/10.1016/S1074-7613(02)00419-3
Peschon JJ, Morrissey PJ, Grabstein KH, Ramsdell FJ, Maraskovsky E, Gliniak BC, et al. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J Exp Med. 1994;180:1955-60. https://doi.org/10.1084/jem.180.5.1955
Instituto de Biotecnología, Universidad Nacional Autónoma de México. Norma Oficial Mexicana NOM-062-ZOO-1999. Especificaciones técnicas para la producción, cuidado y uso de los animales de laboratorio. Fecha de consulta: 15 de enero de 2010. Disponible en: http://www.ibt.unam.mx/computo/pdfs/bioterio.NOM-062.pdf
Bertrand JY, Giroux S, Golub R, Klaine M, Jalil A, Boucontet L, et al. Characterization of purified intra-embryonic hematopoietic stem cells as a tool to define their site of origin. Proc Natl Acad Sci USA. 2005;102:134-9. https://doi.org/10.1073/pnas.0402270102
Cyster JG. Chemokines, sphingosine-1-phosphate, and cell migration in secondary lymphoid organs. Annu Rev Immunol. 2005;23:127-59. https://doi.org/10.1146/annurev.immunol.23.021704.115628
Fu YX, Chaplin DD. Development and maturation of secondary lymphoid tissues. Annu Rev Immunol. 1999;17: 399-433. https://doi.org/10.1146/annurev.immunol.17.1.399
Dias S, Silva H Jr, Cumano A, Vieira P. Interleukin-7 is necessary to maintain the B cell potential in common lymphoid progenitors. J Exp Med. 2005;201:971-9. https://doi.org/10.1084/jem.20042393
Milne CD, Paige CJ. IL-7: A key regulator of B lympho-poiesis. Semin Immunol. 2006;18:20-30. https://doi.org/10. 1016/j.smim.2005.10.003
Ueda Y, Yang K, Foster SJ, Kondo M, Kelsoe G. Inflam-mation controls B lymphopoiesis by regulating chemokine CXCL12 expression. J Exp Med. 2004;199:47-58. https://doi.org/10.1084/jem.20031104
Ueda Y, Kondo M, Kelsoe G. Inflammation and the reciprocal production of granulocytes and lymphocytes in bone marrow. J Exp Med. 2005;201:1771-80. https://doi.org/10.1084/jem.20041419
von Freeden-Jeffry U, Vieira P, Lucian LA, McNeil T, Burdach SE, Murray R. Lymphopenia in interleukin (IL)-7 gene-deleted mice identifies IL-7 as a nonredundant cytokine. J Exp Med. 1995;181:1519-26. https://doi.org/10. 1084/jem.181.4.1519
Morrison SJ, Wright DE, Weissman IL. Cyclophosphamide/granulocyte colony-stimulating factor induces hematopoietic stem cells to proliferate prior to mobilization. Proc Natl Acad Sci USA. 1997;94:1908-13
Inra CN, Zhou BO, Acar M, Murphy MM, Richardson J, Zhao Z, et al. A perisinusoidal niche for extramedullary haematopoiesis in the spleen. Nature. 2015;26:466-71. https://doi.org/10.1038/nature15530
Some similar items:
- Luis A. Franco, Germán E. Matiz, Jairo Calle, Roberto Pinzón, Luis F. Ospina, Antiinflammatory activity of extracts and fractions obtained from Physalis peruviana L. calyces , Biomedica: Vol. 27 No. 1 (2007)
- Luis Ángel Villar, Rosa Margarita Gélvez, Jairo Antonio Rodríguez, Doris Salgado, Beatriz Parra, Lyda Osorio, Irene Bosch, Biomarkers for the prognosis of severe dengue , Biomedica: Vol. 33 (2013): Suplemento 1, Fiebres hemorrágicas
- Wilson Mejía-Naranjo, Myriam Sánchez-Gomez, Protein malnutrition up-regulates growth hormone receptor expression in rat splenic B lymphocytes. , Biomedica: Vol. 24 No. 4 (2004)
- Adriana Cuéllar, Angela Fonseca, Alberto Gómez, Effect of lipopolysaccharides on human dendritic cell cultures and its inhibition by polymyxin B. , Biomedica: Vol. 24 No. 4 (2004)
- Fabián Jaimes, Gisela de la Rosa, Anticoagulation and sepsis: the opportunity for a new use of heparin?. , Biomedica: Vol. 26 No. 1 (2006)
- Henry A. Vargas, Martín Rondón, Rodolfo Dennis, Pharmacological treatment and impairment of pulmonary function in patients with type 2 diabetes: a cross-sectional study , Biomedica: Vol. 36 No. 2 (2016)
- Tania Yadira Martínez-Rodríguez, Mauricio Rey-Buitrago, Alpha sinuclein expression in blood and its relationship with chronic constipation in a population from Bogotá, D.C., with problems of alcohol consumption , Biomedica: Vol. 40 No. 2 (2020)
- Concepción Sánchez-Martínez , Liliana Torres-González , Gabriela Alarcón-Galván , Linda E. Muñoz-Espinosa , Homero Zapata-Chavira , Diana Patricia Moreno-Peña, Homero Náñez-Terreros, Edelmiro Pérez-Rodríguez , Lourdes Garza-Ocañas , Francisco Javier Guzmán-de la Garza , Paula Andrea Cordero, Anti-inflammatory and antioxidant activity of essential amino acid α-ketoacid analogues against renal ischemia–reperfusion damage in Wistar rats , Biomedica: Vol. 40 No. 2 (2020)
- Wbeimar Aguilar-Jiménez, Lizdany Flórez-Álvarez, Daniel S. Rincón, Damariz Marín-Palma, Alexandra Sánchez-Martínez, Jahnnyer Martínez, María Isabel Zapata, John D. Loaiza, Constanza Cárdenas, Fanny Guzmán, Paula A. Velilla, Natalia A. Taborda, Wildeman Zapata, Juan C. Hernández, Francisco J. Díaz, María T. Rugeles, Immune characterization of a Colombian family cluster with SARS-CoV-2 infection , Biomedica: Vol. 41 No. Sp. 2 (2021): Octubre, Infecciones bacterianas y virales
- Alicia Norma Alayón, Ana Patricia Rivadeneira, Carlos Herrera, Heidy Guzmán, Dioneris Arellano, Isabella Echeverri, Metabolic and inflammatory postprandial effect of a highly saturated fat meal and its relationship to abdominal obesity , Biomedica: Vol. 38 No. Sup.1 (2018): Suplemento 1, Enfermedades crónicas
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























