Spatial modeling of soil-transmitted helminthiases in Colombia under climate change scenarios
Abstract
Introduction. Soil-transmitted helminthiases remain a significant public health burden in Colombia, especially in rural and tropical areas. Climate change is expected to alter environmental conditions that favor the survival and transmission of Ascaris lumbricoides, Trichuris trichiura, and hookworms.
Objective. To estimate the current spatial distribution of these infections and project prevalence changes by 2035 under climate change scenarios, with and without public health interventions.
Materials and methods. An ecological study with spatial modeling was conducted, integrating epidemiological, climatic, and biological data. Baseline prevalence data were obtained from the Encuesta Nacional de Parasitismo Intestinal (2012-2014). Climate projections from the ERA5-Land satellite product (2024-2035) were used alongside generalized additive models to estimate environmental suitability. A systematic review defined optimal temperature and humidity thresholds for the development of infective stages. Two scenarios were modeled: one without intervention and another with mass drug administration and improved sanitation.
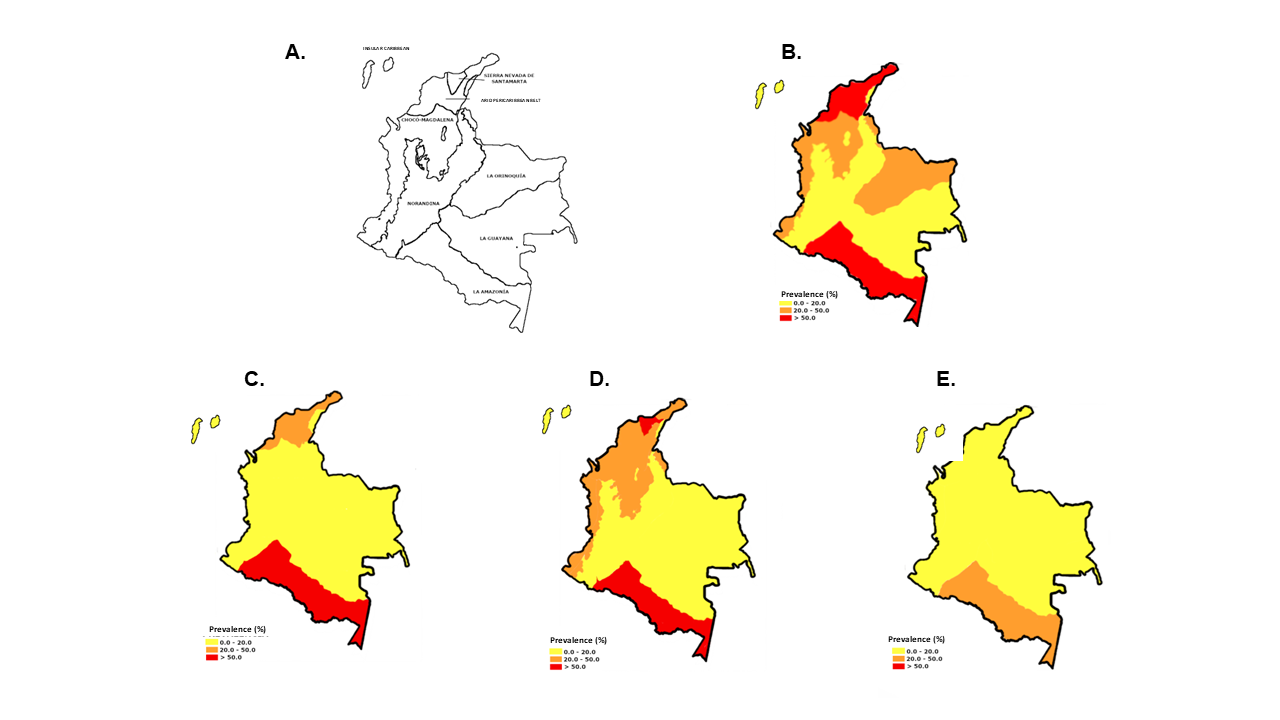
Results. Baseline prevalence was 11.3% for A. lumbricoides, 18.4% for T. trichiura, and 6.4% for hookworms, with highest rates in Amazonia and the Sierra Nevada de Santa Marta. In a no-intervention scenario, projected prevalences increased to 13.6, 21.2, and 8.0%, respectively. The intervention scenario reduced these to 6.8%, 12.7%, and 5.6%. Temperature and humidity were strong positive predictors (p < 0.01), while altitude and forest cover showed negative associations.
Conclusions. Climate change may intensify soil-transmitted helminthiases transmission in Colombia by 2035. However, sustained control strategies could significantly mitigate this impact. Spatial modeling offers a valuable tool to guide targeted interventions and inform public health planning.
Downloads
References
World Health Organization. Soil-transmitted helminth infections. Accessed: July 30, 2025. Available at: https://www.who.int/news-room/fact-sheets/detail/soil-transmitted-helminthinfections
Jourdan PM, Lamberton PHL, Fenwick A, Addiss DG. Soil-transmitted helminth infections. Lancet. 2018;391:252-65. https://doi.org/10.1016/S0140-6736(17)31930-X
Clarke NE, Clements AC, Doi SA, Wang D, Campbell SJ, Gray D, et al. Differential effect of mass deworming and targeted deworming for soil-transmitted helminth control in children: A systematic review and meta-analysis. Lancet. 2017;389:287-97. https://doi.org/10.1016/S0140-6736(16)32123-7
World Health Organization. Preventive chemotherapy in human helminthiasis. Coordinated use of anthelminthic drugs in control interventions: a manual for health professionals and programme managers. Geneva: World Health Organization; 2006. p. 1-62.
Ministerio de Salud y Protección Social, Universidad de Antioquia. Encuesta Nacional de Parasitismo intestinal en Población Escolar 2012-2014. Medellín: Universidad de Antioquia, Ministerio de Salud y Protección Social; 2015. p. 1-173.
Pazmiño FA, Mora-Salamanca AF, Mahecha BSP, Moreno EJP, Olivera MJ, Ospina AK, et al. Prevalence of intestinal parasitism in preschool and school children in Colombia: Systematic review and meta-analysis. Trop Med Int Health. 2022;27:781-94. https://doi.org/10.1111/tmi.13800
Botero D, Restrepo M. Parasitosis humanas. 5a edición. Medellín: Corporación para Investigaciones Biológica; 2012. p. 1-723.
Vanalli C, Mari L, Casagrandi R, Gatto M, Cattadori IM. Helminth ecological requirements shape the impact of climate change on the hazard of infection. Ecol Lett. 2024;27:e14386. https://doi.org/10.1111/ele.14386
Booth M. Climate change and the neglected tropical diseases. Adv Parasitol. 2018;100:39-126. https://doi.org/10.1016/bs.apar.2018.02.001
Chen J, Gong Y, Chen Q, Li S, Zhou Y. Global burden of soil-transmitted helminth infections, 1990-2021. Infect Dis Poverty. 2024;13:77. https://doi.org/10.1186/s40249-024-01238-9
Tidman R, Abela-Ridder B, de Castañeda RR. The impact of climate change on neglected tropical diseases: A systematic review. Trans R Soc Trop Med Hyg. 2021;115:147-68. https://doi.org/10.1093/trstmh/traa192
Schuele SA, Clowes P, Kowuor DO, Nsojo A, Mangu C, Riess H, et al. Ascaris lumbricoides infection and its relation to environmental factors in the Mbeya region of Tanzania, a crosssectional, population-based study. PLoS ONE. 2014;9:e92032. https://doi.org/10.1371/journal.pone.0092032
Kim M, Pyo K, Hwang Y, Park kh, Hwang IG, Chai JY, et al. Effect of temperature on embryonation of Ascaris suum eggs in an environmental chamber. Korean J Parasitol. 2012;50:239-42. https://doi.org/10.3347/kjp.2012.50.3.239
Chammartin F, Guimarães LH, Scholte RG, Bavia ME, Utzinger J, Vounatsou P. Spatiotemporal distribution of soil-transmitted helminth infections in Brazil. Parasit Vectors. 2014;18:440. https://doi.org/10.1186/1756-3305-7-440
Huang SY, Lai YS, Fang YY. The spatial-temporal distribution of soil-transmitted helminth infections in Guangdong Province, China: A geostatistical analysis of data derived from the three national parasitic surveys. PLoS Negl Trop Dis. 2022;16:e0010622. https://doi.org/10.1371/journal.pntd.0010622
Hernández J, Hurtado A, Ortiz R, Walschburger T. Unidades biogeográficas de Colombia. En la diversidad biológica de Iberoamérica. 1a edición. Xalapa: Acta Zoológica Mexicana; 1992. p. 95-135.
Mitra AK, Mawson AR. Neglected tropical diseases: Epidemiology and global burden. Trop Med Infect Dis. 2017;2:36. https://doi.org/10.3390/tropicalmed2030036
Carlson CJ, Albery GF, Merow C, Trisos CH, Zipfel CM, Eskew EA, et al. Climate change increases cross-species viral transmission risk. Nature. 2022;607:555-62. https://doi.org/10.1038/s41586-022-04788-w
Caminade C, McIntyre KM, Jones AE. Impact of recent and future climate change on vectorborne diseases. Ann NY Acad Sci. 2019;1436:157-73. https://doi.org/10.1111/nyas.13950
Rocklöv J, Dubrow R. Climate change: An enduring challenge for vector-borne disease prevention and control. Nat Immunol. 2020;21:479-83. https://doi.org/10.1038/s41590-020-0648-y
Saboyá M, Catalá L, Ault S, Nicholls R. Prevalence and intensity of infection of soil-transmitted helminths in Latin America and the Caribbean countries: Mapping at second administrative level 2000-2010. Washington, D.C: Pan American Health Organization; 2011. p. 1-106.
Candela E, Goizueta C, Sandon L, Muñoz-Antoli C, Periago MV. The relationship between soil-transmitted helminth infections and environmental factors in Puerto Iguazú, Argentina: Cross-sectional study. JMIR Public Health Surveill. 2023;9:e41568. https://doi.org/10.2196/41568
Maddren R, Phillips A, Ower A, Landeryou T, Mengistu B, Anjulo U, et al. Soil-transmitted helminths and schistosome infections in Ethiopia: A systematic review of progress in their control over the past 20 years. Parasit Vectors. 2023;16:203. https://doi.org/10.1186/s13071-023-05815-z
Sartorius B, Cano J, Simpson H, Tusting LS, Marczak LB, Miller-Petrie MK, et al. Prevalence and intensity of soil-transmitted helminth infections of children in sub-Saharan Africa, 2000-18: A geospatial analysis. Lancet Glob Health. 2021;9:e52-e60. https://doi.org/10.1016/S2214-109X(20)30398-3
Newbold LK, Burthe SJ, Oliver AE, Gweon HS, Barnes CJ, Daunt F, et al. Helminth burden and ecological factors associated with alterations in wild host gastrointestinal microbiota. ISME J. 2017;11:663-75. https://doi.org/10.1038/ismej.2016.153
Hall A, Holland C. Geographical variation in Ascaris lumbricoides fecundity and implications for helminth control. Parasitol Today. 2000;16:540-4. https://doi.org/10.1016/s0169-4758(00)01779-8
Ali A, Niaz S, Nasreen N, Khan A, Shafeeq S, Aguilar-Marcelino L, et al. Temporal and spatial patterns of Trichuris trichiura eggs: A potential threat to human health in Pakistan. Helminthologia. 2024;61:11-9. https://doi.org/10.2478/helm-2024-0009
Pullan RL, Kabatereine NB, Quinnell RJ, Brooker S. Spatial and genetic epidemiology of hookworm in a rural community in Uganda. PLoS Negl Trop Dis. 2010;4:e713. https://doi.org/10.1371/journal.pntd.0000713
Chammartin F, Scholte RG, Malone JB, Bavia ME, Nieto P, Utzinger J, et al. Modelling the geographical distribution of soil-transmitted helminth infections in Bolivia. Parasit Vectors. 2013;6:152. https://doi.org/10.1186/1756-3305-6-152
Weatherhead JE, Hotez PJ. Worm infections in children. Pediatr Rev. 2015;36:341-52. https://doi.org/10.1542/pir.36-8-341
Kim MK, Pyo KH, Hwang YS, Park KH, Hwang IG, Chai JY, et al. Effect of temperatura on embryonation of Ascaris suum eggs in an environmental chamber. Korean J Parasitol. 2012;50:239-42. https://doi.org/10.3347/kjp.2012.50.3.239
Weaver HJ, Hawdon JM, Hoberg EP. Soil-transmitted helminthiases: Implications of climate change and human behaviour. Trends Parasitol. 2010;26:574-81. https://doi.org/10.1016/j.pt.2010.06.009
Bethony J, Brooker S, Albonico M, Geiger SM, Loukas A, Diemert D, et al. Soil-transmitted helminth infections: ascariasis, trichiuriasis, and hookworm. Lancet. 2006;367:1521-32. https://doi.org/10.1016/S0140-6736(06)68653-4
Kaliappan S, Ramanujam K, Manuel M, Farzana J, Janagaraj V, Laxmanan S, et al. Soiltransmitted helminth infections after mass drug administration for lymphatic filariasis in rural southern India. Trop Med Int Health. 2022;27:81-91 https://doi.org/10.1111/tmi.13697
Servián A, Garimano N, Santini MS. Systematic review and meta-analysis of soil-transmitted helminth infections in South America (2000-2024). Acta Trop. 2024;260:107400. https://doi.org/10.1016/j.actatropica.2024.107400
Savioli L, Albonico M, Engels D, Montresor A. Progress in the prevention and control of schistosomiasis and soil-transmitted helminthiasis. Parasitol Int. 2004;53:103-13. https://doi.org/10.1016/j.parint.2004.01.001
Some similar items:
- Óscar René Sorto, Alexandra Manoella Portillo, Miguel Ángel Aragón, Martha IdalÍ Saboyá, María Paz Ade, Miguel Ángel Minero, Marta Alicia Hernández, Amada Gloria Mena, Rodolfo Peña, Victor Manuel Mejía, Keith Carter, Prevalence and intensity of infection by soil-transmitted helminths and prevalence of malaria among schoolchildren in El Salvador , Biomedica: Vol. 35 No. 3 (2015)
- Luis Caraballo, Josefina Zakzuk, The evolution of the Th2 immune responses and its relationships with parasitic diseases and allergy , Biomedica: Vol. 32 No. 1 (2012)
Copyright (c) 2025 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |
Funding data

























