Outbreaks and incidence of vector-borne diseases in Colombia (2007-2024): Impact of climate change and deforestation
Abstract
Introduction. Vector-borne diseases pose a public health challenge in Colombia, influenced by climatic and environmental factors. El Niño and deforestation can alter vector habitats, affecting the incidence of dengue, Zika, chikungunya, malaria, cutaneous leishmaniasis, and yellow fever.
Objective. This study analyzes the relationship between these variables and vector-borne diseases incidence in Colombia (2007-2024).
Materials and methods. An ecological study was conducted using incidence and outbreak data for six vector-borne diseases, linked to climate information, El Niño, and deforestation. Regression models and random forests were applied to assess associations.
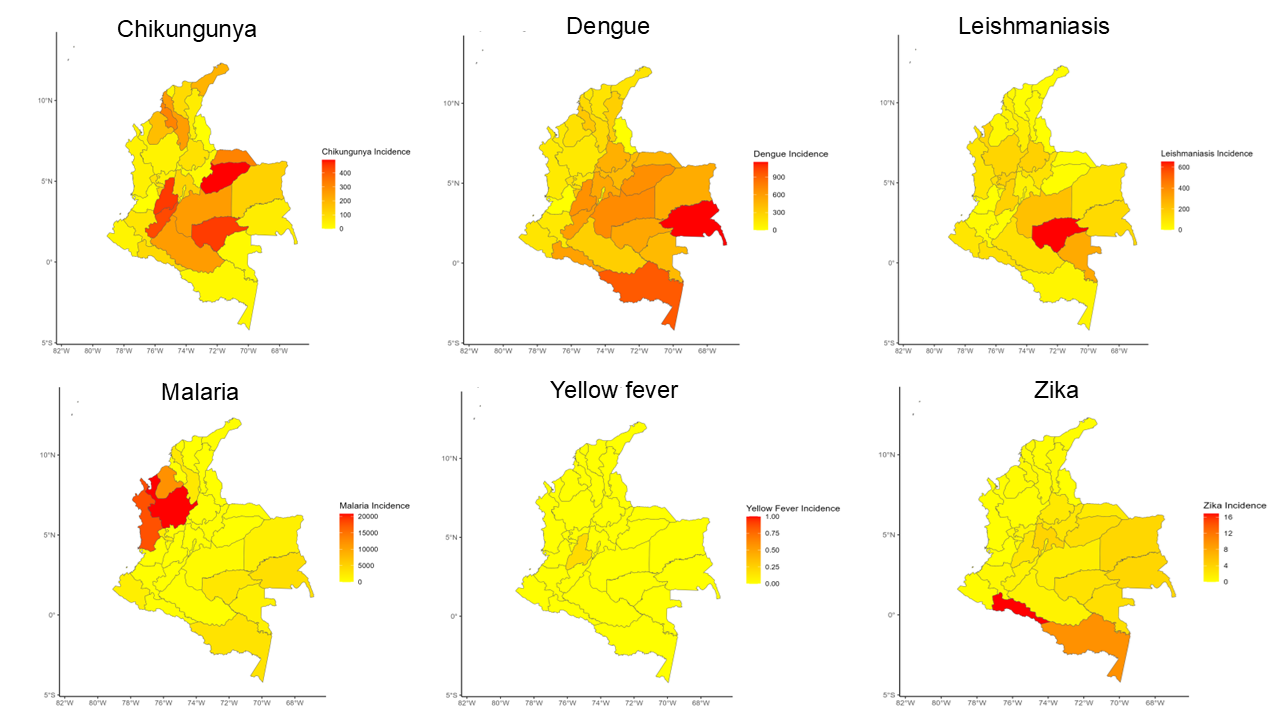
Results. Between 2007 and 2024, 3,283,259 cases of vector-borne diseases were reported in Colombia. Of these, 49.9% (1,639,120) were dengue and 39.8% (1,307,351) malaria, accounting for 89.7% of total cases. El Niño was associated with increased incidence of dengue (β = 213.24; 95% CI: 86.05-338.43), chikungunya (β = 26.41; 95% CI: 17.54-70.36), and Zika (β = 14.12; 95% CI: 10.06-89.30). Maximum temperature showed a positive relationship with dengue (β = 5.74; 95% CI: 2.15-13.63) and malaria (β = 17.28; 95% CI: 3.81-30.75). Deforestation was associated with malaria (β = 12.35; 95% CI: 4.62-20.08) and cutaneous leishmaniasis (β = 8.67; 95% CI: 2.21-15.13). Mean precipitation had negative associations with chikungunya and leishmaniasis.
Conclusions: Climate change and deforestation impact the epidemiology of vector-borne diseases in Colombia. Integrated public health and environmental conservation strategies are needed to mitigate their effects.
Downloads
References
Molina-Guzmán LP, Gutiérrez-Builes LA, Ríos-Osorio LA. Models of spatial analysis for vector-borne diseases studies: A systematic review. Vet World. 2022;15:1975-89. https://doi.org/10.14202/vetworld.2022.1975-1989
El-Sayed A, Kamel M. Climatic changes and their role in emergence and re-emergence of diseases. Environ Sci Pollut Res Int. 2020;27:22336-52. https://doi.org/10.1007/s11356-020-08896-w
Yadav N, Upadhyay RK. Global effect of climate change on seasonal cycles, vector population and rising challenges of communicable diseases: A review. J Atmospheric Sci Res. 2023;6:21-59. https://doi.org/10.30564/jasr.v6i1.5165
Cai W, Santoso A, Collins M, Dewitte B, Karamperidou C, Kug J-S, et al. Changing El Niño- Southern oscillation in a warming climate. Nat Rev Earth Environ. 2021;2:628-44. https://doi.org/10.1038/s43017-021-00199-z
Chretien J-P, Anyamba A, Small J, Britch S, Sánchez JL, Halbach AC, et al. Global climate anomalies and potential infectious disease risks: 2014-2015. PLoS Curr. 2015;26:7. https://doi.org/10.1371/currents.outbreaks.95fbc4a8fb4695e049baabfc2fc8289f
Ortiz DI, Piche-Ovares M, Romero-Vega LM, Wagman J, Troyo A. The impact of deforestation, urbanization, and changing land use patterns on the ecology of mosquito and tick-borne diseases in Central America. Insects. 2021;13:20. https://doi.org/10.3390/insects13010020
Segura NA, Muñoz AL, Losada-Barragán M, Torres O, Rodríguez AK, Rangel H, et al. Minireview: Epidemiological impact of arboviral diseases in Latin American countries, arbovirus-vector interactions and control strategies. Pathog Dis. 2021;79:ftab043. https://doi.org/10.1093/femspd/ftab043
Khan A, Yasin M, Aqueel MA, Farooqi MA, Akram MI, Yousuf HMB, et al. Vector-borne disease and climate change. In: Shields VDC, editor. Arthropods-new advances and perspectives. IntechOpen; 2023. https://doi.org/10.5772/intechopen.107120
Padilla-Rodríguez JC, Olivera MJ, Padilla MC, Abril EP. Malaria epidemics in Colombia, 1970-2019. Rev Soc Bras Med Trop. 202229:55:e05592021. https://doi.org/10.1590/0037-8682-0559-2021
Cardona-Arias JA, Salas-Zapata W, Carmona-Fonseca J. A systematic review of mixed studies on malaria in Colombia 1980-2022: What the “bifocal vision” discovers. BMC Public Health. 2023;23:1169. https://doi.org/10.1186/s12889-023-16098-5
Freitas LP, Carabali M, Yuan M, Jaramillo-Ramírez GI, Balaguera CG, Restrepo BN, et al. Spatio-temporal clusters and patterns of spread of dengue, chikungunya, and Zika in Colombia. PLoS Negl Trop Dis. 2022;16:e0010334. https://doi.org/10.1371/journal.pntd.0010334
Mantilla-Granados JS, Sarmiento-Senior D, Manzano J, Calderón-Peláez M-A, Velandia-Romero ML, Buitrago LS, et al. Multidisciplinary approach for surveillance and risk identification of yellow fever and other arboviruses in Colombia. One Health. 2022:15:100438. https://doi.org/10.1016/j.onehlt.2022.100438
Thomson MC, Stanberry LR. Climate change and vectorborne diseases. N Engl J Med. 2022 4;387:1969-78. https://doi.org/10.1056/NEJMra2200092
Colón-González FJ, Sewe MO, Tompkins AM, Sjödin H, Casallas A, Rocklöv J, et al. Projecting the risk of mosquito-borne diseases in a warmer and more populated world: a multi-model, multi-scenario intercomparison modelling study. Lancet Planet Health. 2021;5:e404-e414 e404-14. https://doi.org/10.1016/S2542-5196(21)00132-7
Colonia CB, Vásquez-Rodríguez AB, Alexander N, de la Hoz Restrepo F. Malaria, relationship with climatic variables and deforestation in Colombia, Latin America and the Caribbean from 2000 to 2020: A systematic review. Malaria J. 2024;23:347. https://doi.org/10.1186/s12936-024-05140-5
Marinho RdSS, Duro RLS, Mota MTdO, Hunter J, Diaz RS, Kawakubo FS, et al. Environmental changes and the impact on the human infections by dengue, chikungunya and Zika viruses in northern Brazil, 2010-2019. Int J Environ Res Public Health. 2022;19:12665. https://doi.org/10.3390/ijerph191912665
Romero-Vega LM, Piche-Ovares M, Soto-Garita C, Barantes Murillo DF, Chaverri LG, Alfaro-Alarcón A, et al. Seasonal changes in the diversity, host preferences and infectivity of mosquitoes in two arbovirus-endemic regions of Costa Rica. Parasit Vectors. 2023;16:34. https://doi.org/10.1186/s13071-022-05579-y
Gagnon AS, Bush AB, Smoyer-Tomic KE. Dengue epidemics and the El Niño southern oscillation. Int J Biometeorol. 2002;46:81-9. https://doi.org/10.1007/s00484-001-0119-6 19. Petrova D, Lowe R, Stewart-Ibarra A, Ballester J, Koopman SJ, Rodó X. Sensitivity of large dengue epidemics in Ecuador to long-lead predictions of El Niño. Climate Services. 2019;15:100096. https://doi.org/10.1016/j.cliser.2019.02.003
Ruiz-Polo AA, Santillan-Valdivia RE, Saavedra-Ríos CY, Núñez-Rodriguez CM, Niño-Mendoza LE. Comportamiento alimentario de Aedes aegypti en brotes de dengue de dos zonas rurales del Perú durante el ciclón Yaku y El Niño Global del 2023. Revista Peruana de Medicina Experimental y Salud Pública. 2024;41:266-72.
Ruiz-Polo AA, Santillan-Valdivia RE, Saavedra-Ríos CY, Núñez-Rodríguez CM, Niño-Mendoza LE. Aedes aegypti feeding behavior during dengue outbreaks in two rural áreas of Perú during the Yaku cyclone and El Niño phenomenon of 2023. Revista Peruana de Medicina Experimental y Salud Pública. 2024;41:266-72.
Messina JP, Kraemer MU, Brady OJ, Pigott DM, Shearer FM, Weiss DJ, et al. Mapping global environmental suitability for Zika virus. Elife. 2016:5:e15272. https://doi.org/10.7554/eLife.15272
Ruiz-Polo AA, Barrera-Rivera LV. Efecto de la temperatura en el ciclo biológico del Aedes aegypti en condiciones de laboratorio. Revista Peruana de Medicina Experimental y Salud Pública. 2024;41:327-8.
Valdez LD, Sibona GJ, Condat C. Impact of rainfall on Aedes aegypti populations. Ecological Modelling. 2018;385:96-105. https://doi.org/10.1016/j.ecolmodel.2018.07.003
Padilla JC, Lizarazo FE, Murillo OL, Mendigaña FA, Pachón E, Vera MJ. Epidemiología de las principales enfermedades transmitidas por vectores en Colombia, 1990-2016. Biomédica. 2017;37(Supl.2) :27-40. https://doi.org/10.7705/biomedica.v37i0.3769
Guzmán NA, Zakzuk J, Moranth RV, Olaciregui AA, Padilla DP. Dengue, Chikunguña y Zika en Colombia 2015-2016. Revista MVZ (Córdoba). 2017;22:2.
Paaijmans KP, Blanford S, Bell AS, Blanford JI, Read AF, Thomas MB. Influence of climate on malaria transmission depends on daily temperature variation. Proc Natl Acad Sci USA. 2010;107:15135-9. https://doi.org/10.1073/pnas.1006422107
Laporta GZ, Ilacqua RC, Bergo ES, Chaves LS, Rodovalho SR, Moresco GG, et al. Malaria transmission in landscapes with varying deforestation levels and timelines in the Amazon: A longitudinal spatiotemporal study. Sci Rep. 2021;11:6477. https://doi.org/10.1038/s41598-021-85890-3
Laporta GZ. Amazonian rainforest loss and declining malaria burden in Brazil. Lancet Planet Health. 2019;3:e4-e5. https://doi.org/10.1016/S2542-5196(18)30243-2
Valle D, Clark J. Improving the modeling of disease data from the government surveillance system: A case study on malaria in the Brazilian Amazon. PLoS Comput Biol. 2013;9:e1003312. https://doi.org/10.1371/journal.pcbi.1003312
Valle D, Clark J. Conservation efforts may increase malaria burden in the Brazilian Amazon. PLoS ONE. 2013;8:e57519. https://doi.org/10.1371/journal.pone.0057519
Hahn MB, Olson SH, Vittor AY, Barcellos C, Patz JA, Pan W. Conservation efforts and malaria in the Brazilian Amazon. Am J Trop Med Hyg. 2014;90:591-4. https://doi.org/10.4269/ajtmh.13-0323
Rodrigues MGdA, Sousa JDdB, Dias ÁLB, Monteiro WM, Sampaio VdS. The role of deforestation on American cutaneous leishmaniasis incidence: Spatial-temporal distribution, environmental and socioeconomic factors associated in the Brazilian Amazon. Trop Med Int Health. 2019;24:348-55. https://doi.org/10.1111/tmi.13196
Burkett-Cadena ND, Vittor AY. Deforestation and vector-borne disease: Forest conversion favors important mosquito vectors of human pathogens. Basic and applied ecology. 2018;26:101-10. Basic Appl Ecol. 2018:26:101-10. https://doi.org/10.1016/j.baae.2017.09.012
Kocher A, Cornuault J, Gantier Jc, Manzi S, Chavy A, Girod R, et al. Biodiversity and vectorborne diseases: Host dilution and vector amplification occur simultaneously for Amazonian leishmaniases. Mol Ecol. 2023;32:1817-31. https://doi.org/10.1111/mec.16341
Caminade C, McIntyre KM, Jones AE. Impact of recent and future climate change on vectorborne diseases. Ann N Y Acad Sci. 2019;1436:157-73. https://doi.org/10.1111/nyas.13950
Mordecai EA, Caldwell JM, Grossman MK, Lippi CA, Johnson LR, Neira M, et al. Thermal biology of mosquito-borne disease. Ecol Lett. 2019;22:1690-708. https://doi.org/10.1111/ele.13335
Molleda P, Serra GV. El Niño Southern Oscillation and the prevalence of infectious diseases. La Granja: revista de Ciencias de la Vida. 2024;40:9-37. https://doi.org/10.17163/lgr.n40.2024.01
Kemarau RA, Eboy OV. Spatial-temporal distribution of malaria risk and its association with El Niño Southern Oscillation (ENSO). Malaysian Journal of Social Sciences and Humanities. 2021;6:276-86. https://doi.org/10.47405/mjssh.v6i4.768
Fletcher IK, Grillet ME, Moreno JE, Drakeley C, Hernández-Villena J, Jones KE, et al. Synergies between environmental degradation and climate variation on malaria reemergence in southern Venezuela: A spatiotemporal modelling study. Lancet Planet Health. 2022;6:e739-e48. https://doi.org/10.1016/S2542-5196(22)00192-9
Hamlet A, Gaythorpe KA, Garske T, Ferguson NM. Seasonal and inter-annual drivers of yellow fever transmission in South America. PLoS Negl Trop Dis. 2021;15:e0008974. https://doi.org/10.1371/journal.pntd.0008974
Hamrick PN, Aldighieri S, Machado G, Leonel DG, Vilca LM, Uriona S, et al. Geographic patterns and environmental factors associated with human yellow fever presence in the Americas. PLoS Negl Trop Dis. 2017;11:e0005897. https://doi.org/10.1371/journal.pntd.0005897
Aliaga-Samanez A, Romero D, Murray K, Cobos-Mayo M, Segura M, Real R, et al. Climate change is aggravating dengue and yellow fever transmission risk. Ecography. 2024;2024:e06942. https://doi.org/10.1111/ecog.06942
Hansen CA, Barrett AD. The present and future of yellow fever vaccines. Pharmaceuticals (Basel). 2021;14:891. https://doi.org/10.3390/ph14090891
Some similar items:
- Elizabeth Borrero, Gabriel Carrasquilla, Neal Alexander, Decentralization and health system reform: What is their impact on malaria incidence in Colombian municipalities? , Biomedica: Vol. 32 (2012): Suplemento 1, Malaria
- Raúl Murillo, Ricardo Cendales, Carolina Wiesner, Marion Piñeros, Sandra Tovar, Effectiveness of cytology-based cervical cancer screening in the Colombian health system , Biomedica: Vol. 29 No. 3 (2009)
- Sandra Lorena Girón, Julio César Mateus, Fabián Méndez, Impact of an open waste disposal site on the occurrence of respiratory symptoms and on health care costs of children , Biomedica: Vol. 29 No. 3 (2009)
- José Joaquín Carvajal, Ligia Inés Moncada, Mauricio Humberto Rodríguez, Ligia del Pilar Pérez, Víctor Alberto Olano, Characterization of Aedes albopictus (Skuse, 1894) (Diptera:Culicidae) larval habitats near the Amazon River in Colombia , Biomedica: Vol. 29 No. 3 (2009)
- Andrés Páez, Gloria Rey, Carlos Agudelo, Alvaro Dulce, Edgar Parra, Hernando Díaz-Granados, Damaris Heredia, Luis Polo, Outbreak of urban rabies transmitted by dogs in Santa Marta, northern Colombia , Biomedica: Vol. 29 No. 3 (2009)
- Patricia Escobar, Katherine Paola Luna, Indira Paola Hernández, César Mauricio Rueda, María Magdalena Zorro, Simon L. Croft, In vitro susceptibility of Trypanosoma cruzi strains from Santander, Colombia, to hexadecylphosphocholine (miltefosine), nifurtimox and benznidazole , Biomedica: Vol. 29 No. 3 (2009)
- Gustavo Pradilla, Julio César Mantilla, Reynaldo Badillo, Human rabies encephalitis by a vampire bat bite in an urban area of Colombia , Biomedica: Vol. 29 No. 2 (2009)
- Mauricio Beltrán, María Cristina Navas, María Patricia Arbeláez, Jorge Donado, Sergio Jaramillo, Fernando De la Hoz, Cecilia Estrada, Lucía del Pilar Cortés, Amalia de Maldonado, Gloria Rey, Seroprevalence of hepatitis B virus and human immunodeficiency virus infection in a population of multiply-transfused patients in Colombia , Biomedica: Vol. 29 No. 2 (2009)
- Rosa Magdalena Uscátegui, Adriana M. Correa, Jaime Carmona-Fonseca, Changes in retinol, hemoglobin and ferritin concentrations in Colombian children with malaria , Biomedica: Vol. 29 No. 2 (2009)
- Claudia M.E. Romero-Vivas, Luis Eduardo Castro, Lila Visbal, Ana María Santos, Esther Díaz, Cutaneous myiasis by Cochliomyia hominivorax (Coquerel) (Díptera Calliphoridae) in Hospital Universidad del Norte, Soledad, Atlántico , Biomedica: Vol. 29 No. 1 (2009)
Copyright (c) 2025 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























