Standardization of the use of opsonized zymosan as stimulus in the 1,2,3-dihydrorhodamine technique for the assessment of neutrophil respiratory burst
Abstract
Introduction. Chronic granulomatous disease is a defect in phagocytosis due to deficiency of gp91phox, p22phox, p47phox, p40phox, and p67phox (classic form of the disease). Recently, EROS and p40phox deficiency were described as responsible for the non-classical form of the disease. The 1,2,3-dihydrorhodamine oxidation technique, with phorbol-12-myristate-13-acetate as a stimulus, is performed to diagnose the classic chronic granulomatous disease. However, oxidation mediated by EROS and p40phox requires stimuli such as zymosan, Escherichia coli, or Staphylococcus aureus.
Objective. To optimize the 1,2,3-dihydrorhodamine technique using zymosan to assess neutrophil respiratory burst and detect the non-classical chronic granulomatous disease.
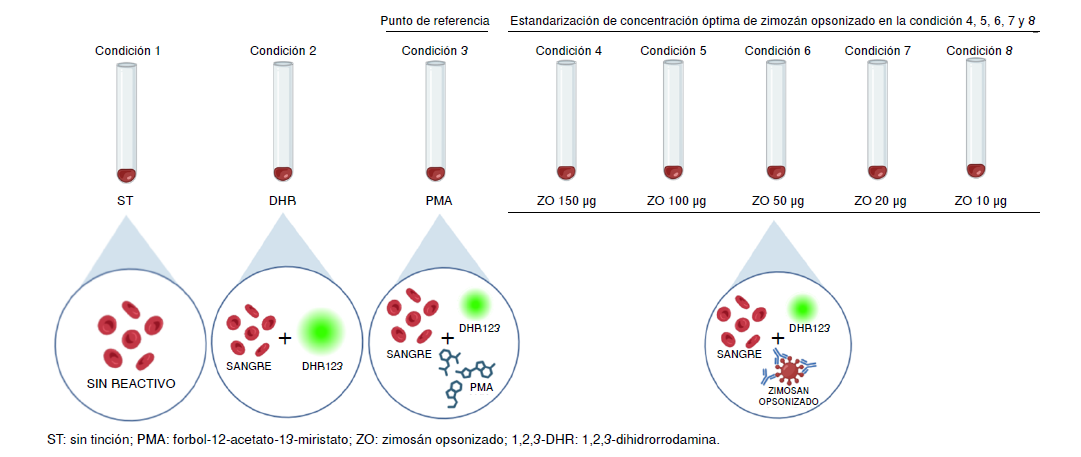
Materials and methods. Blood was obtained from five healthy subjects after the signature of the informed consent. The 1,2,3-dihydrorhodamine technique was performed with phorbol-12-myristate-13-acetate as control and different quantities of opsonized zymosan (150, 100, 50, 20, and 10 μg). We obtained through flow cytometry the mean fluorescence intensity of rhodamine 1,2,3 oxidated in the neutrophil population and calculated the oxidation index. The Kolmogorov-Smirnov test, ANOVA, and Tukey’s post-hoc analysis were used. We considered a p value ≤ 0.05 as statistically significant.
Results. The phorbol-12-myristate-13-acetate increased the rhodamine 1,2,3 mean fluorescence intensity in healthy subjects. Among the different zymosan conditions tested, we selected 50 μg as the optimal and reproducible amount in all controls according to the statistical analysis and cytometric findings.
Conclusions. We present the optimization of the 1,2,3-dihydrorhodamine technique using zymosan. We propose its implementation in clinical diagnostic laboratories to expand the diagnosis of chronic granulomatous disease.
Downloads
References
Yu HH, Yang YH, Chiang BL. Chronic granulomatous disease: A comprehensive review. Clin Rev Allergy Immunol. 2021;61:101-13. https://doi.org/10.1007/s12016-020-08800-x
Randzavola LO, Mortimer PM, Garside E, Dufficy ER, Schejtman A, Roumelioti G, et al. EROS is a selective chaperone regulating the phagocyte NADPH oxidase and purinergic signalling. Elife. 2022;11. https://doi.org/10.7554/eLife.76387
López-Hernández I, Suárez-Gutiérrez M, Santos-Chávez EE, Espinosa S, Blancas-Galicia L. Chronic granulomatous disease. Update and review. Rev Alerg Mex. 2019;66:232-45. https://doi.org/10.29262/ram.v66i2.577
Dinauer MC. Inflammatory consequences of inherited disorders affecting neutrophil function. Blood. 2019;133:2130-9. https://doi.org/10.1182/blood-2018-11-844563
Matute JD, Arias AA, Wright NA, Wrobel I, Waterhouse CC, Li XJ, et al. A new genetic subgroup of chronic granulomatous disease with autosomal recessive mutations in p40phox and selective defects in neutrophil NADPH oxidase activity. Blood. 2009;114:3309-15. https://doi.org/10.1182/blood-2009-07-231498
van de Geer A, Nieto-Patlan A, Kuhns DB, Tool AT, Arias AA, Bouaziz M, et al. Inherited p40phox deficiency differs from classic chronic granulomatous disease. J Clin Invest. 2018;128:3957-75. https://doi.org/10.1172/JCI97116
Neehus AL, Fusaro M, consortium NCF, Levy R, Bustamante J. A new patient with p40phox deficiency and chronic immune thrombocytopenia. J Clin Immunol. 2023;43:1173-7. https://doi.org/10.1007/s10875-023-01498-4
Arnadottir GA, Norddahl GL, Gudmundsdottir S, Agustsdottir AB, Sigurdsson S, Jensson BO, et al. A homozygous loss-of-function mutation leading to CYBC1 deficiency causes chronic granulomatous disease. Nat Commun. 2018;9:4447. https://doi.org/10.1038/s41467-018-06964-x
Thomas DC, Charbonnier LM, Schejtman A, Aldhekri H, Coomber EL, Dufficy ER, et al. EROS/CYBC1 mutations: Decreased NADPH oxidase function and chronic granulomatous disease. J Allergy Clin Immunol. 2019;143:782-5.e1. https://doi.org/10.1016/j.jaci.2018.09.019
Wright M, Chandrakasan S, Okou DT, Yin H, Jurickova I, Denson LA, et al. Early onset granulomatous colitis associated with a mutation in NCF4 resolved with hematopoietic stem cell transplantation. J Pediatr. 2019;210:220-5. https://doi.org/10.1016/j.jpeds.2019.03.042
Chester AE, Kuhns D, Lau K, Holland S, Canfield S, Garcia C, et al. A rare cause of necrotizing pneumonia in an adult woman: p40phox (NCF4) deficient autosomal recessive Chronic Granulomatous Disease. Clin Immunol. 2023;250(Supl.). https://doi.org/10.1016/j.clim.2023.109525
Pérez-Heras I, Tsilifis C, Slatter MA, Brynjolfsson SF, Haraldsson A, Gennery AR. HSCT in two brothers with CGD arising from mutations in CYBC1 corrects the defect in neutrophil function. Clin Immunol. 2021;229:108799. https://doi.org/10.1016/j.clim.2021.108799
Chiriaco M, De Matteis A, Cifaldi C, Di Matteo G, Rivalta B, Passarelli C, et al. Characterization of AR-CGD female patient with a novel homozygous deletion in CYBC1 gene presenting with unusual clinical phenotype. Clin Immunol. 2023;251:109316. https://doi.org/10.1016/j.clim.2023.109316
Mortimer PM, Nichols E, Thomas J, Shanbhag R, Singh N, Coomber EL, et al. A novel mutation in EROS (CYBC1) causes chronic granulomatous disease. Clin Immunol. 2023;255:109761. https://doi.org/10.1016/j.clim.2023.109761
Roos D, de Boer M. Molecular diagnosis of chronic granulomatous disease. Clin Exp Immunol. 2014;175:139-49. https://doi.org/10.1111/cei.12202
Nauseef WM. How human neutrophils kill and degrade microbes: An integrated view. Immunol Rev. 2007;219:88-102. https://doi.org/10.1111/j.1600-065X.2007.00550.x
Askarian F, Wagner T, Johannessen M, Nizet V. Staphylococcus aureus modulation of innate immune responses through Toll-like (TLR), (NOD)-like (NLR) and C-type lectin (CLR) receptors. FEMS Microbiol Rev. 2018;42:656-71. https://doi.org/10.1093/femsre/fuy025
Makni-Maalej K, Chiandotto M, Hurtado-Nedelec M, Bedouhene S, Gougerot-Pocidalo MA, Dang PM, et al. Zymosan induces NADPH oxidase activation in human neutrophils by inducing the phosphorylation of p47phox and the activation of Rac2: involvement of protein tyrosine kinases, PI3Kinase, PKC, ERK1/2 and p38MAPkinase. Biochem Pharmacol. 2013;85:92-100. https://doi.org/10.1016/j.bcp.2012.10.010
Belambri SA, Rolas L, Raad H, Hurtado-Nedelec M, Dang PM, El-Benna J. NADPH oxidase activation in neutrophils: Role of the phosphorylation of its subunits. Eur J Clin Invest. 2018;48(Suppl.2):e12951. https://doi.org/10.1111/eci.12951
Schmidlin M, Alt M, Vogel G, Voegeli U, Brodmann P, Bagutti C. Contaminations of laboratory surfaces with Staphylococcus aureus are affected by the carrier status of laboratory staff. J Appl Microbiol. 2010;109:1284-93. https://doi.org/10.1111/j.1365-2672.2010.04749.x
Bereswill S, Fischer A, Dunay IR, Kuhl AA, Gobel UB, Liesenfeld O, et al. Pro-inflammatory potential of Escherichia coli strains K12 and Nissle 1917 in a murine model of acute ileitis. Eur J Microbiol Immunol (Bp). 2013;3:126-34. https://doi.org/10.1556/EuJMI.3.2013.2.6
Hirt W, Nebe T, Birr C. Phagotest and Bursttest (Phagoburst), test kits for study of phagocyte functions. Wien Klin Wochenschr. 1994;106:250-2.
Rodríguez-Santiago B, Armengol L. Tecnologías de secuenciación de nueva generación en diagnóstico genético pre- y postnatal. Diagn Prenat. 2012;23:56-66. https://doi.org/10.1016/j.diapre.2012.02.001
Some similar items:
- Martín Romero, Diana Chávez, Magalí De los Ríos, Nelson Alvis-Guzmán, Cost-effectiveness of nilotinib, dasatinib and imatinib as first-line treatment for chronic myeloid leukemia in Colombia, 2012 , Biomedica: Vol. 34 No. 1 (2014)
- Yermis Carolina Rocha, Juan Álvaro López, Julio Cesar Orrego, Yadira Coll, Amado Karduss, Sergio Rosenzweig, José Luis Franco, Successful immune reconstitution by means of hematopoietic stem cell transplantation in a Colombian patient with chronic granulomatous disease , Biomedica: Vol. 36 No. 2 (2016)
- Karime Osorio-Arango, Mauricio Beltrán-Durán, Yazmín Arias-Murillo, Franklyn Prieto, Adriana Robayo, Survival in renal transplant recipients in Colombia, 2008-2012 , Biomedica: Vol. 37 No. 2 (2017)
- Nhora Silva, Natalia Calvache, Elizabeth Arrieta, Juan Carlos Bravo, Camilo Salazar, Joaquín Rosales, Francisco Jaramillo, Juan Guillermo Restrepo, Disseminated histoplasmosis as a first clinical manifestation in a patient with small lymphocytic lymphoma: A case report , Biomedica: Vol. 38 No. 3 (2018)
Copyright (c) 2024 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























