Retrospective pharmacogenetic analysis of a pediatric patient under anticoagulant treatment: Clinical case
Abstract
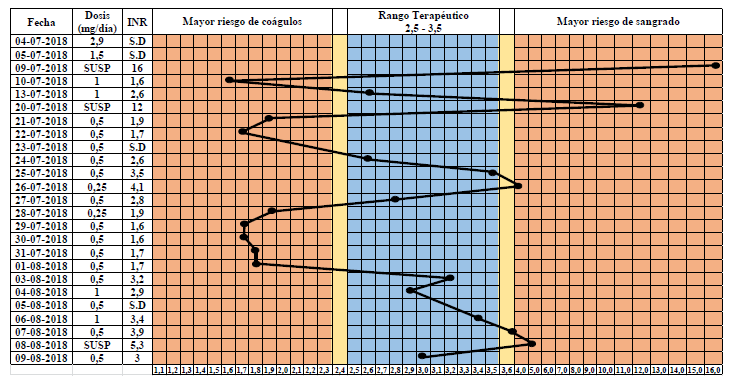
We present the clinical case of a 10-year-old patient diagnosed with dilated cardiomyopathy who registered INR values above 10 upon receiving standard doses of acenocoumarol, as well as other values reported as uncoagulable, forcing the discontinuation and restart of treatment more than once. Expected and stable INR levels were achieved after more than 30 days of treatment, surprisingly with half the recommended dose for a patient of her age and weight.
We decided to conduct a retrospective pharmacogenomic analysis including nucleotide genetic polymorphisms (SNPs) with different degrees of association with the dose/response to antivitamin K (AVK) drugs: rs2108622 (gene CYP4F2), rs9923231, rs7294 (gene VKORC1), rs1799853, and rs1057910 (CYP2C9 gene) using TaqMan® RT-PCR. The patient was homozygous for rs9923231 (VKORC1) and heterozygous for rs2108622 (CYP4F2),a genetic profile strongly associated with a requirement of lower AVK doses as shown by national and international evidence.
In conclusion, the pharmacogenetic analysis confirmed that this patient’s genetic conditions, involving low expression of the VKA therapeutic target, required a lower dose than that established in clinical protocols as recommended by the Food and Drug Administration (FDA) and the PharmGKB® for coumarin drugs. A previous genotypic analysis of the patient would have allowed reaching the therapeutic range sooner, thus avoiding potential bleeding risks. This shows the importance of pharmacogenetic analyses for highly variable treatments with a narrow therapeutic range.
Downloads
References
Organización Mundial de la Salud. Enfermedades cardiovasculares. Fecha de consulta: 5 de julio del 2020. Disponible en: http://www.who.int/cardiovascular_diseases/es/
Vukasovic JL. Miocardiopatía dilatada: aspectos genéticos, infecciosos, inflamatorios y del sistema inmune. Revista Médica Clínica Las Condes. 2015;26:210-6. https://doi.org/10.1016/j.rmclc.2015.04.009
Landefeld CS, Beyth RJ. Anticoagulant-related bleeding: Clinical epidemiology, prediction, and prevention. Am J Med. 1993;95:315-28. https://doi.org/10.1016/0002-9343(93)90285-w
White HD, Gruber M, Feyzi J, Kaatz S, Tse HF, Husted S, et al. Comparison of outcomes among patients randomized to warfarin therapy according to anticoagulant control: Results SPORTIF III and V. Arch Intern Med. 2007;167:239-45. https://doi.org/10.1001/archinte.167.3.239
Florez J, Sedano MC. Farmacología humana: farmacología de la hemostasia, la coagulación y la fibrinólisis. Sexta edición. Barcelona: Editorial Elseiver Masson; 2014. p. 736-8.
U.S. Food and Drug Administration. Table of pharmacogenomic biomarkers in drug abeling. Fecha de consulta: 5 de julio del 2020. Disponible en: https://www.fda.gov/Drugs/ScienceResearch/ucm572698.htm
Whirl-Carrillo M, McDonagh EM, Hebert JM, Gong L, Sangkuhl K, Thorn CF, et al. Pharmacogenomics knowledge for personalized medicine. Clin Pharmacol Ther. 2012;92:414-7. https://doi.org/10.1038/clpt.2012.96
Owen RP, Gong L, Hersh S, Klein TE, Altman RB. VKORC1 pharmacogenomics summary. Pharmacogenet Genomics. 2011;20:642-4. https://doi.org/10.1097/FPC.0b013e32833433b6
Johnson JA, Caudle KE, Gong L, Stein CM, Scott SA, Lee MT, et al. Clinical Pharmacogenetics Implementation Consortium (CPIC) Guideline for pharmacogenetics guided warfarin dosing : 2017 update. Clin Pharmacol Ther. 2017;102:397-404. https://doi.org/10.1002/cpt.668
Roco A, Nieto E, Suárez M, Rojo M, Bertoglia MP, Verón G, et al. A pharmacogenetically guided acenocoumarol dosing algorithm for Chilean patients: A discovery cohort study. Front Pharmacol. 2020;11:1-9. https://doi.org/10.3389/fphar.2020.00325
Bonduel M, Sciuccati G, Hepner M, Feliu-Torres A, Pieroni G, Frontroth JP, et al. Acenocoumarol therapy in pediatric patients. J Thromb Haemost. 2003;1:1740-3. https://doi.org/10.1046/j.1538-7836.2003.00256.x
Holbrook A, Schulman S, Witt DM, Vandvik PO, Fish J, Kovacs MJ, et al. Evidence-based management of anticoagulant therapy: Antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(Supl.2):e152S-e184S. https://doi.org/10.1378/chest.11-2295
Guyatt GH, Akl EA, Crowther M, Gutterman DD, Schünemann HJ. Executive summary: Antithrombotic therapy and prevention of thrombosis. 9th edition. American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(Supl.2):7-47. https://doi.org/10.1378/chest.1412S3
Potamias G, Lakiotaki K, Katsila T, Lee MTM, Topouzis S, Cooper DN, et al. Deciphering next-generation pharmacogenomics: An information technology perspective. Open Biol. 2014;4:421-32. https://doi.org/10.1098/rsob.140071
Some similar items:
- Carlos Isaza, Leonardo Beltrán, Julieta Henao, Gloria Porras, Alfredo Pinzón, Álvaro Vallejos, Jorge Machado, Genetic and bioenvironmental factors associated with warfarin response in Colombian patients , Biomedica: Vol. 30 No. 3 (2010)
- Iván Moreno, Luis Quiñones, Johanna Catalán, Carla Miranda, Angela Roco, Jaime Sasso, Evelyn Tamayo, Dante Cáceres, Andrei N. Tchernitchin, Leonardo Gaete, Iván Saavedra, Influence of CYP3A4/5 polymorphisms in the pharmacokinetics of levonorgestrel: a pilot study , Biomedica: Vol. 32 No. 4 (2012)
- Rubén Gómez, José Luis Penas, Cristina Fleitas, José Emiliano Cascallana, A fatal free floating thrombus in left atrium. , Biomedica: Vol. 25 No. 3 (2005)
- Ricardo A. Cifuentes, Juan Murillo-Rojas, Esperanza Avella-Vargas, Prediction of sensitivity to warfarin based on VKORC1 and CYP2C9 polymorphisms in patients from different places in Colombia , Biomedica: Vol. 36 No. 1 (2016)
- Ángel Tito Alvarado, Ana María Muñoz, Berta Loja, Jessica Michiko Miyasato, Jorge Antonio García, Roberto Andrés Cerro, Luis Abel Quiñones, Nelson Miguel Varela, Study of the allelic variants CYP2C9*2 and CYP2C9*3 in samples of the Peruvian mestizo population , Biomedica: Vol. 39 No. 3 (2019)
- Natalia Osorio, Martha Mónica Reyes, Thromboembolic complications associated with tuberculosis: A pediatric case report , Biomedica: Vol. 40 No. 4 (2020)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























