The perception of different social agents in Colombia regarding biobanks for research purposes
Abstract
Introduction: Biobanks for research purposes are public assets that require active participation from all interested parties; therefore, it is important to discern and investigate the perception that the general public in Colombia has with regards to their participation in a biobank.
Objective: To question different healthy Colombian social agents about the perception they have on donating human biological material to be stored in a biobank for future research purposes.
Materials and methods: We conducted an exploratory quantitative and qualitative research by means of an anonymous survey on healthy members of the Colombian community in order to evaluate their knowledge and stances related to biobanks.
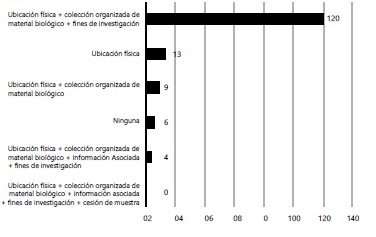
Results: Three hundred sixty-eight (368) individuals completed the survey, including health-care researchers, lawyers and members of research ethics committees. Ninety-six percent (96%) of the subjects stated their willingness to donate biological material to biobanks, although 60% expressed distress about their samples being used for certain research activities, mainly human cloning. Even though about half of the participants reported knowing what a biobank is, less than 3% of the individuals had a clear concept of them.
Conclusion: Participants showed their willingness to donate and store their biological material for research purposes. Similarly, they expressed their opinion about issues related to the management of biobanks. It is necessary to take into account the opinions of the general public in the development of policies that regulate biobanks for research purposes in Colombia.
Downloads
References
Kauffmann F, Cambon-Thomsen A. Tracing biological collections: Between books and clinical trials. JAMA. 2008;299:2316-8. https://doi.org/10.1001/jama.299.19.2316
Hewitt RE. Biobanking: The foundation of personalized me dicine. Curr Opin Oncol. 2011;23:112-9. https://doi.org/10.1097/CCO.0b013e32834161b8
Khoury MJ, Millikan R, Little J, Gwinn M. The emergence of epidemiology in the genomics age. Int J Epidemiol. 2004;33:936-44. https://doi.org/10.1093/ije/dyh278
Barr M. I’m not really read up on genetics: Biobanks and the social context of informed consent. BioSocieties. 2006;1:251-62. https://doi.org/10.1017/S1745855206060029
Jack AL, Womack C. Why surgical patients do not donate tissue for commercial research: Review of records. BMJ. 2003;327:262. https://doi.org/10.1136/bmj.327.7409.262
Cambon-Thomsen A, Rial-Sebbag E, Knoppers BM. Trends in ethical and legal frameworks for the use of human biobanks. Eur Respir J. 2007;30:373-82. https://doi.
org/10.1183/09031936.00165006
Hawkins AK, O’Doherty KC. “Who owns your poop?” Insights regarding the intersection of human microbiome research and the ELSI aspects of biobanking and related
studies. BMC Med Genomics. 2011;4:72. https://doi.org/10.1186/1755-8794-4-72
Serrano-Díaz N, Guío-Mahecha E, Páez-Leal MC. Consentimiento informado para biobancos: un debate ético abierto. Revista Salud UIS. 2016;48:246-56. https://doi.
org/10.18273/revsal.v48n2-2016010
Johnsson L, Helgesson G, Rafnar T, Halldorsdottir I, Chia KS, Eriksson S, et al. Hypothetical and factual willingness to participate in biobank research. Eur J Hum Genet. 2010;18:1261-4. https://doi.org/10.1038/ejhg.2010.106
D’Abramo F, Schildmann J, Vollmann J. Research participant’ perceptions and views on consent for biobank research: A review of empirical data and ethical analysis. BMC Med Ethics. 2015;16:60. https://doi.org/10.1186/s12910-015-0053-5
Garrison NA, Sathe NA, Antommaria AH, Holm IA, Sanderson SC, Smith ME, et al. A systematic literature review of individuals’ perspectives on broad consent and
data sharing in the United States. Genet Med. 2015;18:663-71. https://doi.org/10.1038/gim.2015.138
Helgesson G. In defense of broad consent. Camb Q Healthc Ethics. 2012;21:40-50. https://doi.org/10.1017/S096318011100048X
Hofmann B. Broadening consent-and diluting ethics? J Med Ethics. 2009;35:125-9. http://doi.org/10.1136/jme.2008.024851
Murphy J, Scott J, Kaufman D, Geller G, LeRoy L, Hudson K. Public expectations for return of results from large-cohort genetic research. Am J Bioeth. 2008;8:36-43.
https://doi.org//10.1080/15265160802513093
Miller FA, Hayeems RZ, Li L, Bytautas JP. What does ‘respect for persons’ require? Attitudes and reported practices of genetics researchers in informing research
participants about research. J Med Ethics. 2012;38:48-52. https://doi.org//10.1136/jme.2010.041350
Haga SB, Beskow LM. Ethical, legal, and social implications of biobanks for genetics research. Adv Genet. 2008;60:505- 44. https://doi.org/10.1016/S0065-2660(07)00418-X
Porteri C, Giardina E, Eusebi L. Clinical trial sponsors’ refusal to communicate genetic research results to subjects. Patient Educ Couns. 2014;95:157-8. https://doi.org/10.1016/j.pec.2013.12.001
Some similar items:
- María Consuelo Miranda, Gloria I. Palma, Ernesto Jaramillo, Ethics review committees for human research: the challenge of strengthening this process in Colombia. , Biomedica: Vol. 26 No. 1 (2006)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























