Evaluation of oxidative and methylating DNA damage in painters occupationally exposed to organic solvents and paints
Abstract
Introduction: The exposure to organic solvents and paints has been associated with genotoxicity and a greater risk of neoplasms. However, the type of DNA damage induced in humans by the exposure to these compounds, which would help explain the mechanisms of their genotoxicity, is still not fully characterized. Due to inadequate practices of occupational safety, car painters in the informal sector are a highly exposed group to organic solvents and paints.
Objective: To identify the oxidative and methylating damage in the DNA of lymphocytes of car painters exposed to organic solvents and paints.
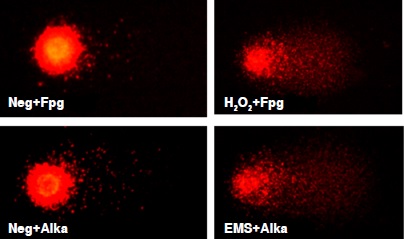
Materials and methods: Isolated peripheral blood lymphocytes from 62 painters and 62 unexposed subjects were analyzed by the modified high-throughput comet assay with the Fpg and AlkA enzymes. The categories used for the evaluation of the DNA damage were basal damage (without enzymes), oxidative and methylating damage. The measurement parameter used to establish the damage was the percentage of DNA in the tail.
Results: The percentage of DNA in the tail was higher in the exposed group compared to the unexposed group (p<0.05). In the exposed group, this percentage was higher in the oxidative damage category than the baseline (16.50 vs. 12.87; p<0.001), whereas methylating damage did not show significant differences (14.00 vs. 12.87; p>0.05).
Conclusion: In this study, exposure to organic solvents and paints was associated with an increase in oxidative lesions in the DNA of car painters’ lymphocytes, such as the production of 8-oxodG and other formamidopyrimidine products which are considered highly mutagenic.
Downloads
References
International Agency for Research on Cancer. Painting, firefighting, and shiftwork: Occupational exposure as a painter. IARC Monogr Eval Carcinog Risks Hum. Lyon, France: International Agency for Research on Cancer; 2010. p. 42-394.
Moro AM, Brucker N, Charao M, Bulcao R, Freitas F, Baierle M, et al. Evaluation of genotoxicity and oxidative damage in painters exposed to low levels of toluene. Mutat Res Genet Toxicol Environ Mutagen. 2012;746:42-8. https://doi.org/10.1016/j.mrgentox.2012.02.007
Cassini C, Calloni C, Bortolini G, García S, Dornelles M, Henriques J, et al. Occupational risk assessment of oxidative stress and genotoxicity in workers exposed to paints during a working week. Int J Occup Environ Health. 2011;24:308-19. https://doi.org/10.2478/s13382-011-0030-2
Londoño-Velasco E, Hidalgo-Cerón V, Escobar-Hoyos LF, Hoyos-Giraldo LS. Assessment of genomic damage and repair on human lymphocytes by paint thinner in vitro. Toxicol Mech Methods. 2014;24:243-9. PubMed PMID: 24236478. https://doi.org/10.3109/15376516.2013.862893
Costa C, De Pasquale R, Silvari V, Barbaro M, Catania S. In vitro evaluation of oxidative damage from organic solvent vapours on human skin. Toxicol In Vitro. 2006;20:324-31. https://doi.org/10.1016/j.tiv.2005.08.007
Martínez-Alfaro M, Palma-Tirado L, Sandoval-Zapata F, Carabez-Trejo A. Correlation between formamidopyrimidine DNA glycosylase (Fpg)-sensitive sites determined by a comet assay, increased MDA, and decreased glutathione during long exposure to thinner inhalation. Toxicol Lett. 2006;163:198-205. https://doi.org/10.1016/j.toxlet.2005.10.021
Hoyos-Giraldo LS, Carvajal S, Cajas-Salazar N, Ruiz M, Sánchez-Gómez A. Chromosome aberrations in workers exposed to organic solvents: Influence of polymorphisms in xenobiotic-metabolism and DNA repair genes. Mutat Res. 2009;666:8-15. https://doi.org/10.1016/j.mrfmmm.2009.03.003
Testa A, Festa F, Ranaldi R, Giachelia M, Tirindelli D, De Marco A, et al. A multi-biomarker analysis of DNA damage in automobile painters. Environ Mol Mutagen. 2005;46:182-8. https://doi.org/10.1002/em.20147
Aksoy H, Yilmaz S, Celik M, Yuzbasioglu D, Unal F. Genotoxicity study in lymphocytes of offset printing workers. J Appl Toxicol. 2006;26:10-5. https://doi.org/10.1002/jat.1098
Villalba-Campos M, Ramírez-Clavijo SR, Sánchez-Corredor MC, Rondón-Lagos M, Ibáñez-Pinilla M, Palma RM, et al. Quantification of cell-free DNA for evaluating genotoxic damage from occupational exposure to car paints. J Occup Med Toxicol. 2016;11:33. https://doi.org/10.1186%2Fs12995-016-0123-8
Londoño-Velasco E, Martínez-Perafán F, Carvajal-Varona S, García-Vallejo F, Hoyos- Giraldo LS. Assessment of DNA damage in car spray painters exposed to organic solvents by the high-throughput comet assay. Toxicol Mech Methods. 2016;26:238-42. https://doi.org/10.3109/15376516.2016.1158892
Collins A, Duthie S, Dobson V. Direct enzymic detection of endogenous oxidative base damage in human lymphocyte DNA. Carcinogenesis. 1993;14:1733.
Tice RR, Agurell E, Anderson D, Burlinson B, Hartmann A, Kobayashi H, et al. Single cell gel/comet assay: Guidelines for in vitro and in vivo genetic toxicology testing. Environ Mol Mutagen. 2000;35:206-21.
Collins AR, Dusinska M, Horska A. Detection of alkylation damage in human lymphocyte DNA with the comet assay. Acta Biochim Pol. 2001;48:611-4.
Blasiak J, Arabski M, Krupa R, Wozniak K, Rykala J, Kolacinska A, et al. Basal, oxidative and alkylative DNA damage, DNA repair efficacy and mutagen sensitivity in breast cancer. Mutat Res. 2004;554:139-48. https://doi.org/10.1016/j.mrfmmm.2004.04.001
World Medical Association. World Medical Association Declaration of Helsinki. Ethical principles for medical research involving human subjects. Bull World Health Organ. 2001;79:373-4.
World Health Organization. Standards and operational guidance for ethics review of health related research with human participants. Geneva: World Health Organization; 2011. p. 55.
Ministerio de Salud. Resolución Número 8430 de 1993 (4 de octubre). Normas científicas, técnicas y administrativas para la investigación en salud. Santafé de Bogotá: Ministerio de Salud; 1993. p. 19.
Singh NP, McCoy MT, Tice RR, Schneider EL. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res. 1988;175:184-91.
Azqueta A, Gutzkow KB, Priestley CC, Meier S, Walker JS, Brunborg G, et al. A comparative performance test of standard, medium- and high-throughput comet assays. Toxicol In Vitro. 2013;27:768-73. https://doi.org/10.1016/j.tiv.2012.12.006
Gutzkow KB, Langleite TM, Meier S, Graupner A, Collins AR, Brunborg G. High-throughput comet assay using 96 minigels. Mutagenesis. 2013;28:333-40. https://doi.org/10.1093/mutage/get012
Lynge E, Anttila A, Hemminki K. Organic solvents and cancer. Cancer Causes Control. 1997;8:406-19.
Ministerio de Salud y Protección Social, Instituto Nacional de Cancerología. Plan Nacional para el control del Cáncer en Colombia, 2012. Fecha de consulta: 14 de septiembre de 2018. Disponible en: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/IA/INCA/plan-nacional-control-cancer.pdf
Wogan GN, Hecht SS, Felton JS, Conney AH, Loeb LA, editors. Environmental and chemical carcinogenesis, 2004: Elsevier. p. 473-486.
Clapp RW, Howe GK, Jacobs MM. Environmental and occupational causes of cancer: A call to act on what we know. Biomed Pharmacother. 2007;61:631-9. https://doi.org/10.1016/j.biopha.2007.08.001
International Agency for Research on Cancer. Chemical agents and related occupations: Occupational exposure as a painter. IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Humans. Lyon: International Agency for Research on Cancer IARC. Working Group on the Evaluation of Carcinogenic Risk to Humans; 2012. p. 509 - 39.
Brown LM, Moradi T, Gridley G, Plato N, Dosemeci M, Fraumeni JF. Exposures in the painting trades and paint manufacturing industry and risk of cancer among men and women in Sweden. J Occup Environ Med. 2002;44:258-64.
Ramanakumar AV, Parent ME, Richardson L, Siemiatycki J. Exposures in painting-related occupations and risk of lung cancer among men: Results from two case-control studies in Montreal. Occup Environ Med. 2011;68:44-51. https://doi.org/10.1136/oem.2009.049957
Alguacil J, Porta M, Malats N, Kauppinen T, Kogevinas M, Benavides FG, et al. Occupational exposure to organic solvents and K-ras mutations in exocrine pancreatic cancer. Carcinogenesis. 2002;23:101-6.
Murata M, Tsujikawa M, Kawanishi S. Oxidative DNA damage by minor metabolites of toluene may lead to carcinogenesis and reproductive dysfunction. Biochem Biophys Res Commun. 1999;261:478-83. https://doi.org/10.1006/bbrc.1999.1041
Midorikawa K, Uchida T, Okamoto Y, Toda C, Sakai Y, Ueda K, et al. Metabolic activation of carcinogenic ethylbenzene leads to oxidative DNA damage. Chem Biol Interact. 2004;150:271-81. https://doi.org/10.1016/j.cbi.2004.09.020
Gaikwad NW, Bodell WJ. Formation of DNA adducts in HL-60 cells treated with the toluene metabolite p-cresol: A potential biomarker for toluene exposure. Chem-Biol Interact. 2003;145:149-58.
Dizdaroglu M. Oxidatively induced DNA damage: Mechanisms, repair, and disease. Cancer Lett. 2012;327:26-47. https://doi.org/10.1016/j.canlet.2012.01.016
Bruner SD, Norman D, Verdine GL. Structural basis for recognition and repair of the endogenous mutagen 8-oxoguanine in DNA. Nature. 2000;403:859. https://doi.org/10.1038/35002510
Kino K, Sugiyama H. Possible cause of G --> C transversion mutation by guanine oxidation product, imidazolone. Chem Biol. 2001;8:369-78.
Arabski M, Klupinska G, Chojnacki J, Kazmierczak P, Wisniewska-Jarosinska M, Drzewoski J, et al. DNA damage and repair in Helicobacter pylori-infected gastric mucosa cells. Mutat Res. 2005;570:129-35. https://doi.org/10.1016/j.mrfmmm.2004.10.006
Hoyos-Giraldo LS, Escobar-Hoyos LF, Saavedra-Trujillo D, Reyes-Carvajal I, Muñoz A, Londoño-Velasco E, et al. Gene-specific promoter methylation is associated with micronuclei frequency in urothelial cells from individuals exposed to organic solvents and paints. J Expo Sci Environ Epidemiol. 2016;26:257-62. https://doi.org/10.1038/jes.2015.28
Moller P. Assessment of reference values for DNA damage detected by the comet assay in human blood cell DNA. Mutat Res. 2006;612:84-104. https://doi.org/10.1016/j.mrrev.2005.10.001
Mladinic M, Kopjar N, Milic M, Dasovic AB, Huzak M, Zeljezic D. Genomic instability in a healthy elderly population: A pilot study of possible cytogenetic markers related to ageing. Mutagenesis. 2010;25:455-62. https://doi.org/10.1093/mutage/geq027
Fracasso ME, Doria D, Carrieri M, Bartolucci GB, Quintavalle S, De Rosa E. DNA single and double-strand breaks by alkaline- and immuno-comet assay in lymphocytes of workers exposed to styrene. Toxicol Lett. 2009;185:9-15. https://doi.org/10.1016/j.toxlet.2008.11.010
Sul D, Lee D, Im H, Oh E, Kim J, Lee E. Single strand DNA breaks in T- and B-lymphocytes and granulocytes in workers exposed to benzene. Toxicol Lett. 2002;134:87-95.
Roma-Torres J, Teixeira JP, Silva S, Laffon B, Cunha LM, Méndez J, et al. Evaluation of genotoxicity in a group of workers from a petroleum refinery aromatics plant. Mutat Res. 2006;604:19-27. https://doi.org/10.1016/j.mrgentox.2005.12.005
Sardas S, Omurtag GZ, Tozan A, Gul H, Beyoglu D. Evaluation of DNA damage in construction-site workers occupationally exposed to welding fumes and solvent-based paints in Turkey. Toxicol Ind Health. 2010;26:601-8. https://doi.org/10.1177/0748233710374463
Zhu CQ, Lam TH, Jiang CQ. Lymphocyte DNA damage in bus manufacturing workers. Mutat Res Genet Toxicol Environ Mutagen. 2001;491:173-81.
Bonassi S, Znaor A, Ceppi M, Lando C, Chang WP, Holland N, et al. An increased micronucleus frequency in peripheral blood lymphocytes predicts the risk of cancer in humans. Carcinogenesis. 2006;28:625. https://doi.org/10.1093/carcin/bgl177
Bonassi S, Hagmar L, Stromberg U, Montagud AH, Tinnerberg H, Forni A, et al. Chromosomal aberrations in lymphocytes predict human cancer independently of exposure to carcinogens. European Study Group on Cytogenetic Biomarkers and Health. Cancer Res. 2000;60:1619-25.
Heuser VD, Erdtmann B, Kvitko K, Rohr P, da Silva J. Evaluation of genetic damage in Brazilian footwear-workers: Biomarkers of exposure, effect, and susceptibility. Toxicology. 2007;232:235-47. https://doi.org/10.1016/j.tox.2007.01.011
Martino-Roth M, Viegas J, Roth D. Occupational genotoxicity risk evaluation through the comet assay and the micronucleus test. Genet Mol Res. 2003;2:410-7.
Pereira da Silva VH, Gomes de Moura CF, Spadari-Bratfisch RC, Ribeiro DA. Cytogenetic biomonitoring of peripheral blood and oral mucosa cells from car painters. Toxicol Mech Methods. 2012;22:497-501. https://doi.org/10.3109/15376516.2012.680621
Some similar items:
- Wellman Ribón, Claudia Castro, Liliana González, Juan Carlos Rozo, Gloria Puerto, Biosafety evaluation of the DNA extraction protocol for Mycobacterium tuberculosis complex species, as implemented at the Instituto Nacional de Salud, Colombia , Biomedica: Vol. 29 No. 4 (2009)
- Brian Alejandro Suárez, Claudia Liliana Cuervo, Concepción Judith Puerta, The intergenic region of the histone h2a gene supports two major lineages of Trypanosoma rangeli , Biomedica: Vol. 27 No. 3 (2007)
- Rafael José Vivero, Maria Angélica Contreras-Gutiérrez, Eduar Elías Bejarano, Analysis of the primary and secondary structure of the mitochondrial serine transfer RNA in seven species of Lutzomyia , Biomedica: Vol. 27 No. 3 (2007)
- Diana Carolina López, Carlos Jaramillo, Felipe Guhl, Population structure and genetic variability of Rhodnius prolixus (Hemiptera: Reduviidae) from different geographic areas of Colombia. , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Ingrid Yamile Pulido, José Ramón Mantilla, Emilia María Valenzuela, María Teresa Reguero, Elsa Beatriz González, Distribution of extended spectrum β-lactamases-codifying genes in Klebsiella pneumoniae isolates from hospitals of Bogota, D.C., Colombia , Biomedica: Vol. 31 No. 1 (2011)
- Sandy Milena Caldera, María Cristina Jaramillo, Suljey Cochero, Alveiro Pérez-Doria, Eduar Elías Bejarano, Genetic differences between populations of Aedes aegypti from municipalities in northern Colombia, with high and low dengue incidence , Biomedica: Vol. 33 (2013): Suplemento 1, Fiebres hemorrágicas
- Leonardo F. Jurado, Martha I. Murcia, Patricia Hidalgo, John E. Leguizamón, Lorena R. González, Phenotypic and genotypic diagnosis of bone and miliary tuberculosis in an HIV+ patient in Bogotá, Colombia , Biomedica: Vol. 35 No. 1 (2015)
- Andrea Casas-Vargas, Liza M. Romero, William Usaquén, Sara Zea, Margarita Silva, Ignacio Briceño, Alberto Gómez, José Vicente Rodríguez, Mitochondrial DNA diversity in prehispanic bone remains on the eastern Colombian Andes , Biomedica: Vol. 37 No. 4 (2017)
- Adrián Peñata, Richard Salazar, Tatiana Castaño, Julián Bustamante, Sigifredo Ospina, Molecular diagnosis of extrapulmonary tuberculosis and sensitivity to rifampicin with an automated real-time method , Biomedica: Vol. 36 (2016): Suplemento 1, Microbiología médica
- Luz Adriana Pérez, Freddy Rodríguez, Carl Henrik Langebaek, Helena Groot, Real-time quantification to analyze historical Colombian samples detecting a short fragment of hypervariable region II of mitochondrial DNA , Biomedica: Vol. 36 No. 3 (2016)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























