A pathogenic homozygous variant of the BBS10 gene in a patient with Bardet Biedl syndrome
Abstract
The Bardet-Biedl syndrome is an autosomal recessive hereditary disorder with vast locus heterogeneity that belongs to the so-called ciliopathies, whose proteins are localized in the primary cilia and present functional deficiency. The multisystemic features of the disease include ocular, renal, cognitive, skeletal, as well as gonadal involvement and obesity, among others, with high inter- and intrafamilial variability.
We describe the clinical case of an adolescent male patient with Bardet-Biedl syndrome, including the approach, the results from a 22-gene sequencing panel, and the analysis of updated scientific literature.
We collected the clinical data of the patient and, after obtaining the informed consent, we conducted a multigenic sequencing panel oriented to known implicated genes.
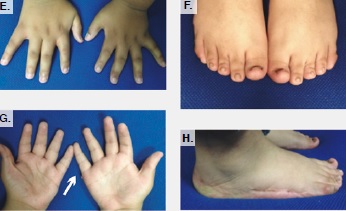
The patient was born to consanguineous parents and was the first affected member of the family. He presented with postaxial polydactyly, obesity, micropenis, retinitis pigmentosa, and learning disability.
The multigenic panel allowed the identification of the homozygous pathogenic variant c.39_46del in the BBS10 gene and in other BBS genes variants associated with obesity. As the Bardet-Biedl syndrome is a rare disease, it is challenging to interpret its pleiotropism and gene/allelic heterogeneity. Its confirmation by molecular tests allows an adequate approach, follow-up, and genetic counseling of the patient and the family.
Downloads
References
Laurence JZ, Moon RC. Four cases of “retinitis pigmentosa” occurring in the same family, and accompanied by general imperfections of development. Obes Res. 1995;3:400-3. https://doi.org/10.1002/j.1550-8528.1995.tb00166.x
Wilhelm V, Pacheco V. Síndrome de Laurence-Moon-Biedl. Rev Chil Pediatr. 1979;50:77-80. https://doi.org/10.4067/S0370-41061979000100013
Forsythe E, Beales PL. Bardet–Biedl syndrome. Eur J Hum Genet. 2013;21:8-13. https://doi.org/10.1038/ejhg.2012.115
Forsythe E, Beales PL. Bardet-Biedl syndrome. Gene Reviews® [Internet]. Fecha de consulta: 17 de octubre de 2017. Disponible en: http://www.ncbi.nlm.nih.gov/books/NBK1363/
Beales PL, Elcioglu N, Woolf AS, Parker D, Flinter FA. New criteria for improved diagnosis of Bardet-Biedl syndrome: results of a population survey. J Med Genet. 1999;36:437-46. https://doi.org/10.1136/jmg.36.6.437
Daniels AB, Sandberg MA, Chen J, Weigel-DiFranco C, Hejtmancik JF, Berson EL. Genotype-phenotype correlations in Bardet-Biedl syndrome. Arch Ophthalmol. 2012;130:901-7. https://doi.org/10.1001/archophthalmol.2012.89
Green JS, Parfrey PS, Harnett JD, Farid NR, Cramer BC, Johnson G, et al. The cardinal manifestations of Bardet-Biedl syndrome, a form of Laurence-Moon-Biedl syndrome. N Engl J Med. 1989;321:1002-9. https://doi.org/10.1056/NEJM198910123211503
Tobin JL, Beales PL. Bardet-Biedl syndrome: beyond the cilium. Pediatr Nephrol. 2007;22:926-36. https://doi.org/10.1007/s00467-007-0435-0
Pawlik B, Mir A, Iqbal H, Li Y, Nürnberg G, Becker C, et al. A novel familial BBS12 mutation associated with a mild phenotype: implications for clinical and molecular diagnostic strategies. Mol Syndromol. 2010;1:27-34. https://doi.org/10.1159/000276763
Hostelley TL, Lodh S, Zaghloul NA. Whole organism transcriptome analysis of zebrafish models of Bardet-Biedl syndrome and Alström syndrome provides mechanistic insight into shared and divergent phenotypes. BMC Genomics. 2016;17:318. https://doi.org/10.1186/s12864-016-2679-1
Heon E, Kim G, Qin S, Garrison JE, Tavares E, Vincent A, et al. Mutations in C8ORF37 cause Bardet Biedl syndrome (BBS21). Human Mol Genet. 2016;25:2283-94. https://doi.org/10.1093/hmg/ddw096
Lindstrand A, Frangakis S, Carvalho CM, Richardson EB, McFadden KA, Willer JR, et al. Copy-number variation contributes to the mutational load of Bardet-Biedl syndrome. Am J Hum Genet. 2016;99:318-36. https://doi.org/10.1016/j.ajhg.2015.04.023
Ministerio de Salud. Resolución 8430 de 1993. Por la cual se establecen las normas científicas, técnicas y administrativas para la investigación en salud. Fecha de consulta: 17 de octubre de 2017. Disponible en: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/DE/DIJ/RESOLUCION-8430-DE-1993.PDF
Ministerio de la Protección Social. Resolución 2378 de 2008. Por la que se adoptan las buenas prácticas clínicas para las instituciones que conducen investigación con medicamentos en seres humanos. Fecha de consulta:17 de octubre de 2017. Disponible en: http://www.alcaldiabogota.gov.co/sisjur/normas/Norma1.jsp?i=31169
World Medical Association. Declaración de Helsinki -Principios éticos para las investigaciones médicas en seres humanos. 64a Asamblea General, octubre de 2013. Fecha de consulta: 17 de octubre de 2017. Disponible en: https://www.wma.net/es/policies-post/declaracion-de-helsinkide-la-amm-principios-eticos-para-las-investigacionesmedicas-en-seres-humanos/
Wang K, Li M, Hakonarson H. ANNOVAR: Functional annotation of genetic variants from next-generation sequencing data. Nucleic Acids Research. 2010;38:e164.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and Guidelines for the Interpretation of Sequence Variants: A Joint Consensus Recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405-24. https://doi.org/10.1038/gim.2015.30
Schwarz JM, Cooper DN, Schuelke M, Seelow D. Mutation Taster2: mutation prediction for the deep-sequencing age. Nat Mehtods. 2014;11:361-2. https://doi.org/10.1038/nmeth.2890
McLaren W, Gil L, Hunt SE, Riat HS, Ritchie GR, Thormann A, et al. The Ensemble Variant Effect Predictor. Genome Biol. 2016;17:122. https://doi.org/10.1186/s13059-016-0974-4
Li Q, Wang K. InterVar: Clinical Interpretation of Genetic Variants vy the 2015 ACMG-AMP Guidelines. Am J Hum Genet. 2017;100:267-80. https://doi.org/10.1016/j.ajhg.2017.01.004
Slavotinek AM. McKusick-Kaufman Syndrome. Gene Reviews®. Fecha de consulta: 17 de octubre de 2017. Disponible en: http://www.ncbi.nlm.nih.gov/books/NBK1502/
Marshall JD, Maffei P, Beck S, Barrett TG, Paisey R, Naggert JK. Clinical utility gene card for: Alström Syndrome- update 2013. Eur J Hum Genet. 2013;21. https://doi.org/10.1038/ejhg.2013.61
Marshall JD, Paisey RB, Carey C, Macdermott S. Alström syndrome. GeneReviews®. Fecha de consulta: 17 de octubre de 2017. Disponible en: http://www.ncbi.nlm.nih.gov/books/NBK1267/24. Parisi M, Glass I. Joubert Syndrome and Related Disorders. GeneReviews®. Fecha de consulta: 17 de octubre de 2017. Disponible en: https://www.ncbi.nlm.nih.gov/books/NBK1325/
Farag TI, Teebi AS. High incidence of Bardet Biedl syndrome among the Bedouin. Clin Genet. 1989;36:463-4. https://doi.org/10.1111/j.1399-0004.1989.tb03378.x
Hjortshøj TD, Grønskov K, Brøndum-Nielsen K, Rosenberg T. A novel founder BBS1 mutation explains a unique high prevalence of Bardet-Biedl syndrome in the Faroe Islands. Br J Ophthalmol. 2009;93:409-13. https://doi.org/10.1136/bjo.2007.131110
Nachury MV, Loktev AV, Zhang Q, Westlake CJ, Peränen J, Merdes A, et al. A core complex of BBS proteins cooperates with the GTPase Rab8 to promote ciliary membrane biogenesis. Cell. 2007;129:1201-13. https://doi.org/10.1016/j.cell.2007.03.053
Loktev AV, Zhang Q, Beck JS, Searby CC, Scheetz TE, Bazan JF, et al. A BBSome subunit links ciliogenesis, microtubule stability, and acetylation. Dev Cell. 2008;15:854-65. https://doi.org/10.1016/j.devcel.2008.11.001
Seo S, Baye LM, Schulz NP, Beck JS, Zhang Q, Slusarski DC, et al. BBS6, BBS10, and BBS12 form a complex with CCT/TRiC family chaperonins and mediate BBSome assembly. Proc Natl Acad Sci USA. 2010;107:1488-93. https://doi.org/10.1073/pnas.0910268107
Cardenas-Rodriguez M, Badano JL. Ciliary biology: understanding the cellular and genetic basis of human ciliopathies. Am J Med Genet C Semin Med Genet. 2009;151C:263-80. https://doi.org/10.1002/ajmg.c.30227
Young JC, Agashe VR, Siegers K, Hartl FU. Pathways of chaperone-mediated protein folding in the cytosol. Nat Rev Mol Cel Biol. 2004;5:781-91. https://doi.org/10.1038/nrm1492
Álvarez-Satta M, Castro-Sánchez S, Valverde D. Bardet-Biedl syndrome as a chaperonopathy: dissecting the major role of chaperonin-like BBS proteins (BBS6-BBS10-BBS12). Front Mol Biosci. 2017;4:55. https://doi.org/10.3389/fmolb.2017.00055
Marion V, Stutzmann F, Gérard M, De Melo C, Schaefer E, Claussmann A, et al. Exome sequencing identifies mutations in LZTFL1, a BBSome and smoothened trafficking regulator, in a family with Bardet--Biedl syndrome with situs inversus and insertional polydactyly. J Med Genet. 2012;49:317-21. https://doi.org/10.1136/jmedgenet-2012-100737
Lindstrand A, Davis EE, Carvalho CM, Pehlivan D, Willer JR, Tsai IC, et al. Recurrent CNVs and SNVs at the NPHP1 locus contribute pathogenic alleles to Bardet-Biedl syndrome. Am J Hum Genet. 2014;94:745-54. https://doi.org/10.1016/j.ajhg.2014.03.017
Priya S, Nampoothiri S, Sen P, Sripriya S. Bardet-Biedl syndrome: Genetics, molecular pathophysiology, and disease management. Indian J Ophthalmol. 2016;64:620-7. https://doi.org/10.4103/0301-4738.194328
ET, Liu YP, Chan Y, Tiinamaija T, Käräjämäki A, Madsen E, et al. A novel test for recessive contributions to complex diseases implicates Bardet-Biedl syndrome gene BBS10 in idiopathic type 2 diabetes and obesity. Am J Hum Genet. 2014;95:509-20. https://doi.org/10.1016/j.ajhg.2014.09.015
Stoetzel C, Muller J, Laurier V, Davis EE, Zaghloul NA, Vicaire S, et al. Identification of a novel BBS gene (BBS12) highlights the major role of a vertebrate-specific branch of chaperonin-related proteins in Bardet-Biedl syndrome. Am J Hum Genet. 2007;80:1-11. https://doi.org/10.1086/510256
Zhang Q, Yu D, Seo S, Stone EM, Sheffield VC. Intrinsic protein-protein interaction-mediated and chaperoninassisted sequential assembly of stable Bardet-Biedl syndrome protein complex, the BBSome. J Biol Chem. 2012;287:20625-35. https://doi.org/10.1074/jbc.M112.341487
Cognard N, Scerbo MJ, Obringer C, Yu X, Cosata F, Haser E, et al. Comparing the Bbs10 complete knockout phenotype with a specific renal epithelial knockout one highlights the link between renal defects and systemic inactivation in mice. Cilia. 2015;4:10. https://doi.org/10.1186/s13630-015-0019-8
Stoezel C, Laurier V, Davis EE, Muller J, Rix S, Badano JL, et al. BBS10 encodes a vertebrate-specific chaperoninlike protein and is a major BBS locus. Nat Genet. 2006;38:521-4. Erratum in: Nat Genet. 2006;38(6):727. https://doi.org/10.1038/ng1771
Leroux MR, Hartl FU. Protein folding: versatility of the cytosolic chaperonin TRiC/CCT. Curr Biol. 2000;10:R260-4. https://doi.org/10.1016/S0960-9822(00)00432-2
Cuellar JP. Caracterización estructural y funcional de la interacción entre las chaperonas CCT y Hsc70. [disertación]. Madrid: Universidad Autónoma de Madrid; 2008. p. 7-8.
Leiden Open Variation Database. Gene BBS10. Fecha de consulta: 17 de marzo de 2018. Disponible en: https://lovd.euro-wabb.org/variants.php?select_db=BBS10&action=view&view=0000955%2C0000029%2C0
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, et al. A method and server for predicting damaging missense mutations. Nat Methods. 2010;7(4):248-9. https://doi.org/10.1038/nmeth0410-248
NHLBI GO Exome Sequencing Project (ESP). Exome Variant Server. Fecha de consulta: 17 de octubre de 2017. Disponible en http://evs.gs.washington.edu/EVS/
Rouskas K, Paletas K, Kalogeridis A, Sarigianni M, Ioannidou-Papagiannaki E, Tsapas A, et al. Association between BBS6/MKKS gene polymorphisms, obesity and metabolic syndrome in the Greek population. Int J Obes. 2008;32:1618-25. https://doi.org/10.1038/ijo.2008.167
Ajmal M, Imran M, Neveling K, Tayyab A, Jaffar S, Sadeque A, et al. Exome sequencing identifies a novel and a recurrent BBS1 mutation in Pakistani families with Bardet-Bield syndrome. Mol Vis. 2013;19:644-53.
Shim H, Kim JH, Kim CY, Hwang S, Kim H, Yang S, et al. Function-driven discovery of disease genes in zebrafish using an integrated genomics big data resource. Nucleic Acids Res. 2016;44:9611-23. https://doi.org/10.1093/nar/gkw897
Oud MM, Lamers IJ, Arts HH. Ciliopathies: genetics in pediatric medicine. J Pediatr Genet. 2017;6:18-29. https://doi.org/10.1055/s-0036-1593841
Sengillo JD, Justus S, Tsai YT, Cabral T, Tsang SH. Gene and cell-based therapies for inherited retinal disorders: An update. Am J Med Genet C Semin Med Genet. 2016;172:349-66. https://doi.org/10.1002/ajmg.c.31534
Sengillo JD, Justus S, Cabral T, Tsang SH. Correction of monogenic and common retinal disorders with gene therapy. Genes. 2017;8:53. https://doi.org/10.3390/genes8020053
Congreso de Colombia. Ley 1392 de 2010. Por medio de la cual se reconocen las enfermedades huérfanas como de especial interés y se adoptan normas tendientes a garantizar la protección social por parte del Estado colombiano a la población que padece enfermedades huérfanas y sus cuidadores. Fecha de consulta: 17 de octubre de 2017. Disponible en: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/DE/DIJ/ley-1392-de-2010.pdf
De Castro M, Restrepo CM. Genetics and genomic medicine in Colombia. Mol Genet Genomic Med. 2015;3:84-91. https://doi.org/10.1002/mgg3.139
Some similar items:
- Liliana Franco-Hincapié, Constanza Elena Duque, María Victoria Parra, Natalia Gallego, Alberto Villegas, Andrés Ruiz-Linares, Gabriel Bedoya, Association between polymorphism in uncoupling proteins and type 2 diabetes in a northwestern Colombian population , Biomedica: Vol. 29 No. 1 (2009)
- Elpidia Poveda, Diana Giraldo, Yibby Forero, Carlos Mendivil, Self-reported physical activity in comparison with anthropometric body fat indicators in school children , Biomedica: Vol. 28 No. 3 (2008)
- Juan Bernardo Pinzón, Norma Cecilia Serrano, Luis Alfonso Díaz, Gerardo Mantilla, Harvey Mauricio Velasco, Luz Ximena Martínez, Paula Andrea Millán, Sandra Milena Acevedo, Daniel Moreno, Impact of the new definitions in the prevalence of the metabolic syndrome in an adult population at Bucaramanga, Colombia , Biomedica: Vol. 27 No. 2 (2007)
- Nora Elena Múnera, Rosa Magdalena Uscátegui, Beatriz Elena Parra, Luz Mariela Manjarrés, Fredy Patiño, Claudia María Velásquez, Alejandro Estrada, Gabriel Bedoya, Vicky Parra, Angélica María Muñoz, Ana Carolina Orozco, Gloria María Agudelo, Environmental risk factors and metabolic syndrome components in overweight youngsters , Biomedica: Vol. 32 No. 1 (2012)
- Carolina Guerra, Carlos Santiago Uribe, Alejandro Guerra, Olga H. Hernández, Bickerstaff brain encephalitis: case report and review , Biomedica: Vol. 33 No. 4 (2013)
- Tomas Carvajal, Diana L. Franco, Adriana Martínez, Iván M. Peña, Wernicke’s encephalopathy and polyneuropathy associated with vitamin B complex deficiencyafter a bariatric surgery , Biomedica: Vol. 32 No. 4 (2012)
- Greta Rodríguez-Arroyo, Irene Paradisi, Merlyn Vívenes-Lugo, Dinorah Castro-Guerra, Álvaro Rodríguez-Larralde, LEP, LDLR and APOA4 gene polymorphisms and their relationship with the risk of overweight, obesity and chronic diseases in adults of the State of Sucre, Venezuela , Biomedica: Vol. 36 No. 1 (2016)
- Ana C. Orozco, Angélica M. Muñoz, Claudia M. Velásquez, Rosa M. Uscátegui, María V. Parra, Fredy A. Patiño, Luz M. Manjarres, Beatriz E. Parra, Alejandro Estrada, Gloria M. Agudelo, Variant in CAPN10 gene and environmental factors show evidence of association with excess weight among young people in a Colombian population , Biomedica: Vol. 34 No. 4 (2014)
- Harry Pachajoa, Vania A. Villota, Luz Marina Cruz, Yoseth Ariza, Prevalence of birth defects according to the level of care in two hospitals, Cali, Colombia, 2012-2013 , Biomedica: Vol. 35 No. 2 (2015)
- Oscar F. Herrán, Gonzalo A. Patiño, Sara E. Del Castillo, Dietary transition and excess weight in adults according to the Encuesta de la Situación Nutricional en Colombia, 2010 , Biomedica: Vol. 36 No. 1 (2016)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























