Potential models of the geographic and climatic distribution of the Amblyomma cajennense complex (Acari: Ixodidae), a potential vector of Rickettsia rickettsii in Colombia
Abstract
Introduction: The species of the Amblyoma cajennense complex, potential vectors involved in the transmission of Rickettsia rickettsii in Central and South America, have a wide geographical distribution in the Neotropics. In Colombia, the knowledge about these species, their distribution, and many aspects of their ecology is still limited.
Objective: To explore the ecology of A. cajennense complex in Colombia based on the study of its habitat selection, as well as the generation of potential geographic and climatic distribution models.
Materials and methods: We used unique records of these ticks to study the selection patterns of the climatic habitat using the Ecological Niche Factor Analysis (ENFA), and we built distribution models using the MaxEnt and GARP algorithms.
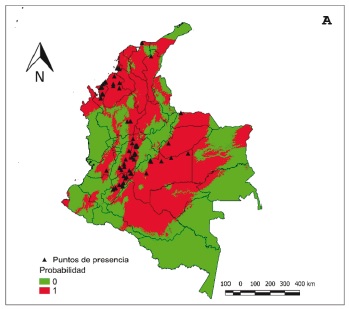
Results: The record of distributions showed that A. cajennense complex is present in all five natural regions of Colombia with 64.71% of the records found in the departments of Antioquia, Cundinamarca, and Huila. The analysis of their habitat selection showed that these ticks preferably select geographical areas (ENFA: 96.03%) according to the isothermality conditions and the precipitation of the driest quarter of the year. The potential distribution models indicated habitat suitability in 31 to 51% of the area of the country depending on the algorithm.
Conclusions: The distribution of A. cajennense complex in Colombia is highly influenced by a climatebased habitat selection, a characteristic that favored the creation of distribution models. Field studies will allow the validation of the potential distribution models and further genetic studies will be required in the country to identify species within the complex and study their potential hybridization areas.
Downloads
References
Beati L, Nava S, Burkman EJ, Barros-Battesti DM, Labruna MB, Guglielmone AA, et al. Amblyomma cajennense (Fabricius, 1787) (Acari: Ixodidae), the Cayenne tick: Phylogeography and evidence for allopatric speciation. BMC Evol Biol. 2013;13:267. https://doi.org/10.1186/1471-2148-13-267
Nava S, Beati L, Labruna MB, Cáceres AG, Mangold AJ, Guglielmone AA. Reassessment of the taxonomic status of Amblyomma cajennense (Fabricius, 1787) with the description of three new species, Amblyomma tonelliae n. sp., Amblyomma interandinum n. sp. and Amblyomma patinoi n. sp., and reinstatement of Amblyomma mixtum, and Amblyomma sculptum. Ticks Tick Borne Dis. 2014;5:252-276. https://doi.org/10.1016/j.ttbdis.2013.11.004
Estrada-Peña A, Guglielmone AA, Mangold AJ. The distribution and ecological ‘preferences’ of the tick Amblyomma cajennense (Acari: Ixodidae), an ectoparasite of humans and other mammals in the Americas. Ann Trop Med Parasitol. 2004;98:283-92. https://doi.org/10.1179/000349804225003316
Nava S, Guglielmone AA. A meta-analysis of host specificity in Neotropical hard ticks (Acari: Ixodidae). Bull Entomol Res. 2013;103:216-24. https://doi.org/10.1017/S00 07485312000557
Labruna MB, Mattar S, Nava S, Venzal JM, Dolz G, Abarca K, et al. Rickettsioses in Latin America, Caribbean, Spain and Portugal. Rev MVZ Córdoba. 2011;16:2435-57.
Parola P, Labruna MB, Raoult D. Tick-borne rickettsioses in America: Unanswered questions and emerging diseases. Curr Infect Dis Rep. 2009;11:40-50. https://doi.org/10.1007/s11908-009-0007-5
Scoles GA, Ueti MW. Amblyomma cajennense is an intrastadial biological vector of Theileria equi. Parasit Vectors. 2013;6:306. https://doi.org/10.1186/1756-3305-6-306
Sanders DM, Parker JE, Walker WW, Buchholz MW, Blount K, Kiel JL. Field collection and genetic classification of tick-borne Rickettsiae and Rickettsiae-like pathogens from South Texas: Coxiella burnetii isolated from field collected Amblyomma cajennense. Ann N Y Acad Sci. 2008;1149:208-11. https://doi.org/10.1196/annals.1428.022
Linthicum KJ, Logan TM, Bailey CL, Gordon SW, Peters CJ, Monath TP, et al. Venezuelan Equine Encephalomyelitis Virus infection in and transmission by the tick Amblyomma cajennense (Arachnida: Ixodidae). J Med Entomol. 1991;28:405-9. https://doi.org/10.1093/jmedent/28.3.405
Dunn H. Notes on some insects and other arthropods affecting man and animals in Colombia. Am J Trop Med Hyg. 1929;1:493-508. https://doi.org/10.4269/ajtmh.1929.s1-9.493
López G, Zúñiga I, Villar C, Osorio D. Distribución de garrapatas en 25 municipios de departamento de Antioquia. Rev ICA. 1985;20:40-4.
Patino L. Nuevas observaciones sobre un tercer foco de fiebre petequial (maculosa) en el hemisferio americano. Boletín Oficina Sanitaria Panamericana. 1941;20:1112-24.
Hidalgo M, Orejuela L, Fuya P, Carrillo P, Hernández J, Parra E, et al. Rocky mountain spotted fever, Colombia. Emerg Infect Dis. 2007;13:1058-60. https://doi.org/10.3201/eid1307.060537
Acosta J, Urquijo L, Díaz A, Sepúlveda M, Mantilla G, Heredia D, et al. Brote de Rickettsia Rickettsii en Necoclí, Antioquia, Colombia, 2006. Inf Quinc Epidemiol Nac. 2006;11:161-76.
Hidalgo M, Miranda J, Heredia D, Zambrano P, Vesga JF, Lizarazo D, et al. Outbreak of Rocky Mountain spotted fever in Córdoba, Colombia. Mem Inst Oswaldo Cruz.
;106:117-8. https://doi.org/10.1590/S0074-02762011000100019
Rivera-Páez FA, Labruna MB, Martins TF, Sampieri BR, Camargo-Mathias MI. Amblyomma mixtum Koch, 1844 (Acari: Ixodidae): First record confirmation in Colombia using
morphological and molecular analyses. Ticks Tick Borne Dis. 2016;7:842-8. https://doi.org/10.1016/j.ttbdis.2016.03.020
Faccini-Martínez AA, Costa FB, Hayama-Ueno TE, Ramírez-Hernández A, Cortés-Vecino JA, Labruna MB, et al. Rickettsia rickettsii in Amblyomma patinoi ticks, Colombia. Emerg Infect Dis. 2015;21:537-9. https://doi.org/10.3201/eid2013.140721
Morrone JJ. Biogeographic areas and transition zones of Latin America and the Caribbean islands based on panbiogeographic and cladistic analyses of the entomofauna. Annu Rev Entomol. 2006;51:467-94. https://doi.org/10.1146/annurev.ento.50.071803.130447
Olson DM, Dinerstein E, Wikramanayake ED, Burgess ND, Powell GV, Underwood EC, et al. Terrestrial ecoregions of the world: A new map of life on earth a new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. BioScience. 2001;51:933-8. https://doi.org/10.1641/0006-3568(2001)051[0933:TEOTWA]2.0.CO;2
Sua S, Mateus RD, Vargas JC. Georreferenciación de registros biológicos y gacetero digital de localidades. Bogotá D.C.: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt.; 2004. p. 69.
Broxton PD, Zeng X, Scheftic W, Troch PA. A MODISbased 1 km maximum green vegetation fraction dataset. J Appl Meteorol Clim. 2014;53:1996-2004. https://doi.org/10.1175/JAMC-D-13-0356.1
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A. Very high resolution interpolated climate surfaces for global land areas. J Appl Meteorol. 2005;25:1965-78. https://doi.org/10.1002/joc.1276
Basille M, Calenge C, Marboutin E, Andersen R, Gaillard JM. Assessing habitat selection using multivariate statistics: Some refinements of the ecological-niche factor analysis. Ecol Modell. 2008;211:233-40. https://doi.org/10.1016/j.ecolmodel.2007.09.006
Calenge C. The package “adehabitat” for the R software: A tool for the analysis of space and habitat use by animals. Ecol Modell. 2006;197:516-9. https://doi.org/10.1016/j.ecolmodel.2006.03.017
Hirzel AH, Hausser J, Chessel D, Perrin N. Ecological niche factor analysis: How to compute habitat-suitability maps without absence data? Ecology. 2002;83:2027-36. https://doi.org/10.1890/0012-9658(2002)083[2027:ENFAHT]2.0.CO;2
Phillips SJ, Anderson RP, Schapire RE. Maximum entropy modeling of species geographic distributions. Ecol Model. 2006;190:231-59. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Stockwell D. The GARP modelling system: Problems and solutions to automated spatial prediction. Int J Geogr Inf Sci. 1999;13:143-58. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Merow C, Smith MJ, Silander JA. A practical guide to MaxEnt for modeling species’ distributions: What it does, and why inputs and settings matter. Ecography. 2013;36:1058-69. https://doi.org/10.1111/j.1600-0587.2013.07872.x
Peterson AT, Papeş M, Soberón J. Rethinking receiver operating characteristic analysis applications in ecological niche modeling. Ecol Model. 2008;213:63-72. https://doi.org/10.1016/j.ecolmodel.2007.11.008
Labruna MB, Mattar S, Nava S, Venzal JM, Dolz G, Abarca K, et al. Rickettsioses in Latin America, Caribbean, Spain and Portugal. Revista MVZ Córdoba. 2011;16:2435-57.
Mastropaolo M, Nava S, Guglielmone AA, Mangold AJ. Biological differences between two allopatric populations of Amblyomma cajennense (Acari: Ixodidae) in Argentina. Exp Appl Acarol. 2011;53:769-74. https://doi.org/10.1007/s10493-010-9404-6
Estrada-Peña A, Tarragona EL, Vesco U, Meneghi D de, Mastropaolo M, Mangold AJ, et al. Divergent environmental preferences and areas of sympatry of tick species in the Amblyomma cajennense complex (Ixodidae). Int J Parasitol. 2014;44:1081-9. https://doi.org/10.1016/j.ijpara.2014.08.007
Rivera-Páez FA, Labruna MB, Martins TF, Pérez JE, Castaño-Villa GJ, Ossa-López PA, et al. Contributions to the knowledge of hard ticks (Acari: Ixodidae) in Colombia. Ticks Tick Borne Dis. 2018;9:57-66. http://doi.org/10.1016/j.ttbdis.2017.10.008
Álvarez C, Bonilla M, Chacón G. Distribución de la garrapata Amblyomma cajennense (Acari: Ixodidae) sobre Bos taurus y Bos indicus en Costa Rica. Revista de Biología Tropical. 2000;48:129-35.
Martins TF, Barbieri AR, Costa FB, Terassini FA, Camargo LM, Peterka CR, et al. Geographical distribution of Amblyomma cajennense (sensu lato) ticks (Parasitiformes: Ixodidae) in Brazil, with description of the nymph of A. cajennense (sensu stricto). Parasit Vectors. 2016;9:186. https://doi.org/10.1186/s13071-016-1460-2
Strey OF, Teel PD, Longnecker MT, Needham GR. Survival and water-balance characteristics of unfed adult Amblyomma cajennense (Acari: Ixodidae). J Med Entomol. 1996;33:63-73.
Szabó MP, Pinter A, Labruna MB. Ecology, biology and distribution of spotted-fever tick vectors in Brazil. Front Cell Infect Microbiol. 2013;3:27. https://doi.org/10.3389/fcimb.2013.00027
Some similar items:
- María Elena Torres, Hugo Luis Rojas , Luis Carlos Alatorre , Luis Carlos Bravo , Mario Iván Uc , Manuel Octavio González , Lara Cecilia Wiebe , Alfredo Granados , Biogeographical factors determining Triatoma recurva distribution in Chihuahua, México, 2014 , Biomedica: Vol. 40 No. 3 (2020)
- Jaime Badel-Mogollón, Laura Rodríguez-Figueroa, Gabriel Parra-Henao, Spatio-temporal analysis of the biophysical and ecological conditions of Triatoma dimidiata (Hemiptera: Reduviidae: Triatominae) in the northeast region of Colombia , Biomedica: Vol. 37 No. Sup. 2 (2017): Suplemento 2, Entomología médica, 2017
- Jorge Miranda, Verónica Contreras, Yesica Negrete, Marcelo B. Labruna, Salim Mattar, Surveillance of Rickettsia sp. infection in capybaras (Hydrochoerus hydrochaeris) a potential model of epidemiological alert in endemic areas , Biomedica: Vol. 31 No. 2 (2011)
- Carolina Firacative, Germán Torres, María Claudia Rodríguez, Patricia Escandón, First environmental isolation of Cryptococcus gattii serotype B, from Cúcuta, Colombia , Biomedica: Vol. 31 No. 1 (2011)
- Leonor Chacín-Bonilla, Fernando Barrios, Cyclospora cayetanensis: biology, environmental distribution and transfer , Biomedica: Vol. 31 No. 1 (2011)
- Gabriel Parra-Henao, Laura Suárez, Mosquitoes (Diptera: Culiciadae) as potential vectors of arbovirused in the Urabá region, Northwest of Colombia , Biomedica: Vol. 32 No. 2 (2012)
- Alexandra Castañeda, Sandra Huérfano, Maria Claudia Rodríguez, Elizabeth Castañeda, Recovery of Cryptococcus neoformans var. gattii serotype C from almond trees detritus , Biomedica: Vol. 21 No. 1 (2001)
- Alexandra Castañeda, Elizabeth Castañeda, lsolation of Cryptococcus species from Eucalyptus trees located in a park in Bogotá , Biomedica: Vol. 21 No. 1 (2001)
- Alexander Salazar-Ceballos, Lídice Álvarez-Miño, Dengue incidence and its relationship with El Niño oceanic index, as a sensitive variable to anticipate outbreaks in the Colombian Caribbean region , Biomedica: Vol. 45 No. Sp. 2 (2025): Cambio climático y salud
- Alexandra Castañeda, Juan McEwen, Marylin Hidalgo, Elizabeth Castañeda, Cryptococcus spp. DNA extraction from environmental samples. , Biomedica: Vol. 24 No. 3 (2004)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























