Ten years of the immunogenetics laboratory performance assessment programme and its impact on the donor and transplant network
Abstract
Introduction. The use of immunological tests before solid organ transplantation is essential to reduce the risk of rejection and post-transplant complications. Therefore, quality control systems in laboratories performing them are necessary for clinical practice. The Colombian Instituto Nacional de Salud implemented the external evaluation program of transplant immunogenetics laboratory performance in 2014.
Objective. To evaluate the performance of the laboratories that carried out five of the immunological tests for transplants in Colombia between 2014 and 2023, according to information from the external evaluation program of transplant immunogenetics laboratory performance.
Materials and methods. We conducted a study of laboratory performance considering five immunological tests for transplantation: HLA, qualitative and quantitative PRA (Panel Reactive Antibodies), isolated antigen, and cross-matching tests. We collected data from reports of each laboratory. Based on the comparisons between laboratories, their performance was rated as “good”, “acceptable”, or “unacceptable” for each test. We calculated proportions and an analysis of predicted values with a 95% confidence interval.
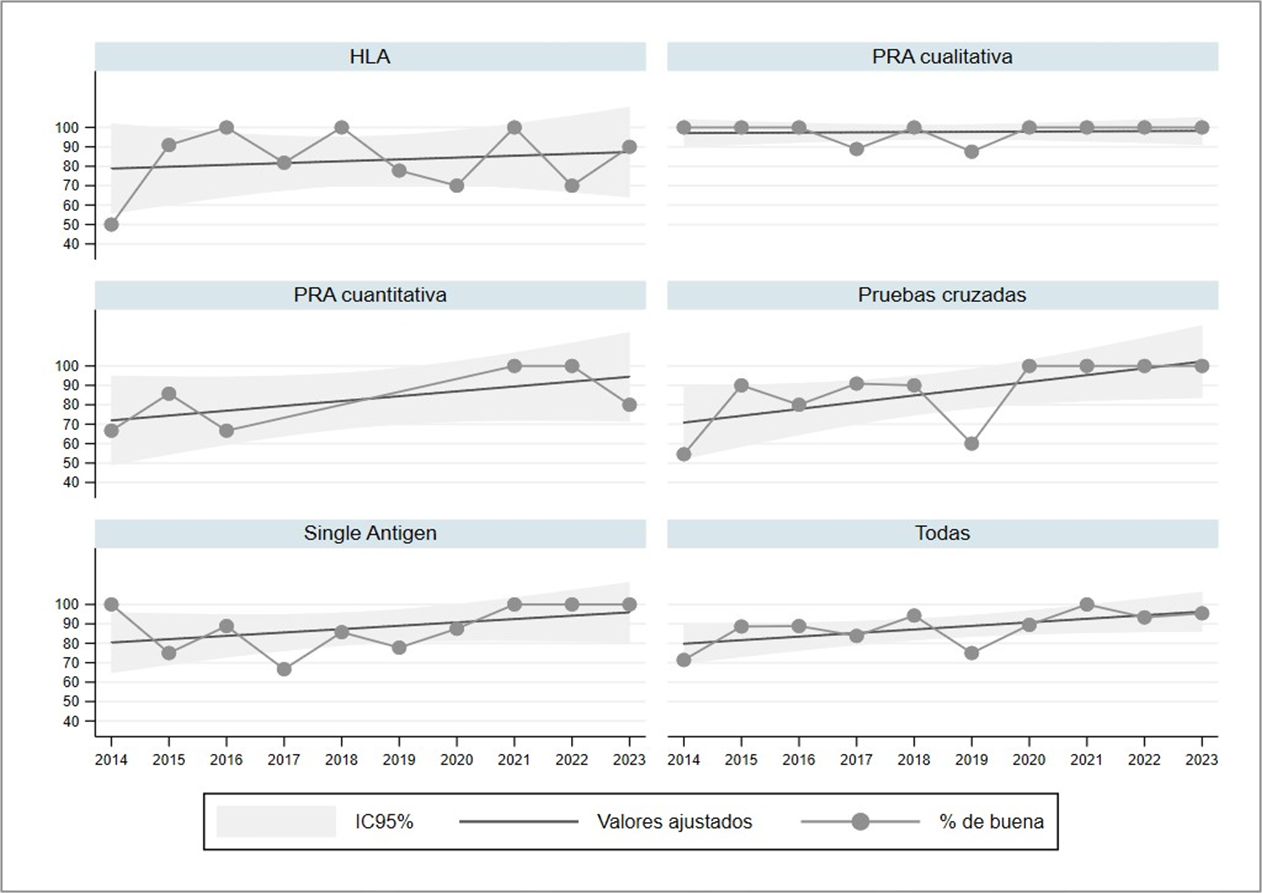
Results. The number of participating laboratories varied between 5 and 12, depending on the test. The proportion of laboratories with “good” performance was lower in the first year. The best performance was for qualitative PRA, rated as good in all the laboratories for eight years. In HLA (2014), qualitative PRA (2017 and 2019), crossmatch tests (2019), and single antigen (2017 and 2019) tests, the laboratories had a lower percentage of “good” performance than expected.
Conclusion. “Good” performance was observed in all the laboratories in each test during the last three years, except for HLA and quantitative PRA.
Downloads
References
Prieto L, Salinas MA, Arias YR, Rodríguez LM, Giraldo MC, Suárez DM, et al. Estándares de calidad para laboratorios que realizan pruebas de inmunogenética para trasplante de órganos. 2013. Fecha de consulta: 24 de febrero de 2024. Disponible en: https://www.ins.gov.co/Direcciones/RedesSaludPublica/GestiondeCalidadLaboratorios/ConsultaEstandaresparaLaboratoriosdeInmunologia/Estandares%20para%20laboratorios%20de%20inmunologia%20version%20online%202013.pdf
The American Society for Histocompatibility and Immunogenetics. Quality assurance and standards. Fecha de consulta: 24 de febrero de 2024. Disponible en: https://www.ashi-hla.org/page/standards
European Federation for Immunogenetics. Standards for histocompatibility immunogenetics. Fecha de consulta: 24 de febrero de 2024. Disponible en: https://efi-web.org/fileadmin/Efi_web/Standardv8_280819.pdf
McHugh ML. Interrater reliability: The kappa statistic. Biochem Med (Zagreb). 2012;22:276-82.
Ministerio de Salud. Resolución 8430 del 4 de octubre de 1993. Fecha de consulta: 30 de mayo de 2024. Disponible en: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/DE/DIJ/RESOLUCION-8430-DE-1993.pdf
Zhao X, Ma S, Wang B, Jiang X, The Han100K Initiative, Xu S. PGG.MHC: Toward understanding the diversity of major histocompatibility complexes in human populations. Nucleic Acids Res. 2023;51:D1102-8. https://doi.org/10.1093/nar/gkac997
Apps R, Qi Y, Carlson JM, Chen H, Gao X, Thomas R, et al. Influence of HLA-C expression level on HIV control. Science. 2013;340:87-91. https://doi.org/10.1126/science.1232685
Díaz-Peña R, Castro-Santos P, Aransay AM, Brüges-Armas J, Pimentel-Santos FM, López-Larrea C. Genetic study confirms association of HLA-DPA1*01:03 subtype with ankylosing spondylitis in HLA-B27-positive populations. Hum Immunol. 2013;74:764-7. https://doi.org/10.1016/j.humimm.2013.02.001
Migliorini F, Torsiello E, Spiezia F, Oliva F, Tingart M, Maffulli N. Association between HLA genotypes and COVID-19 susceptibility, severity and progression: A comprehensive review of the literature. Eur J Med Res. 2021;26:84. https://doi.org/10.1186/s40001-021-00563-1
Zeestraten ECM, Reimers MS, Saadatmand S, Goossens-Beumer IJ, Dekker JWT, Liefers GJ, et al. Combined analysis of HLA class I, HLA-E and HLA-G predicts prognosis in colon cancer patients. Br J Cancer. 2014;110:459-68. https://doi.org/10.1038/bjc.2013.696.
Montgomery RA, Tatapudi VS, Leffell MS, Zachary AA. HLA in transplantation. Nat Rev Nephrol. 2018;14:558-70. https://doi.org/10.1038/s41581-018-0039-x
Arias-Murillo YR, Salinas-Nova Ma, Montaño Ji. Criterios de asignación para trasplante renal en Colombia. Fecha de consulta: 12 de junio de 2024. Disponible en: https://www.ins.gov.co/BibliotecaDigital/consenso-criterios-asignacion-trasplante-renal-colombia.pdf
Salinas-Nova MA, Arias-Murillo YR, Patiño-Amaya YA, Rivera T, Jiménez-Urrea V, Pineda-Lozano JC, et al. Conferencia de consenso de recomendaciones para la construcción de un documento nacional de criterios de asignación para trasplante de corazón en Colombia. Instituto Nacional de Salud; 2022. Fecha de consulta: 12 de junio de 2024. Disponible en: https://www.ins.gov.co/BibliotecaDigital/consenso-criterios-de-asignacion-para-trasplantecardiaco-en-colombia.pdf
Instituto Nacional de Salud. Conferencia de consenso de criterios de asignación para trasplante de pulmón en Colombia. 2022. Fecha de consulta: 12 de junio de 2024. Disponible en: https://www.ins.gov.co/BibliotecaDigital/resultados-consenso-de-pulmon.pdf
Instituto Nacional de Salud. Informe ejecutivo Red de Donación y Trasplantes 2023. Fecha de consulta: 12 de junio de 2024. Disponible en: https://www.ins.gov.co/BibliotecaDigital/informe-ejecutivo-colombia-2023-anual.pdf
Instituto Nacional de Salud. Inscripción a los programas de evaluación externa del desempeño (PEED). Fecha de consulta: 12 de junio de 2024. Disponible en: https://www.ins.gov.co/TyS/programas-de-calidad
Levering WH, van den Beemd R, Marvelde JG, van Beers WA, Hooijkaas H, Sintnicolaas K, et al. External quality assessment of flow cytometric HLA-B27 typing. Cytometry. 2000;42:95-105.
Macardle PJ, McEvoy R, Jovanovich S. HLA-B27 expression by flow cytometry: An analysis of seven years quality assurance data. J Immunol Methods. 2000;243:51-7. https://doi.org/10.1016/s0022-1759(00)00227-1
Voorter CEM, Groeneveld L, Heidt S, Wieten L. Evaluation of 19 years of international external proficiency testing for high-resolution HLA typing. Front Genet. 2024;14:1290915. https://doi.org/10.3389/fgene.2023.1290915
Harmer A, Mascaretti L, Petershofen E. Accreditation of histocompatibility and immunogenetics laboratories: Achievements and future prospects from the European Federation for Immunogenetics Accreditation Programme. HLA. 2018;92:67-73. https://doi.org/10.1111/tan.13289
Lin G, Zhang K, Li J. A national proficiency scheme for human leucocyte antigen typing by next-generation sequencing. Clin Chim Acta. 2022;533:85-8. https://doi.org/10.1016/j.cca.2022.06.015
Duquesnoy RJ, Marrari M. Multilaboratory evaluation of serum analysis for HLA antibody and crossmatch reactivity by lymphocytotoxicity methods. Arch Pathol Lab Med. 2003;127:149-56. https://doi.org/10.5858/2003-127-149-MEOSAF
Chong AS. Mechanisms of organ transplant injury mediated by B cells and antibodies: Implications for antibody-mediated rejection. Am J Transplant. 2020;20:23-32. https://doi.org/10.1111/ajt.15844
Loupy A, Hill GS, Jordan SC. The impact of donor-specific anti-HLA antibodies on late kidney allograft failure. Nat Rev Nephrol. 2012;8:348-57. https://doi.org/10.1038/nrneph.2012.81
Tait BD, Süsal C, Gebel HM, Nickerson PW, Zachary AA, Claas FHJ, et al. Consensus guidelines on the testing and clinical management issues associated with HLA and non-HLA antibodies in transplantation. Transplantation. 2013;95:19-47. https://doi.org/10.1097/TP.0b013e31827a19cc
Reinsmoen NL, Lai CH, Vo A, Cao K, Ong G, Naim M, et al. Acceptable donor-specific antibody levels allowing for successful deceased and living donor kidney transplantation after desensitization therapy. Transplantation. 2008;86:820-5. https://doi.org/10.1097/TP.0b013e3181856f98
Cecka JM. Current methodologies for detecting sensitization to HLA antigens. Curr Opin Organ Transplant. 2011;16:398-403. https://doi.org/10.1097/MOT.0b013e328348980a
Rodríguez-Ramírez S. Al Jurdi AD. Konvalinka AG. Riella L. Antibody-mediated rejection: Prevention, monitoring and treatment dilemmas. Curr Opin Organ Transplant. 2022;27:405-14. https://doi.org/10.1097/MOT.0000000000001011
Tait BD, Hudson F, Brewin G, Cantwell L, Holdsworth R. Solid phase HLA antibody detection technology--challenges in interpretation. Tissue Antigens. 2010;76:87-95. https://doi.org/10.1111/j.1399-0039.2010.01486.x
Oh EJ, Park H, Park KU, Kang ES, Kim HS, Song EY. Interlaboratory comparison of the results of lifecodes LSA class I and class II single antigen kits for human leukocyte antigen antibody detection. Ann Lab Med. 2015;35:321-8. https://doi.org/10.3343/alm.2015.35.3.321
Some similar items:
- Paula Ximena Pavía, Nubia Lucía Roa, Ana María Uribe, Concepción Judith Puerta, Using S35-S36 and TcH2AF-R primer-based PCR tests to follow-up a Chagas’ disease patient who had undergone a heart transplant , Biomedica: Vol. 31 No. 2 (2011)
- John F. Arboleda, Luis F. García, Cristiam M. Álvarez, ILT3+/ILT4+ tolerogenic dendritic cells and their influence on allograft survival , Biomedica: Vol. 31 No. 2 (2011)
- Lorenzo Cáceres, José Rovira, Rolando Torres, Arsenio García, José Calzada, Manuel De La Cruz, Characterization of Plasmodium vivax malaria transmission at the border of Panamá and Costa Rica , Biomedica: Vol. 32 No. 4 (2012)
- Carmen E. Alarcón, Hernán Hurtado, Jaime E. Castellanos, Avian antibodies: an alternative in production and diagnosis , Biomedica: Vol. 20 No. 4 (2000)
- Mario García, Liliana Alejandra Chicaíza, Hoover Quitián, Adriana Linares, Óscar Ramírez, Cost-effectiveness of consolidation treatments for acute myeloid leukemia in high-risk pediatric patients within the Colombian healthcare system , Biomedica: Vol. 35 No. 4 (2015)
- Yazmin Rocío Arias, Karime Osorio-Arango, Brayan Bayona, Guadalupe Ercilla, Mauricio Beltrán-Durán, Determination of HLA-A, -B and -DRB1 polymorphism in brain dead organ donors representative of the Colombian general population, 2007-2014 , Biomedica: Vol. 37 No. 2 (2017)
- Lina Echeverri-Toro, Andrés Arango, Sigifredo Ospina, Carlos Agudelo, Bordetella bronchiseptica recurrent bacteraemia in a patient with bone marrow transplantation , Biomedica: Vol. 35 No. 3 (2015)
- Fiona Xacur-García, Rodrigo Díaz-Novelo, Linnete Herrera-David, Paulina Moreno-Arjona, Nina Méndez-Domínguez, Manifestaciones clínicas y evolución a largo plazo de tres casos de rosácea ocular atendidos en un hospital de alta especialidad del sureste de México , Biomedica: Vol. 40 No. 3 (2020)
- Julio Cesar Padilla-Rodríguez , Mario Javier Olivera, Pablo Chaparro , Martha Lucía Quiñonez , José Pablo Escobar , Gilberto Álvarez , The malaria eradication campaign in Colombia, 1959-1979 , Biomedica: Vol. 42 No. 2 (2022)
- Andrés Felipe Zea-Vera, Lina María Castaño-Jaramillo, Cruising the transition: Challenges and opportunities when caring for immunity inborn error patients , Biomedica: Vol. 44 No. Sp. 2 (2024): Inmunología clínica
Copyright (c) 2024 Biomedica

This work is licensed under a Creative Commons Attribution 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























