Microscopic and molecular evaluation of Strongyloides venezuelensis in an experimental life cycle using Wistar rats
Abstract
Introduction: Strongyloides venezuelensis is a nematode whose natural host is rats. It is used as a model for the investigation of human strongyloidiasis caused by S. stercoralis. The latter is a neglected tropical disease in Ecuador where there are no specific plans to mitigate this parasitic illness.
Objective: To evaluate the stages of S. venezuelensis in an experimental life cycle using Wistar rats.
Materials and methods: Male Wistar rats were used to replicate the natural biological cycle of S. venezuelensis and describe its morphometric characteristics, as well as its parasitic development. Furthermore, the production of eggs per gram of feces was quantified using two diagnostic techniques and assessment of parasite load: Kato-Katz and qPCR.
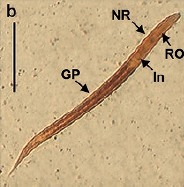
Results: Viable larval stages (L1, L2, L3) could be obtained up to 96 hours through fecal culture. Parthenogenetic females were established in the duodenum on the fifth day postinfection. Fertile eggs were observed in the intestinal tissue and fresh feces where the production peak occurred on the 8th. day post-infection. Unlike Kato-Katz, qPCR detected parasitic DNA on days not typically reported.
Conclusions: The larval migration of S. venezuelensis within the murine host in an experimental environment was equivalent to that described in its natural biological cycle. The Kato-Katz quantitative technique showed to be quick and low-cost, but the qPCR had greater diagnostic precision. This experimental life cycle can be used as a tool for the study of strongyloidiasis or other similar nematodiasis.
Downloads
References
Buonfrate D, Mena MA, Angheben A, Requema-Méndez A, Muñoz J, Gobbi F, et al. Prevalence of strongyloidiasis in Latin America: A systematic review of the literature.
Epidemiol Infect. 2015;143:452-60. https://doi.org/10.1017/S0950268814001563
Viney ME, Lok JB. The biology of Strongyloides spp. Wormbook: The online review of C. elegans biology. 2015:1-17. Access date: 11-18-2019. Available from: https://doi.org/10.1895/wormbook.1.141.2
Jacobsen KH, Ribeiro PS, Quist BK, Rydbeck BV. Prevalence of intestinal parasites in young Quichua children in the highlands of rural Ecuador. J Health Popul Nutr. 2007;25:399-405.
Reyes-Chacón JA. Evaluación del RT-PCR en el diagnóstico de 6 parásitos intestinales en un área con parasitismo de baja intensidad en el trópico, Ecuador. Quito: Universidad San Francisco de Quito; 2012. Available from: https://repositorio.usfq.edu.ec/handle/23000/2043
Júnior AF, Gonçalves-Pires MR, Silva DA, Gonçalves ALR, Costa-Cruz JM. Parasitological and serological diagnosis of Strongyloides stercoralis in domesticated dogs from southeastern Brazil. Vet Parasitol. 2006;136:137-45. https://doi.org/10.1016/j.vetpar.2005.10.022
Mati VLT, Raso P, de Melo AL. Strongyloides stercoralis infection in marmosets: Replication of complicated and uncomplicated human disease and parasite biology. Parasit Vectors. 2014;7:579. https://doi.org/10.1186/s13071-014-0579-2
Nutman TB. Human infection with Strongyloides stercoralis and other related Strongyloides species. Parasitology. 2017;144:263-73. https://doi.org/10.1017/S0031182016000834
Ruano AL. Óxido nítrico como modulador de la estrongiloidosis. Salamanca: Universidad de Salamanca; 2008. Available from: https://dialnet.unirioja.es/servlet/tesis?codigo=102628
Viney M, Kikuchi T. Strongyloides ratti and S. venezuelensis-rodent models of Strongyloides infection. Parasitology. 2017;144:285-94. https://doi.org/10.1017/S0031182016000020
Chaves LA, Gonçalves ALR, Paula FM, Silva NM, Silva CV, Costa-Cruz JM, et al. Comparison of parasitological, immunological and molecular methods for evaluation of faecal samples of immunosuppressed rats experimentally infected with Strongyloides venezuelensis. Parasitology. 2015;142:1715-21. https://doi.org/10.1017/S0031182015001298
Marques PD, Malta FM, Meisel DMCL, Corral MA, Pinho JR, Costa-Cruz JM, et al. Diagnosis of the strongyloid nematode Strongyloides venezuelensis in experimentally infected rats. J Helminthol. 2016;90:422-7. https://doi.org/10.1017/S0022149X15000528
Viney M. Strongyloides. Parasitology. 2017;144:259-62. https://doi.org/10.1017/S0031182016001773
Bosqui LR, Marques PD, de Melo GB, Gonçalves-Pires MR, Malta FM, Pavanelli WR, et al. Molecular and immune diagnosis: Further testing for human strongyloidiasis. Mol Diagn Ther. 2018;22:485-91. https://doi.org/10.1007/s40291-018-0340-1
Organización Mundial de la Salud. Medios auxiliares para el diagnóstico de las parasitosis intestinales. Ginebra: OMS; 1994. Available from: https://www.paho.org/es/documentos/medios-auxiliares-para-diagnostico-parasitosis-intestinales
Girard de Kaminsky R. Métodos para laboratorios de atención primaria de salud. Segunda edición. Tegucigalpa: Universidad Nacional Autónoma de Honduras y Hospital-Escuela; 2003. Available from: http://www.bvs.hn/Honduras/pdf/Manual%20Parasitologia%202007.pdf
Rugai E, Mattos T, Brisola AP. A new technic for the isolation of nematode larvae from faeces; modification of Baermann’s method. Rev Inst Adolfo Lutz. 1954;14:5-8.
Verweij JJ, Canales M, Polman K, Ziem J, Brienen EAT, Polderman AM, et al. Molecular diagnosis of Strongyloides stercoralis in faecal samples using real-time PCR. Trans R Soc Trop Med Hyg. 2009;103:342-46. https://doi.org/10.1016/j.trstmh.2008.12.001
Beaver PCh, Jung RC, Cupp EW. Rhabditida: Strongyloides y formas relacionadas. Parasitología clínica de Craig Faust. Tercera edición. México DF: Masson Doyma S. A.; 2003.
Mehlhorn H. Encyclopedia of parasitology. Third edition. New York: Springer. 2008.
Marra NM, Chiuso-Minicucci F, Machado GC, Zorzella-Pezavento SF, França TG, Ishikawa LL, et al. Migratory route of Strongyloides venezuelensis in Lewis rats: Comparison of histological analyses and PCR. Exp Parasitol. 2011;127:334-9. https://doi.org/10.1016/j.exppara.2010.08.006
Gonçalves ALR, Silva CV, Ueta MT, Costa-Cruz JM. Antigen, antibody and immune complex detection in serum samples from rats experimentally infected with Strongyloides venezuelensis. Exp Parasitol. 2012;130:205-8. https://doi.org/10.1016/j.exppara.2012.01.007
Paula FM, Sitta RB, Malta FM, Gottardi M, Corral MA, Gryschek RCB, et al. Parasitological and molecular diagnosis in experimental Strongyloides venezuelensis infection. Rev Inst Med Trop São Paulo. 2013;55:141-3. https://doi.org/10.1590/S0036-46652013000200015
Vieira FM, Lima SS, Bessa EC. Morfologia e biometria de ovos e larvas de Strongyloides sp. grassi, 1879 (Rhabditoidea: Strongyloididae) parasito gastrointestinal de Hydrochaeris hydrochaeris (Linnaeus, 1766) (Rodentia: Hydrochaeridae), no Município de Juiz de Fora, Minas Gerais. Rev Bras Parasitol Vet. 2006;15:7-12.
El-Malky M, Maruyama H, Hirabayashi Y, Shimada S, Yoshida A, Amano T, et al. Intraepithelial infiltration of eosinophils and their contribution to the elimination of adult intestinal nematode, Strongyloides venezuelensis in mice. Parasitol Int. 2003;52:71-9. https://doi.org/10.1016/s1383-5769(02)00086-7
El-Malky MA, Maruyama H, Al-Harthi SA, El-Beshbishi SN, Ohta N. The role of B-cells in immunity against adult Strongyloides venezuelensis. Parasit Vectors. 2013;6:148. https://doi.org/10.1186/1756-3305-6-148
Sasaki Y, Yoshimoto T, Maruyama H, Tegoshi T, Ohta N, Arizono N, et al. IL-18 with IL-2 protects against Strongyloides venezuelensis infection by activating mucosal mast celldependent type 2 innate immunity. J Exp Med. 2005;202:607-16. https://doi.org/10.1084/jem.20042202
Takamure A. Migration route of Strongyloides venezuelensis in rodents. Int J Parasitol. 1995;25:907-11. https://doi.org/10.1016/0020-7519(95)00014-s
Wynn TA. IL-13 effector functions. Annu Rev Immunol. 2003;21:425-56. https://doi.org/10.1146/annurev.immunol.21.120601.141142
Gabriel JA, Rueda MM, Canales M, Sánchez A. Utilidad del método Kato-Katz para diagnóstico de uncinarias: experiencia en una zona rural de Honduras, 2011. Rev Med Hondur. 2012;80:96-101.
Easton AV, Oliveira RG, Walker M, O’Connell EM, Njenga SM, Mwandawiro ChS, et al. Sources of variability in the measurement of Ascaris lumbricoides infection intensity by Kato-Katz and qPCR. Parasit Vectors. 2017;10:256. https://doi.org/10.1186/s13071-017-2164-y
Gonçalves ALR, Silva CV, Carvalho EFG, Ueta MT, Costa-Cruz JM. Transmammary transmission of strongyloidiasis in immunosuppressed rats. Neotrop Helminthol. 2013;7:195-200. https://doi.org/10.2307/3285443
Some similar items:
- Elpidia Poveda, Pilar Trujillo, Francisco Ruiz, Elizabeth Lopez, Glucose and insulin levels in Wistar rats submitted to high fat diet and treatment with mimetic leptin peptides , Biomedica: Vol. 28 No. 1 (2008)
- Biviana Andrea Duque, Diego Aranzazu, Piedad Agudelo-Flórez, Andrés F. Londoño, Víctor H. Quiroz, Juan David Rodas, Rattus norvegicus as an indicator of circulation of Capillaria hepatica and Taenia taeniaeformis on a groceries trade center of Medellín, Colombia , Biomedica: Vol. 32 No. 4 (2012)
- Luis Solórzano-Alava, Francisco Sánchez-Amador, Talia Valverde, Angiostrongylus (Parastrongylus) cantonensis on intermediate and definitive hosts in Ecuador, 2014-2017 , Biomedica: Vol. 39 No. 2 (2019)
- Mario Francisco Guerrero, Elements for the effective evaluation of natural products with possible antihypertensive effects , Biomedica: Vol. 29 No. 4 (2009)
- Lauro Figueroa, Francisco Díaz, Avelardo Camacho, Eliseo Díaz, Rolando Marvin, Activity induced by androsterone and hemisuccinate of androsterone on perfusion pressure and vascular resistance , Biomedica: Vol. 29 No. 4 (2009)
- Diana Urrego, Alejandro Múnera, Julieta Troncoso, Peripheral facial nerve lesion induced long-term dendritic retraction in pyramidal cortico-facial neurons , Biomedica: Vol. 31 No. 4 (2011)
- Ángela Liliana Londoño-Franco, Juliana Loaiza-Herrera, Fabiana María Lora-Suárez, Jorge Enrique Gómez-Marín, Blastocystis sp. frequency and sources among children from 0 to 5 years of age attending public day care centers in Calarcá, Colombia , Biomedica: Vol. 34 No. 2 (2014)
- Elpidia Poveda, Paola Ayala, Milena Rodríguez, Edgar Ordóñez, Cesar Baracaldo, Willman Delgado, Martha Guerra, Effects of vegetal oil supplementation on the lipid profile of Wistar rats . , Biomedica: Vol. 25 No. 1 (2005)
- Silvana Marisa Montenegro, María Cristina Tarrés, Juan Carlos Picena, Stella Maris Martínez, Feeding behavior and glycemic profile in two lines of rats with genetic diabetes. , Biomedica: Vol. 25 No. 4 (2005)
- Beatriz Eugenia Alvarado, Luis Reinel Vásquez, Social determinants, feeding practices and nutritional consequences of intestinal parasitism in young children. , Biomedica: Vol. 26 No. 1 (2006)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























