Prediction of sensitivity to warfarin based on VKORC1 and CYP2C9 polymorphisms in patients from different places in Colombia
Abstract
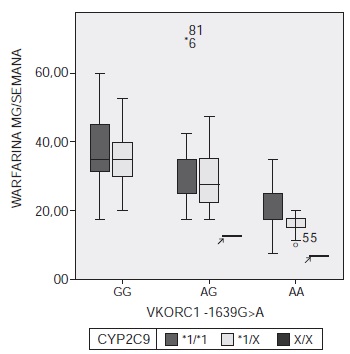
Introduction: In the search to prevent hemorrhages associated with anticoagulant therapy, a major goal is to validate predictors of sensitivity to warfarin. However, previous studies in Colombia that included polymorphisms in the VKORC1 and CYP2C9 genes as predictors reported different algorithm performances to explain dose variations, and did not evaluate the prediction of sensitivity to warfarin. Objective: To determine the accuracy of the pharmacogenetic analysis, which includes the CYP2C9 *2 and *3 and VKORC1 1639G>A polymorphisms in predicting patients’ sensitivity to warfarin at the Hospital Militar Central, a reference center for patients born in different parts of Colombia. Materials and methods: Demographic and clinical data were obtained from 130 patients with stable doses of warfarin for more than two months. Next, their genotypes were obtained through a melting curve analysis. After verifying the Hardy-Weinberg equilibrium of the genotypes from the polymorphisms, a statistical analysis was done, which included multivariate and predictive approaches. Results: A pharmacogenetic model that explained 52.8% of dose variation (p<0.001) was built, which was only 4% above the performance resulting from the same data using the International Warfarin Pharmacogenetics Consortium algorithm. The model predicting the sensitivity achieved an accuracy of 77.8% and included age (p=0.003), polymorphisms *2 and *3 (p=0.002) and polymorphism 1639G>A (p<0.001) as predictors. Conclusions: These results in a mixed population support the prediction of sensitivity to warfarin based on polymorphisms in VKORC1 and CYP2C9 as a valid approach in Colombian patients.
Downloads
References
Carlquist JF, Horne BD, Muhlestein JB, Lappe DL, Whiting BM, Kolek MJ, et al . Genotypes of the cytochrome p450 isoform, CYP2C9 , and the vitamin K epoxide reductase complex subunit 1 conjointly determine stable warfarin dose: A prospective study. J Thromb Thrombolysis. 2006;22: 191-7. http://dx.doi.org/10.1007/s11239-006-9030-7
Johnson JA, Gong L, Whirl-Carrillo M, Gage BF, Scott SA, Stein CM, et al . Clinical Pharmacogenetics Implementation Consortium Guidelines for CYP2C9 and VKORC1 genotypes and warfarin dosing. Clin Pharmacol Ther. 2011;90:625-9. http://dx.doi.org/10.1038/clpt.2011.185
Stehle S, Kirchheiner J, Lazar A, Fuhr U. Pharmacogenetics of oral anticoagulants: A basis for dose individualization. Clin Pharmacokinet. 2008;47:565-94.
Kamali F, Wynne H. Pharmacogenetics of warfarin. Annu Rev Med. 2010;61:63-75. http://dx.doi.org/10.1146/annurev.med.070808.170037
Hirsh J, Fuster V, Ansell J, Halperin JL. American Heart Association/American College of Cardiology Foundation guide to warfarin therapy. Circulation. 2003;107:1692-711. http://dx.doi.org/10.1161/01.CIR.0000063575.17904.4E
Li T, Lange LA, Li X, Susswein L, Bryant B, Malone R, et al . Polymorphisms in the VKORC1 gene are strongly associated with warfarin dosage requirements in patients receiving anticoagulation. J Med Genet. 2006;43:740-4. http://dx.doi.org/10.1136/jmg.2005.040410
Bilen O, Teruya J. Complications of anticoagulation. Dis Mon. 2012;58:440-7. http://dx.doi.org/10.1016/j.disamonth.2012.04.002
Goldstein JN, Rosand J, Schwamm LH. Warfarin reversal in anticoagulant-associated intracerebral hemorrhage. Neurocrit Care. 2008;9:277-83. http://dx.doi.org/10.1007/s12028-008-9049-z
Pirmohamed M, Burnside G, Eriksson N, Jorgensen AL, Toh CH, Nicholson T, et al . A randomized trial of genotype- guided dosing of warfarin. N Engl J Med. 2013;369:2294-303. http://dx.doi.org/10.1056/NEJMoa1311386
Gage BF, Eby C, Johnson JA, Deych E, Rieder MJ, Ridker PM, et al . Use of pharmacogenetic and clinical factors to predict the therapeutic dose of warfarin. Clin Pharmacol Ther. 2008;84:326-31 . http://dx.doi.org/10.1038/clpt.2008.10
Wadelius M, Pirmohamed M. Pharmacogenetics of warfarin: Current status and future challenges. Pharmacogenomics J. 2007;7:99-111. http://dx.doi.org/10.1038/sj.tpj.6500417
Wang D, Chen H, Momary KM, Cavallari LH, Johnson JA, Sadee W. Regulatory polymorphism in vitamin K epoxide reductase complex subunit 1 ( VKORC1 ) affects gene expression and warfarin dose requirement. Blood. 2008;112:1013-21. http://dx.doi.org/10.1182/blood-2008-03-144899
Dean L. Warfarin therapy and the genotypes CYP2C9 and VKORC1 . Medical genetics summaries. Bethesda, MD: National Center for Biotechnology Information; 2013.
Finkelman BS, Gage BF, Johnson JA, Brensinger CM, Kimmel SE. Genetic warfarin dosing: Tables versus algorithms. J Am Coll Cardiol. 2011;57:612-8. http://dx.doi.org/10.1016/j.jacc.2010.08.643
Klein TE, Altman RB, Eriksson N, Gage BF, Kimmel SE, Lee MT, et al . Estimation of the warfarin dose with clinical and pharmacogenetic data. N Engl J Med. 2009;360:753-64. http://dx.doi.org/10.1056/NEJMoa0809329
Bazan NS, Sabry NA, Rizk A, Mokhtar S, Badary O. Validation of pharmacogenetic algorithms and warfarin dosing table in Egyptian patients. Int J Clin Pharm. 2012;34:837-44. http://dx.doi.org/10.1007/s11096-012-9678-3
Liang R, Li L, Li C, Gao Y, Liu W, Hu D, et al . Impact of CYP2C9 *3, VKORC1 -1639, CYP4F2rs2108622 genetic polymorphism and clinical factors on warfarin maintenance dose in Han-Chinese patients. J Thromb Thrombolysis. 2012;34:120-5. http://dx.doi.org/10.1007/s11239-012-0725-7
Salzano FM. Interethnic variability and admixture in Latin America--social implications. Rev Biol Trop . 2004;52:405-15.
Roper N, Storer B, Bona R, Fang M. Validation and comparison of pharmacogenetics-based warfarin dosing algorithms for application of pharmacogenetic testing. J Mol Diagn. 2010;12:283-91 . http://dx.doi.org/10.2353/jmoldx.2010.090110
Palacio L, Falla D, Tobón I, Mejía F, Lewis JE, Martínez AF, et al . Pharmacogenetic impact of VKORC1 and CYP2C9 allelic variants on warfarin dose requirements in a hispanic population isolate. Clin Appl Thromb Hemost. 2010;16:83-90. http://dx.doi.org/10.1177/1076029608330472
Isaza C, Beltrán L, Henao J, Porras G, Pinzón A, Vallejos A, et al . Factores genéticos y ambientales asociados con la respuesta a warfarina en pacientes colombianos. Biomédica. 2010;30:410-20 . http://dx.doi.org/10.7705/biomedica.v30i3.275
Saleh MI. Clinical predictors associated with warfarin sensitivity . Am J Ther. 2015. http://dx.doi.org/10.1097/MJT.0000000000000248
Burian M, Grosch S, Tegeder I, Geisslinger G. Validation of a new fluorogenic real-time PCR assay for detection of CYP2C9 allelic variants and CYP2C9 allelic distribution in a German population. Br J Clin Pharmacol. 2002;54:518-21. http://dx.doi.org/10.1046/j.1365-2125.2002.01693.x
Veenstra DL, You JH, Rieder MJ, Farin FM, Wilkerson HW, Blough DK, et al . Association of vitamin K epoxide reductase complex 1 ( VKORC1 ) variants with warfarin dose in a Hong Kong Chinese patient population. Pharmacogenet Genomics. 2005;15:687-91.
Cifuentes RA, Barreto E. Supervised selection of single nucleotide polymorphisms in chronic fatigue syndrome. Biomédica. 2011;31:613-21 . http://dx.doi.org/10.1590/S0120-41572011000400017
Frank E, Hall M, Trigg L, Holmes G, Witten IH. Data mining in bioinformatics using Weka. Bioinformatics. 2004;20:2479- 81. http://dx.doi.org/10.1093/bioinformatics/bth261
Williams D, Feely J. Pharmacokinetic-pharmacodynamic drug interactions with HMG-CoA reductase inhibitors. Clin Pharmacokinet. 2002;41:343-70. http://dx.doi.org/10.2165/00003088-200241050-00003
Andrus MR. Oral anticoagulant drug interactions with statins: Case report of fluvastatin and review of the literature. Pharmacotherapy. 2004;24:285-90. http://dx.doi.org/10.1592/phco.24.2.285.33137
McKenney JM. Efficacy and safety of rosuvastatin in treatment of dyslipidemia. Am J Health Syst Pharm. 2005;62:1033-47.
Yunis JJ, Acevedo LE, Campo DS, Yunis EJ. Geno- geographic origin of Y-specific STR haplotypes in a sample of Caucasian-Mestizo and African-descent male individuals from Colombia. Biomédica. 2013;33:459-67. http://dx.doi.org/10.7705/biomedica.v33i3.807
Departamento Administrativo Nacional de Estadística . Colombia una nación multicultural. Su diversidad étnica - 2007. Fecha de consulta: 14 de julio de 2015. Disponible en: https://www.dane.gov.co/files/censo2005/etnia/sys/colombia_nacion.pdf.
Flockhart DA, O´Kane D, Williams MS, Watson MS, Gage B, Gandolfi R, et al . Pharmacogenetic testing of CYP2C9 and VKORC1 alleles for warfarin. Genet Med. 2008;10:139- 50. http://dx.doi.org/10.1097/GIM.0b013e318163c35f
Holbrook AM, Pereira JA, Labiris R, McDonald H, Douketis JD, Crowther M, et al . Systematic overview of warfarin and its drug and food interactions. Arch Intern Med. 2005;165:1095-106. http://dx.doi.org/10.1001/archinte.165.10.1095
Geisen C, Watzka M, Sittinger K, Steffens M, Daugela L, Seifried E, et al . VKORC1 haplotypes and their impact on the inter-individual and inter-ethnical variability of oral anticoagulation. Thromb Haemost. 2005;94:773-9. http://dx.doi.org/10.1160/TH05-04-0290
Rieder MJ, Reiner AP, Gage BF, Nickerson DA, Eby CS, McLeod HL, et al . Effect of VKORC1 haplotypes on transcriptional regulation and warfarin dose. N Engl J Med. 2005;352:2285-93. http://dx.doi.org/10.1056/NEJMoa044503
Kadian-Dodov DL, van der Zee SA, Scott SA, Peter I, Martis S, Doheny DO, et al . Warfarin pharmacogenetics: A controlled dose-response study in healthy subjects. Vasc Med. 2013;18:290-7. http://dx.doi. org/10.1177/1358863X13503193
Nutescu E, Chuatrisorn I, Hellenbart E. Drug and dietary interactions of warfarin and novel oral anticoagulants: An update. J Thromb Thrombolysis. 2011;31:326-43. http://dx.doi.org/10.1007/s11239-011-0561-1
Chan TY. Adverse interactions between warfarin and nonsteroidal antiinflammatory drugs: Mechanisms, clinical significance, and avoidance. Ann Pharmacother. 1995;29: 1274-83.
Izzo AA, Ernst E. Interactions between herbal medicines and prescribed drugs: An updated systematic review. Drugs. 2009;69:1777-98. http://dx.doi.org/10.2165/11317010-000000000-00000
Motsinger-Reif AA, Wagner MJ. Clinical trial evidence of the promise of pharmacogenomics warfarin dosing algorithms. Pharmacogenomics. 2012;13:861-3. http://dx.doi.org/10.2217/pgs.12.65
Wang M, Lang X, Cui S, Fei K, Zou L, Cao J, et al . Clinical application of pharmacogenetic-based warfarin-dosing algorithm in patients of Han nationality after rheumatic valve replacement: A randomized and controlled trial. Int J Med Sci. 2012;9:472-9. http://dx.doi.org/10.7150/ijms.4637
Some similar items:
- Luz Elena Velásquez, Catalina Gómez, Erika Valencia, Laura Salazar, Eudoro Casas, Paragonimosis in the peri-urban zone of Medellín, Antioquia , Biomedica: Vol. 28 No. 3 (2008)
- Carlos Isaza, Leonardo Beltrán, Julieta Henao, Gloria Porras, Alfredo Pinzón, Álvaro Vallejos, Jorge Machado, Genetic and bioenvironmental factors associated with warfarin response in Colombian patients , Biomedica: Vol. 30 No. 3 (2010)
- Sandra Milena Arias, Lina Marcela Salazar, Eudoro Casas, Alexandra Henao, Luz Elena Velásquez, Paragonimus sp. in crabs and awareness of the educational community to aquatic ecosystems in La Miel and La Clara, Caldas, Antioquia , Biomedica: Vol. 31 No. 2 (2011)
- Jaiberth Cardona-Arias, Luz Peláez-Vanegas, Juan López-Saldarriaga, Marcela Duque-Molina, Oscar Leal-Álvarez, Health related quality of life in adults with HIV/AIDS in Colombia , Biomedica: Vol. 31 No. 4 (2011)
- Carlos Alberto Palacio, Jenny García, María Patricia Arbeláez, Ricardo Sánchez, Beatriz Aguirre, Isabel Cristina Garcés, Gabriel Jaime Montoya, Juliana Gómez, Angela Agudelo, Carlos Alberto López, Jorge Julián Calle, Carlos Alberto Cardeño, Juan Fernando Cano, María Cecilia López, Patricia Montoya, Claudia Patricia Herrera, Natalia González, Alejandro González, Gabriel Bedoya, Andrés Ruiz, Jorge Ospina, Validation of the Diagnostic Interview for Genetic Studies (DIGS) in Colombia. , Biomedica: Vol. 24 No. 1 (2004)
- German Eduardo Rueda, Luis Alfonso Díaz, Adalberto Campo, Jaider Alfonso Barros, Gissel Constanza Avila, Lutzi Tathiana Oróstegui, Betty Cecilia Osorio, Laura del Pilar Cadena, Validation of the SCOFF questionnaire for screening of eating disorders in university women. , Biomedica: Vol. 25 No. 2 (2005)
- Oscar F. Herrán, María F. Ardila, Internal validity and reliability of the CAGE test in Bucaramanga, Colombia. , Biomedica: Vol. 25 No. 2 (2005)
- Ángel Tito Alvarado, Ana María Muñoz, Berta Loja, Jessica Michiko Miyasato, Jorge Antonio García, Roberto Andrés Cerro, Luis Abel Quiñones, Nelson Miguel Varela, Study of the allelic variants CYP2C9*2 and CYP2C9*3 in samples of the Peruvian mestizo population , Biomedica: Vol. 39 No. 3 (2019)
- Mirta Cavieres , Marcelo Suárez , Gabriel Verón , Luis Abel Quiñones , Nelson Miguel Varela, Retrospective pharmacogenetic analysis of a pediatric patient under anticoagulant treatment: Clinical case , Biomedica: Vol. 41 No. 3 (2021)
- Ana Yibby Forero Torres, Luis Carlos Forero, Comparison between anthropometric measurements and indices to evaluate general and abdominal obesity, Colombia ENSIN 2015 , Biomedica: Vol. 43 No. Sp. 3 (2023): Enfermedades crónicas no transmisibles
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























