Streptozotocin diabetogenic action in an experimental neonatal induction model
Abstract
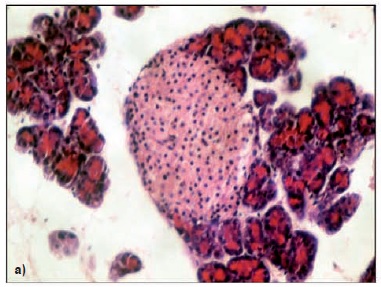
Introduction: The use of experimental models is essential to study the pathophysiological mechanisms of diabetes. Objectives: To compare in adult Wistar rats the diabetogenic action of streptozotocin according to the moment and route of administration during the neonatal period by evaluating biochemical, metabolic and histological variables. Materials and methods: Eight groups of neonatal female Wistar rats (n=10) were formed. We evaluated the induction with streptozotocin (100 mg/kg of body weight) on days 2 and 5 after birth, as well as the administration routes (subcutaneous or intraperitoneal). Controls were injected with sodium citrate buffer. Blood glucose level, body weight, food and water intake were monitored for 12 weeks. We also performed tolerance tests for oral glucose and glycosylated hemoglobin, and a histopathological pancreas morphometric study. Results: The mortality rate was about 100% among rats given streptozotocin on their fifth day of life. All rats receiving the drug on day 2 of life survived, and they showed a marked hyperglycemia, polyphagia, polydipsia and decreased body weight gain in addition to increased glycosylated hemoglobin rates and impaired results in the oral glucose tolerance test. Histopathological lesions of the pancreas as well as a decreased number of islets were significantly more frequent in rats receiving the drug subcutaneously on day 2, which confirms that streptozotocin administered subcutaneously produces greater damage. Conclusions: Subcutaneous injection of streptozotocin in a dose of 100 mg/kg of body weight in the second day after birth induced moderate diabetes in adult Wistar rats more effectively.
Downloads
References
Damasceno DC, Netto AO, Iessi IL, Gallego FQ, Corvino SB, Dallaqua B, et al . Streptozotocin-induced diabetes models: Pathophysiological mechanisms and fetal outcomes. Biomed Res Int. 2014;2014:819065. http://dx.doi.org/10.1155/2014/819065
Srinivasan K, Ramarao P. Animal models in type 2 diabetes research: An overview. Indian J Med Res. 2007;125:451-72.
Casado M. Modelos animales en el estudio de la diabetes. Av Diabetol. 2007;23:432-8.
Ashcroft FM, Rorsman P. Diabetes mellitus and the beta cell: The last ten years. Cell. 2012;148:1160-71. http://dx.doi.org/10.1016/j.cell.2012.02.010
Comité Ejecutivo de la Asociación Latinoamericana de Diabetes. Guías de diagnóstico, control y tratamiento de la diabetes mellitus tipo 2 con medicina basada en evidencia. Rev ALAD. 2013;1-142.
Arulmozhi DK, Veeranjaneyulu A, Bodhankar SL. Neonatal streptozotocin-induced rat model of type 2 diabetes mellitus: A glance. Indian J Pharmacol. 2004;36:217-21.
Jawerbaum A, White V. Animal models in diabetes and pregnancy. Endocr Rev. 2010;31:680-701. http://dx.doi.org/10.1210/er.2009-0038
Kiss AC, Woodside B, Sinzato YK, Bernardi MM, Kempinas WG, Anselmo-Franci JA, et al . Neonatally induced mild diabetes: Influence on development, behavior and reproductive function of female Wistar rats. Diabetol Metab Syndr. 2013;5:2-10. http://dx.doi.org/10.1186/1758-5996-5-61
Kiss AC, Lima PH, Sinzato YK, Takaku M, Takeno MA, Rudge MV, et al. Animal models for clinical and gestational diabetes: Maternal and fetal outcomes. Diabetol Metab Syndr. 2009;1:1-7. http://dx.doi.org/10.1186/1758-5996-1-21
Junod A, Lambert A, Stauffacher W, Renold A. Diabetogenic action of streptozotocin: Relationship of dose to metabolic response. J Clin Invest. 1969;48:2129-39.
Portha B, Picolon L, Rosselin G. Chemical diabetes in the adult rat as the spontaneous evolution of neonatal diabetes. Diabetologia. 1979;17:371-7.
Damasceno DC, Sinzato YK, Bueno A, Netto AO, Dallaqua B, Gallego FQ, et al . Mild diabetes models and their maternal-fetal repercussions. J Diabetes Res. 2013:2013;473575. http://dx.doi.org/10.1155/2013/473575
Sinzato YK, Lima PH, de Campos KE, Kiss AC, Rudge MV, Damasceno DC. Neonatally-induced diabetes: Lipid profile outcomes and oxidative stress status in adult rats. Rev Assoc Med Bras. 2009;55:384-8. http://dx.doi.org/10.1590/S0104-42302009000400010
Ernsberger P, Koletsky R. The glucose tolerance test as a laboratory tool with clinical implications. In: Chackrewarthy S, editor. Glucose tolerance. Rijeka: InTech; 2012. p. 3-14. http://dx.doi.org/10.5772/54785
Ishii Y, Ohta T, Sasase T. Non-obese type 2 diabetes animals models. In: Chackrewarthy S, editor. Glucose tolerance. Rijeka: InTech; 2012. p. 224-42. http://dx.doi.org/10.5772/54785
Weykamp C, John WG, Mosca A. A review of the challenge in measuring hemoglobin A1c. J Diabetes Sci Technol. 2009; 3:439-45. http://dx.doi.org/10.1177/193229680900300306
American Diabetes Association. Standards of medical care in diabetes-2014. Diabetes Care. 2014;37(Supl.1):s5-13. http://dx.doi.org/10.2337/dc14-S005
Rosales A. Determinación de hemoglobina glicada (HbA1) en ratas con diabetes inducida con estreptozotocina. México, DF: Universidad Autónoma Metropolitana Unidad Iztapalapa; 1994.
Del Toro M. Efecto de la cocarboxilasa sobre la glucemia en ratas Wistar inducidas a diabetes (tesis). Colima, México: Universidad de Colima; 2001.
Ciobotaru E. Spontaneous diabetes mellitus in animals. In: Oguntibeju OO, editor. Diabetes mellitus - Insights and perspectives. Rijeka: InTech; 2013. p. 271-96. http://dx.doi.org/10.5772/48170
Xuesong X, Zhu W, Yuming Z, Lihua B, Yuexin Y. Dosage of streptozocin in inducing rat model of type 2 diabetes mellitus. Wei Sheng Yan Jiu. 2010;39:138-42.
Ahimadi S, Karimian SM, Sotoudeh M, Bahadori M. Histological and immunohistochemical study of pancreatic islet beta cells of diabetic rats treated with oral vanadyl sulphate. MJIRI. 2002;16:173-8.
Alimohammadi S, Hobbenaghi R, Javanbakht J, Kheradmand D, Mortezaee R, Tavakoli R, et al . Protective and antidiabetic effects of extract from Nigella sativa on blood glucose concentrations against streptozotocin (STZ)-induced diabetic in rats: An experimental study with histopathological evaluation. Diagn Pathol. 2013;8:137-44. http://dx.doi.org/10.1186/1746-1596-8-137
Abunasef SK, Amin HA, Abdel-Hamid GA. A histological and immunohistochemical study of beta cells in streptozotocin diabetic rats treated with caffeine. Folia Histochem Cytobiol. 2014;52:42-50. http://dx.doi.org/10.5603/FHC.2014.0005
Mir SH, Baqui A, Bhagat RC, Darzi MM, Shah AW. Biochemical and histomorphological study of streptozotocin-induced diabetes mellitus in rabbits. Pak J Nut. 2008; 7:359-64.
Cubillos V, López C, Alberdi A. Estudio histopatológico e inmunohistoquímico de páncreas en perros diabéticos inducidos con aloxano. Arch Med Vet. 2008;40:169-77.
Bequer L, Gómez T, Molina JL, López F, Gómez CL, Clapés S. Inducción de hiperglicemias moderadas en ratas Wistar por inoculación neonatal de estreptozotocina. ¿Inyección subcutánea o intraperitoneal? Rev Argent Endocrinol Metab. 2014;51:178-84.
Figueroa MC, Pérez IH, Mejía R. Caracterización de un modelo de diabetes tipo 2 en ratas Wistar hembra. Rev MVZ Córdoba. 2013;18:3699-707.
Bonner-Weir S, Trent DF, Honey RN, Weir GC. Responses of neonatal rat islets to streptozotocin: Limited beta -cell regeneration and hyperglycemia. Diabetes. 1981;30:64-9. http://dx.doi.org/10.2337/diab.30.1.64
Deeds MC, Anderson JM, Armstrong AS, Gastineau DA, Hiddinga HJ, Jahangir A, et al . Single dose streptozotocin-induced diabetes: Considerations for study design in islet transplantation models. Lab Anim. 2011;45:131-40. http://dx.doi.org/10.1258/la.2010.010090
Some similar items:
- Juan Carlos Miguel, Ariana Erazo, Fernanda Beduino, Juan Carlos Picena, María Isabel Luciano, Gustavo Pizzuti, María Cristina Tarrés, Silvana Montenegro, Stella Maris Martínez, Chronic bronchial dilatations in different colonies of laboratory rats , Biomedica: Vol. 22 No. 2 (2002)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























