Trasplante haploidéntico de células progenitoras hematopoyéticas con ciclofosfamida después del trasplante en errores innatos de la inmunidad: experiencia en un centro de referencia en Colombia
Resumen
Introducción. Los errores innatos de la inmunidad son enfermedades raras causadas por más de 400 mutaciones genéticas que afectan al sistema inmunológico y aumentan la susceptibilidad a infecciones, la autoinmunidad y las neoplasias malignas. El trasplante de progenitores hematopoyéticos es una opción curativa para algunos errores innatos de la inmunidad y, específicamente, el trasplante haploidéntico es una alternativa viable en ausencia de un donante idéntico.
Objetivo. Determinar la supervivencia, utilidad del monitoreo semanal del quimerismo, reconstitución inmunológica y complicaciones de los pacientes con errores innatos de la inmunidad sometidos a trasplante haploidéntico de progenitores hematopoyéticos en un centro de referencia en Colombia.
Materiales y métodos. Se realizó un estudio observacional, retrospectivo, de serie de casos, con pacientes pediátricos sometidos a trasplante haploidéntico de progenitores hematopoyéticos, usando ciclofosfamida después del trasplante y haciendo seguimiento semanal de quimerismo. Se analizó la supervivencia con el método de Kaplan-Meier.
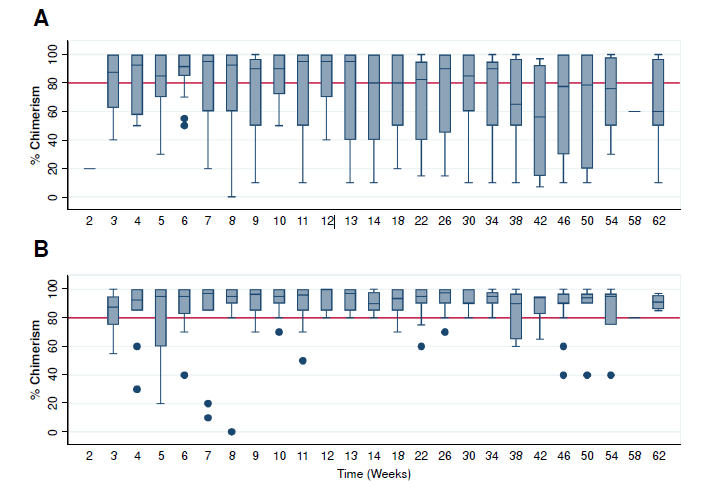
Resultados. Se incluyeron 16 pacientes con trasplante de donante familiar haploidéntico. El diagnóstico más frecuente fue inmunodeficiencia combinada grave (n = 5). Once de diecisiete recibieron un esquema de acondicionamiento no mieloablativo. La enfermedad aguda de injerto contra huésped se desarrolló en 12 de 16 pacientes y en 3 fue de grado III-IV. Las infecciones posteriores al trasplante afectaron a 14 pacientes, con predominio de agentes bacterianos. La mediana de quimerismo de células T fue mayor al 80 % durante el seguimiento. La reconstitución de linfocitos B y T se logró en más de 11 pacientes La supervivencia global a cinco años fue del 81 % y, a 100 días, fue del 91 %.
Conclusión. El trasplante haploidéntico de progenitores hematopoyéticos utilizando ciclofosfamida postrasplante, es una alternativa factible en pacientes con errores innatos de la inmunidad en ausencia de un donante idéntico. El monitoreo seriado del quimerismo es útil para el seguimiento del injerto.
Descargas
Referencias bibliográficas
Tangye SG, Al-Herz W, Bousfiha A, Chatila T, Cunningham-Rundles C, Etzioni A, et al. Human inborn errors of immunity: 2019 update on the classification from the International Union of Immunological Societies Expert Committee. J Clin Immunol. 2020;40:24-64. https://doi.org/10.1007/s10875-019-00737-x
Bousfiha A, Moundir A, Tangye SG, Picard C, Jeddane L, Al-Herz W, et al. The 2022 Update of IUIS phenotypical classification for human inborn errors of immunity. J Clin Immunol. 2022;42:1508-20.
Leiva LE, Bezrodnik L, Oleastro M, Condino-Neto A, Costa-Carvalho BT, Sevciovic Grumach A, et al. Primary immunodeficiency diseases in Latin America: Proceedings of the Second Latin American Society for Immunodeficiencies (LASID) Advisory Board. Allergol Immunopathol (Madr). 2011;39:106-10. https://doi.org/10.1007/s10875-019-00737-x
Vásquez E, Villada F, Orrego J, Franco J. Espectro de las inmunodeficiencias primarias en Colombia: reporte del Centro Nacional de Referencia Jeffrey Modell para diagnóstico e investigación en inmunodeficiencias primarias (CJM-UDEA). Iatreia. 2013;26(Suppl.3):S43. https://doi.org/10.17533/udea.iatreia.15760
Mccurdy SR, Luznik L. How we perform haploidentical stem cell transplantation with posttransplant cyclophosphamide. Blood. 2019;134:1802-10. https://doi.org/10.1182/blood.2019001323
Gyurkocza B, Sandmaier BM. Conditioning regimens for hematopoietic cell transplantation: One size does not fit all. Blood. 2014;124:344-53. https://doi.org/10.1182/blood-2014-02-514778
Yanagimachi M, Kato K, Iguchi A, Sasaki K, Kiyotani C, Koh K, et al. Hematopoietic cell transplantation for chronic granulomatous disease in Japan. Front Immunol. 2020;11:1617. https://doi.org/10.3389/fimmu.2020.01617
Laberko A, Gennery AR. Clinical considerations in the hematopoietic stem cell transplant management of primary immunodeficiencies. Expert Rev Clin Immunol. 2018;14:297-306. https://doi.org/10.1080/1744666X.2018.1459189
Pérez G, Olaya V, Del Campo M, Gaytán M, Mújica G, Juárez N, et al. Reconstitución inmunológica en niños receptores de trasplante de células progenitoras hematopoyéticas. Alerg Asma Inmunol Pediatr. 2012;21:72-9.
Rifón JJ. Trasplante de progenitores hemopoyéticos. An Sist Sanit Navar. 2006:29(Suppl.2):137-51.
Cruz CH, Carnot Uria J, Perurena JM, Llauger KC, Pardo I, Hospital R, et al. El trasplante alogénico haploidéntico: un traje hecho a la medida de nuestras condiciones y necesidades. Rev Cubana Med. 2017;56.
Trujillo ÁM, Karduss AJ, Suárez G, Pérez R, Ruiz G, Cardona A, et al. Haploidentical hematopoietic stem cell transplantation with post-transplantation cyclophosphamide in children with high-risk leukemia using a reduced-intensity conditioning regimen and peripheral blood as the stem cell source. Transplant Cell Ther. 2021;27:427.e1-427.e7. https://doi.org/10.1016/j.jtct.2021.02.010
Fernandes JF, Nichele S, Arcuri LJ, Ribeiro L, Zamperlini-Netto G, Loth G, et al. Outcomes after haploidentical stem cell transplantation with post-transplantation cyclophosphamide in patients with primary immunodeficiency diseases. Biol Blood Marrow Transplant. 2020;26:1923-9. https://doi.org/10.1016/j.bbmt.2020.07.003
Jones RJ. Haploidentical transplantation: Repurposing cyclophosphamide. Biol Blood Marrow Transplant. 2012;18:1771-2. https://doi.org/10.1016/j.bbmt.2012.09.005
Faraci M, Bagnasco F, Leoni M, Giardino S, Terranova P, Subissi L, et al. Evaluation of chimerism dynamics after allogeneic hematopoietic stem cell transplantation in children with nonmalignant diseases. Biol Blood Marrow Transplant. 2018;24:1088-93. https://doi.org/10.1016/j.bbmt.2017.12.801
Olaya M, Franco A, Chaparro M, Estupiñán M, Aristizábal D, Builes-Restrepo N, et al. Hematopoietic stem cell transplantation in children with inborn errors of immunity: A multicenter experience in Colombia. J Clin Immunol. 2020;40:1116-23. https://doi.org/10.1007/s10875-020-00856-w
Kuhns DB, Alvord WG, Heller T, Feld JJ, Pike KM, Marciano BE, et al. Residual NADPH oxidase and survival in chronic granulomatous disease. N Engl J Med. 2010;363:2600-10. https://doi.org/10.1056/NEJMoa1007097
Conley ME, Notarangelo LD, Etzioni A. Diagnostic criteria for primary immunodeficiencies. Representing PAGID (Pan-American Group for Immunodeficiency) and ESID (European Society for Immunodeficiencies). Clin Immunol. 1999;93:190-7. https://doi.org/10.1006/clim.1999.4799
Jordan MB, Allen CE, Weitzman S, Filipovich AH, McClain KL. How I treat hemophagocytic lymphohistiocytosis. Blood. 2011;118:4041-52. https://doi.org/10.1182/blood-2011-03-278127
Seidel MG, Kindle G, Gathmann B, Quinti I, Buckland M, van Montfrans J, et al. The European Society for Immunodeficiencies (ESID) registry working definitions for the clinical diagnosis of inborn errors of immunity. J Allergy Clin Immunol Pract. 2019;7:1763-70. https://doi.org/10.1016/j.jaip.2019.02.004
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: Working definitions. Biol Blood Marrow Transplant. 2009;15:1628-33. https://doi.org/10.1016/j.bbmt.2009.07.004
Lum SH, Hoenig M, Gennery AR, Slatter MA. Conditioning regimens for hematopoietic cell transplantation in primary immunodeficiency. Curr Allergy Asthma Rep. 2019;19:52. https://doi.org/10.1007/s11882-019-0883-1
Al-Homsi AS, Roy TS, Cole K, Feng Y, Duffner U. Post-transplant high-dose cyclophosphamide for the prevention of graft-versus-host disease. Biol Blood Marrow Transplant. 2015;21:604-11. https://doi.org/10.1016/j.bbmt.2014.08.014
Kanda Y, Hyo R, Yamashita T, Fujimaki K, Oshima K, Onoda M, et al. Effect of blood cyclosporine concentration on the outcome of hematopoietic stem cell transplantation from an HLA-matched sibling donor. Am J Hematol. 200;81:838-44. https://doi.org/10.1002/ajh.20710
Rowlings PA, Przepiorka D, Klein JP, Gale RP, Passweg JR, Henslee-Downey J, et al. IBMTR Severity Index for grading acute graft-versus-host disease: Retrospective comparison with Glucksberg grade. Br J Haematol. 1997;97:855-64. https://doi.org/10.1046/j.1365-2141.1997.1112925.x
Carreras E, Dufour C, Mohty M, Kröger N. The EBMT Handbook: Hematopoietic stem cell transplantation and cellular therapies [internet]. 7th edition. Cham (CH): Springer; 2019. https://doi.org/10.1007/978-3-030-02278-5
Rujkijyanont P, Morris C, Kang G, Gan K, Hartford C, Triplett B, et al. Risk-adapted donor lymphocyte infusion based on chimerism and donor source in pediatric leukemia. Blood Cancer J. 2013;3:e137. https://doi.org/10.1038/bcj.2013.39
Umeda K, Adachi S, Tanaka S, Miki M, Okada K, Hashii Y, et al. Comparison of second transplantation and donor lymphocyte infusion for donor mixed chimerism after allogeneic stem cell transplantation for nonmalignant diseases. Pediatr Blood Cancer. 2016;63:2221-9. https://doi.org/10.1002/pbc.26141
Kim HT, Armand P. Clinical endpoints in allogeneic hematopoietic stem cell transplantation studies: The cost of freedom. Biol Blood Marrow Transplant. 2013;19:860-6. https://doi.org/10.1016/j.bbmt.2013.01.003
Comans-Bitter WM, De Groot R, van Den Beemd R, Neijens HJ, Hop WC, Groeneveld K, et al. Immunophenotyping of blood lymphocytes in childhood: Reference values for lymphocyte subpopulations. J Pediatr. 1997;130:388-93. https://doi.org/10.1016/s0022-3476(97)70200-2
Hladun R, Badell I, González M, Martínez AM, Sánchez de Toledo J, Olivé MT, et al. Survival analysis of hematopoietic stem cell transplantation in children with primary immunodeficiency in Spain. An Pediatr (Barc). 2015;82:62-7. https://doi.org/10.1016/j.anpedi.2014.04.014
Ariffin H, Ab Rahman S, Jawin V, Foo JC, Amram NF, Mahmood NM, et al. Haematopoietic stem cell transplantation for inborn errors of immunity: 25-year experience from University of Malaya Medical Centre, Malaysia. J Paediatr Child Health. 2020;56:379-83. https://doi.org/10.1111/jpc.14621
Gennery AR, Slatter MA, Grandin L, Taupin P, Cant AJ, Veys P, et al. Transplantation of hematopoietic stem cells and long-term survival for primary immunodeficiencies in Europe: Entering a new century, do we do better? J Allergy Clin Immunol. 2010;126:602-10.e1-11. https://doi.org/10.1016/j.jaci.2010.06.015
Fernandes JF, Nichele S, Daudt LE, Tavares RB, Seber A, Kerbauy FR, et al. Transplantation of hematopoietic stem cells for primary immunodeficiencies in Brazil: Challenges in treating rare diseases in developing countries. J Clin Immunol. 2018;38:917-26. https://doi.org/10.1007/s10875-018-0564-1
Yi ES, Choi YB, Lee NH, Lee JW, Sung KW, Koo HH, et al. Allogeneic hematopoietic cell transplantation in patients with primary immunodeficiencies in Korea: Eleven-year experience in a single center. J Clin Immunol. 2018;38:757-66. https://doi.org/10.1007/s10875-018-0542-7
Heimall J, Logan BR, Cowan MJ, Notarangelo LD, Griffith LM, Puck JM, et al. Immune reconstitution and survival of 100 SCID patients post-hematopoietic cell transplant: A PIDTC natural history study. Blood. 2017;130:2718-27. https://doi.org/10.1182/blood-2017-05-781849
Klein OR, Chen AR, Gamper C, Loeb D, Zambidis E, Llosa N, et al. Alternative-donor hematopoietic stem cell transplantation with post-transplantation cyclophosphamide for nonmalignant disorders. Biol Blood Marrow Transplant. 2016;22:895-901. https://doi.org/10.1016/j.bbmt.2016.02.001
Neven B, Diana JS, Castelle M, Magnani A, Rosain J, Touzot F, et al. Haploidentical hematopoietic stem cell transplantation with post-transplant cyclophosphamide for primary immunodeficiencies and inherited disorders in children. Biol Blood Marrow Transplant. 2019;25:1363-73. https://doi.org/10.1016/j.bbmt.2019.03.009
Díaz de Heredia C, Ortega JJ, Díaz MA, Olivé T, Badell I, González-Vicent M, et al. Unrelated cord blood transplantation for severe combined immunodeficiency and other primary immunodeficiencies. Bone Marrow Transplant. 2008;41:627-33. https://doi.org/1010.1038/sj.bmt.170594
Scarselli A, Di Cesare S, Capponi C, Cascioli S, Romiti ML, Di Matteo G, et al. Longitudinal evaluation of immune reconstitution and B-cell function after hematopoietic cell transplantation for primary immunodeficiency. J Clin Immunol. 2015;35:373-83. https://doi.org/10.1007/s10875-015-0154-4
Uppuluri R, Jayaraman D, Sivasankaran M, Patel S, Vellaichamy Swaminathan V, Vaidhyanathan L, et al. Hematopoietic stem cell transplantation for primary immunodeficiency disorders: Experience from a referral center in India. Indian Pediatr. 2018;55:661-4.
Chang YJ, Xu LP, Wang Y, Zhang XH, Chen H, Chen YH, et al. Effects of Low-dose glucocorticoid prophylaxis on chronic graft-versus-host disease and graft-versus-host disease-free, relapse-free survival after haploidentical transplantation: Long-term follow-up of a controlled, randomized open-label trial. Biol Blood Marrow Transplant. 2019;25:529-37. https://doi.org/10.1016/j.bbmt.2018.11.020
Patiroglu T, Akar H, Ozdemir A, Karakukcu M. Hematopoietic stem cell transplant for primary immunodeficiency diseases: A single-center experience. Exp Clin Transplant. 2017;15:337-43. https://doi.org/10.6002/ect.2015.0233
Haddad E, Logan BR, Griffith LM, Buckley RH, Parrott RE, Prockop SE, et al. SCID genotype and 6-month posttransplant CD4 count predict survival and immune recovery. Blood. 2018;132:1737-49. https://doi.org/10.1182/blood-2018-03-840702
Thakar MS, Logan BR, Puck JM, Dunn EA, Buckley RH, Cowan MJ, et al. Measuring the effect of newborn screening on survival after haematopoietic cell transplantation for severe combined immunodeficiency: A 36-year longitudinal study from the Primary Immune Deficiency Treatment Consortium. Lancet. 2023;402:129-40. https://doi.org/10.1016/S0140-6736(23)00731-6
Algunos artículos similares:
- Paula Ximena Pavía, Nubia Lucía Roa, Ana María Uribe, Concepción Judith Puerta, Seguimiento de paciente con enfermedad de Chagas y trasplante de corazón mediante las PCR S35-S36 y TcH2AF-R , Biomédica: Vol. 31 Núm. 2 (2011)
- John F. Arboleda, Luis F. García, Cristiam M. Álvarez, Papel de las células dendríticas tolerogénicas ILT3+/ILT4+ en la respuesta inmunitaria a trasplante de órganos y tejidos , Biomédica: Vol. 31 Núm. 2 (2011)
- Mario García, Liliana Alejandra Chicaíza, Hoover Quitián, Adriana Linares, Óscar Ramírez, Costo-efectividad de los tratamientos de consolidación para la leucemia mieloide aguda en niños en riesgo alto en el sistema de salud colombiano , Biomédica: Vol. 35 Núm. 4 (2015)
- Yazmin Rocío Arias, Karime Osorio-Arango, Brayan Bayona, Guadalupe Ercilla, Mauricio Beltrán-Durán, Determinación del polimorfismo HLA -A, -B, -DRB1 en donantes de órganos en muerte encefálica representativos de la población general colombiana, 2007-2014 , Biomédica: Vol. 37 Núm. 2 (2017)
- Lina Echeverri-Toro, Andrés Arango, Sigifredo Ospina, Carlos Agudelo, Bacteriemia recurrente por Bordetella bronchiseptica en un paciente con trasplante de medula ósea , Biomédica: Vol. 35 Núm. 3 (2015)
- Fiona Xacur-García, Rodrigo Díaz-Novelo, Linnete Herrera-David, Paulina Moreno-Arjona, Nina Méndez-Domínguez, Manifestaciones clínicas y evolución a largo plazo de tres casos de rosácea ocular atendidos en un hospital de alta especialidad del sureste de México , Biomédica: Vol. 40 Núm. 3 (2020)
Derechos de autor 2024 Biomédica

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























