Seroprevalencia de HCoV-NL63 y HCoV-HKU1 y su relación con las características clínicas de pacientes con COVID-19 de Villavicencio, Colombia
Resumen
Introducción. Debido a la reactividad cruzada entre SARS-CoV-2 y los coronavirus humanos comunes, las infecciones previas con estos virus podrían contribuir a la protección cruzada serológica o celular contra la COVID-19 grave. Sin embargo, la inmunidad protectora puede no desarrollarse o la inmunidad preexistente podría generar COVID-19 grave.
Objetivo. Determinar la seroprevalencia de anticuerpos IgG frente a HCoV-NL63 y HCoVHKU1, y correlacionar su previa exposición con los signos de COVID-19 en pacientes de Villavicencio.
Materiales y métodos. Se realizó un estudio retrospectivo observacional analítico y transversal. Se utilizó la técnica ELISA para buscar anticuerpos IgG contra HCoV-NL3 y HCoV-HKU1 en pacientes con resultado positivo de RT-qPCR para SARS-CoV-2. Los pacientes se agruparon según los signos de COVID-19 en cuatro grupos: grupo 1:
asintomáticos (n = 23); grupo 2: hospitalizados (n = 24); grupo 3: unidad de cuidados intensivos (n = 24), y grupo 4: fallecidos (n = 22).
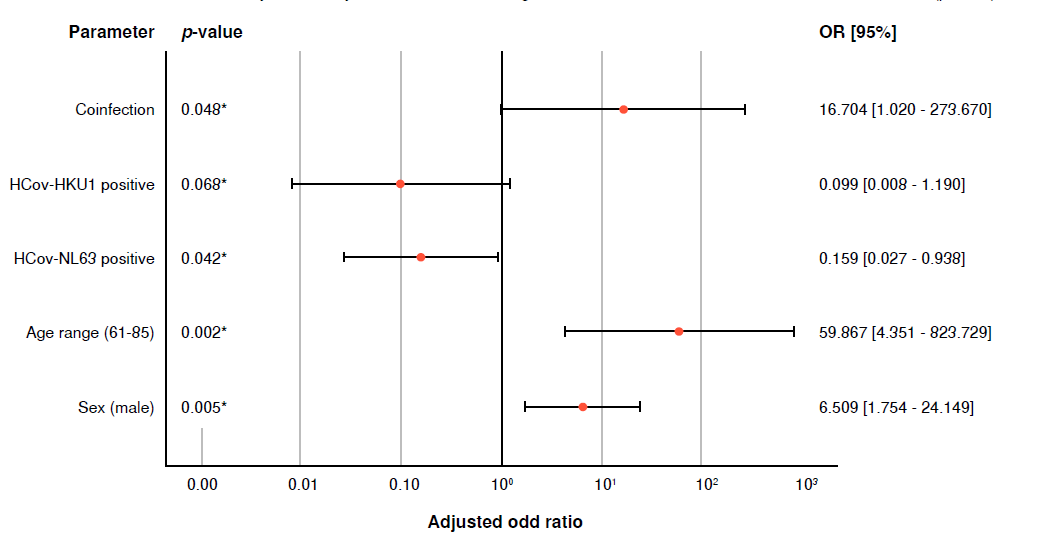
Resultados. La seroprevalencia general de IgG anti-HCoV fue de 74.2 % (n = 69; IC95%: 65,3-83,1), con 66,7 % de HCoV-NL63 (n = 62; IC95% :57,1-76,2) y 25,8 % de HCoV-HKU1 (n = 24; [IC95%:16,9-34,7). Según el análisis de las tablas de contingencia, la exposición previa a HCoV-NL63 se asoció con protección de una COVID-19 grave (p = 0,042; OR ajustado = 0,159; IC95%: 0,027-0,938) y la previa coinfección de HCoV-NL63 y HCoV-HKU1 se asoció con padecimiento de signos clínicos graves por COVID-19 (p = 0,048; OR ajustado = 16,704; IC95%: 1,020- 73,670).
Conclusión. Según la literatura revisada hasta la fecha, este es el primer estudio sobre la seroprevalencia de anticuerpos IgG de HCoV en Colombia y Latinoamérica. La exposición previa a HCoV-NL63 podría proteger contra la COVID-19 grave, mientras que los pacientes con coinfección subyacente de HCoV-NL63 y HCoV-HKU1 podrían resultar hospitalizados con signos graves de COVID-19.
Descargas
Referencias bibliográficas
Lai MM, Perlman S, Anderson LJ. Coronaviridae. In: Knipe DM, Howley PM, editors. Fields Virology. Philadelphia: Lippincott Williams & Wilkins; 2007. p. 1305-36.
Cui J, Li F, Shi ZL. Origin and evolution of pathogenic coronaviruses. Nat Rev Microbiol. 2019;17:181-92. https://doi.org/10.1038/s41579-018-0118-9
Drosten C, Günther S, Preiser W, van der Werf S, Brodt H, Becker S, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967-76. https://www.nejm.org/doi/10.1056/NEJMoa030747
van der Hoek L, Pyrc K, Jebbink MF, Vermeulen-Oost W, Berkhout RJ, Wolthers K, et al. Identification of a new human coronavirus. Nat Med. 2004;10:368-73. https://doi.org/10.1038/nm1024
Woo PC, Lau SK, Chu CM, Chan KH, Tsoi HW, Huang Y, et al. Characterization and complete genome sequence of a novel coronavirus, coronavirus HKU1, from patients with pneumonia. J Virol. 2005;79:884-95. https://doi.org/10.1128/jvi.79.2.884-895.2005
Chen B, Tian E-K, He B, Tian L, Han R, Wang S, et al. Overview of lethal human coronaviruses. Signal Transduc Target Ther. 2020;5:89.
https://doi.org/10.1038/s41392-020-0190-2
Phan T. Novel coronavirus: From discovery to clinical diagnostics. Infect Genet Evol. 2020;79:104211. https://doi.org/10.1016/j.meegid.2020.104211
World Health Organization. Director-General’s opening remarks at the media briefing on COVID-19 - 11 March 2020. New York: WHO Office at the United Nations; 2020. Accessed: March 7, 2023. Available at: https://www.who.int/director-general/speeches/detail/whodirector-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020
Johns Hopkins University. COVID-19 Dashboard. Maryland: Center for Systems Science and Engineering (CSSE); 2023. Accessed: July 7, 2023. Available at: https://coronavirus.jhu.edu/map.html
Guan W, Ni Z, Hu Y, Wen-Hua L, Chun-Quan O, He J, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-20. https://doi.org/10.1016/j.jemermed.2020.04.004
Greenberg SB. Update on rhinovirus and coronavirus infections. Semin Respir Crit Care Med. 2011;32:433-46. https://doi.org/10.1055/s-0031-1283283
Shah MM, Winn A, Dahl RM, Kniss KL, Silk BJ, Killerby M. Seasonality of common human coronaviruses, United States, 2014-2021. Emerg Infect Dis. 2022;28:1970-6. https://doi.org/10.3201/eid2810.220396
Gaunt ER, Hardie A, Claas EC, Simmonds P, Templeton KE. Epidemiology and clinical presentations of the four human coronaviruses 229E, HKU1, NL63, and OC43 detected over 3 years using a novel multiplex real-time PCR method. J Clin Microbiol. 2010;48:2940-7. https://doi.org/10.1128/JCM.00636-10
Chiu SS, Chan KH, Chu KW, Kwan SW, Guan Y, Poon LL, et al. Human coronavirus NL63 infection and other coronavirus infections in children hospitalized with acute respiratory disease in Hong Kong, China. Clin Infect Dis. 2005;40:1721-9. https://doi.org/10.1086/430301
Yu X, Lu R, Wang Z, Zhu N, Wang W, Druce J, et al. Etiology and clinical characterization of respiratory virus infections in adult patients attending an emergency department in Beijing. PLoS ONE. 2012;7:e32174. https://doi.org/10.1371/journal.pone.0032174
Reina J, López-Causapé C, Rojo-Molinero E, Rubio R. Características de las infecciones respiratorias agudas causadas por los coronavirus OC43, NL63 y 229E. Rev Clin Esp. 2014;214:499-504. https://doi.org/10.1016/j.rce.2014.05.020
Kanwar A, Selvaraju S, Esper F. Human coronavirus-HKU1 infection among adults in Cleveland, Ohio. Open Forum Infect Dis. 2017;4. https://doi.org/10.1093/ofid/ofx052
Cabeça TK, Carraro E, Watanabe A, Granato C, Bellei N. Infections with human coronaviruses NL63 and OC43 among hospitalised and outpatient individuals in São Paulo, Brazil. Mem Inst Oswaldo Cruz. 2012;107:693-4. https://doi.org/10.1590/s0074-02762012000500020
Grifoni A, Weiskopf D, Ramírez SI, Mateus J, Dan J, Moderbacher CR, et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell. 2020;181:1489-501. https://doi.org/10.1016/j.cell.2020.05.015
Tso FY, Lidenge SJ, Peña PB, Clegg A, Ngowind J, Mwaiselage J, et al. High prevalence of pre-existing serological cross-reactivity against severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) in Sub-Saharan Africa. Int J Infect Dis. 2021;102:577-83. https://doi.org/10.1016/j.ijid.2020.10.104
Mateus J, Grifoni A, Tarke A, Sidney J, Ramírez S, Dan J, et al. Selective and cross-reactive SARS-CoV-2 T cell epitopes in unexposed humans. Science. 2020;370:89-94. https://doi.org/10.1126/science.abd3871
Braun J, Loyal L, Frentsch M, Wendisch D, Georg P, Kurth F, et al. SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19. Nature. 2020;587:270-4. https://doi.org/10.1038/s41586-020-2598-9
Lesmes-Rodríguez LC, Lambarey H, Chetram A, Riou C, Wilkinson R, Joyimbana W, et al. Previous exposure to common coronavirus HCoV-NL63 is associated with reduced COVID-19 severity in patients from Cape Town, South Africa. Front Virol. 2023;3:1125448. https://doi.org/10.3389/fviro.2023.1125448
Reina J. Posible efecto del «pecado antigénico original» en la vacunación frente a las nuevas variantes del SARS-CoV-2. Rev Clin Esp 2022;222:91-2. https://doi.org/10.1016/j.rce.2021.05.003
Roncati L, Palmieri B. What about the original antigenic sin of the humans versus SARSCoV-2? Med Hypotheses. 2020;142:109824. https://doi.org/10.1016/j.mehy.2020.109824
Instituto Nacional de Salud. Distribución de casos por municipios COVID-19 en Colombia. Bogotá: INS; 2020. Accessed: July 27, 2023. Available at: https://www.ins.gov.co/Noticias/Paginas/Coronavirus.aspx
Dijkman R, Jebbink MF, Idrissi NBE, Pyrc K, Müller M, Kuijpers T, et al. Human coronavirus Nl63 and 229e seroconversion in children. J Clin Microbiol. 2008;46:2368-73. https://doi.org/10.1128/JCM.00533-08
Edridge AWD, Kaczorowska J, Hoste ACR, Bakker M, Klein M, Loens K, et al. Seasonal coronavirus protective immunity is short-lasting. Nat Med. 2020;26:1691-3. https://doi.org/10.1038/s41591-020-1083-1
World Medical Association. Declaration of Helsinki. Ethical principles for medical research involving human subjects. Bull World Health Organ. 2001;79:373-4.
Ministerio de Salud de Colombia. Resolución 008430, por la cual se establecen las normas científicas, técnicas y administrativas para la administración en salud. Bogotá: Ministerio de Salud; 1993. Accessed: July 6, 2023. Available at: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/DE/DIJ/RESOLUCION-8430-DE-1993.PDF
Severance E, Bossis I, Dickerson F, Stallings C, Origoni A, Sullens A, et al. Development of a nucleocapsid-based human coronavirus immunoassay and estimates of individuals exposed to coronavirus in a U.S. Metropolitan Population. Clin Vaccine Immunol. 2021;15:1805-10. https://doi.org/10.1128/CVI.00124-08
Khan T, Rahman M, Ali FA, Huang S, Ata M, Zhang Q, et al. Distinct antibody repertoires against endemic human coronaviruses in children and adults. JCI Insight. 2021;6:e144499. https://doi.org/10.1172/jci.insight.144499
Gorse GJ, Patel GB, Vitale JN, O’Connor T. Prevalence of antibodies to four human coronaviruses is lower in nasal secretions than in serum. Clin Vaccine Immunol. 2010;17:1875-80. https://doi.org/10.1128/CVI.00278-10
Galipeau Y, Siragam V, Laroche G, Marion E, Greig Y, McGuinty M, et al. Relative ratios of human seasonal coronavirus antibodies predict the efficiency of cross-neutralization of SARS-CoV-2 spike binding to ACE2. EBioMedicine. 2021;74:103700. https://doi.org/10.1016/j.ebiom.2021.103700
Gao X, Zhou H, Wu C, Xiao Y, Ren L, Paranhos-Baccalà G, et al. Antibody against nucleocapsid protein predicts susceptibility to human coronavirus infection. J Infect. 2015;71:599-602. https://doi.org/10.1016/j.jinf.2015.07.002
Sagar M, Reifler K, Rossi M, Miller N, Sinha P, White L, et al. Recent endemic coronavirus infection is associated with less-severe COVID-19. J Clin Invest. 2021;131:e143380. https://doi.org/10.1172/JCI143380
Dugas M, Grote-Westrick T, Merle U, Fontenay M, Kremer A, Hanses F, et al. Lack of antibodies against seasonal coronavirus OC43 nucleocapsid protein identifies patients at risk of critical COVID-19. J Clin Virol. 2021;139:104847. https://doi.org/10.1016/j.jcv.2021.104847
Gouma S, Weirick ME, Bolton MJ, Arévalo C, Goodwin E, Anderson E, et al. Health care worker seromonitoring reveals complex relationships between common coronavirus antibodies and COVID-19 symptom duration. JCI Insight. 2021;6:e150449. https://doi.org/10.1172/jci.insight.150449
Lavell AH, Sikkens JJ, Edridge AW, Gils M, Hoek L, Oomen M, et al. Recent Infection with HCoV-OC43 may be associated with protection against SARS-CoV-2 Infection. iScience. 2022;25:105105. https://doi.org/10.1016/j.isci.2022.105105
Liu D, Chen C, Chen D, Zhu A, Li F, Zhuang Z, et al. Mouse models susceptible to HCoV-229E and HCoV-NL63 and cross protection from challenge with SARS-CoV-2. Proc Natl Acad Sci USA. 2023;120:e2202820120. https://doi.org/10.1073/pnas.2202820120
Rees E, Waterlow N. Centre for the Mathematical Modelling of Infectious Diseases COVID-19 Working group, Lowe R, Adam J. Estimating the duration of seropositivity of human seasonal coronaviruses using seroprevalence studies. Wellcome Open Res. 2021;6:138. https://doi.org/10.12688/wellcomeopenres.16701.3
Anderson EM, Goodwin EC, Verma A, Arévalo C, Bolton M, Weirick M, et al. Seasonal human coronavirus antibodies are boosted upon SARS-CoV-2 infection but not associated with protection. Cell. 2021;184:1858-64.e10. https://doi.org/10.1016/j.cell.2021.02.010
Imai K, Matsuoka M, Tabata S, Kitagawac Y, Ikeda M, Kubota K, et al. Cross-reactive humoral immune responses against seasonal human coronaviruses in COVID-19 patients with different disease severities. Int J Infect Dis. 2021;111:68-75. https://doi.org/10.1016/j.ijid.2021.08.026
Sayama Y, Okamoto M, Saito M, Obata M, Tamaki R, Joboco CD, et al. Seroprevalence of four endemic human coronaviruses and, reactivity and neutralization capability against SARS-CoV-2 among children in the Philippines. Sci Rep. 2023;13:2310. https://doi.org/10.1038/s41598-023-29072-3
Guo L, Wang Y, Kang L, Hu Y, Wang L. Joboco DC, et al. Cross-reactive antibody against human coronavirus Oc43 spike protein correlates with disease severity in COVID-19 patients: A retrospective study. Emerg Microbes Infect. 2021;10:664-76. https://doi.org/10.1080/22221751.2021.1905488
McNaughton AL, Paton RS, Edmans M, Youngs J, Wellens J. Phalora P, et al. Fatal COVID-19 outcomes are associated with an antibody response targeting epitopes shared with endemic coronaviruses. JCI Insight. 2022;7:e156372. https://doi.org/10.1172/jci.insight.156372
Vatti A, Monsalve DM, Pacheco Y, Chang C, Anaya J, Gershwin ME. Original antigenic sin: A comprehensive review. J Autoimmun. 2017;83:12-21. https://doi.org/10.1016/j.jaut.2017.04.008
Steiner S, Sotzny F, Bauer S, Na I, Schmueck M. Corman V, et al. HCoV and SARS-CoV-2 cross-reactive T cells in COVID patients. Front Immunol. 2020;11:607918. https://doi.org/10.3389/fimmu.2020.607918
Lin C-Y, Wolf J, Brice DC, Sun Y, Locke M. Cherry S, et al. Pre-existing humoral immunity to human common cold coronaviruses negatively impacts the protective SARS-CoV-2 antibody response. Cell Host Microbe. 2022;30:83-96.e4. https://doi.org/10.1016/j.chom.2021.12.005
Odendahl M, Endler I, Haubold B, Rodionov R, Bornstein S, Tonn T, et al. SARS-CoV-2-specific humoral immunity in convalescent patients with mild COVID-19 is supported by CD4+ T-cell help and negatively correlated with alphacoronavirus-specific antibody titer. Immunol Lett. 2022;251-252:38-46. https://doi.org/10.1016/j.imlet.2022.09.007
Ma J, Dushoff J, Earn D. Age-specific mortality risk from pandemic influenza. J Theor Biol. 2011;288:29-34. https://doi.org/10.1016/j.jtbi.2011.08.003
Ajmeriya S, Kumar A, Karmakar S, Rana S, Singh H. Neutralizing antibodies and antibodydependent enhancement in COVID-19: A perspective. J Indian Inst Sci. 2022;102:671-87. https://doi.org/10.1007/s41745-021-00268-8
Vahidy F, Pan AP, Ahnstedt H, Munshi Y, Choi H. Tiruneh Y, et al. Sex differences in susceptibility, severity, and outcomes of coronavirus disease 2019: Cross-sectional analysis from a diverse US metropolitan area. PLoS ONE. 2021;16:e0245556. https://doi.org/10.1371/journal.pone.0245556
Kragholm K, Andersen MP, Gerds TA, Butt J, Ostergaard L, Polcwiartek C, et al. Association between male sex and outcomes of coronavirus disease 2019 (COVID-19)-a Danish nationwide, register-based study. Clin Infect Dis. 2021;73:e4025-30. https://doi.org/10.1093/cid/ciaa924
Zhang J, Dong X, Liu GH, Gao Y. Risk and protective factors for COVID-19 morbidity, severity, and mortality. Clinic Rev Allerg Immunol. 2023;64:90-107. https://doi.org/10.1007/s12016-022-08921-5
Tizazu AM, Mengist HM, Demeke G. Aging, inflammaging and immunosenescence as risk factors of severe COVID-19. Immun Ageing. 2022;19:53. https://doi.org/10.1186/s12979-022-00309-5
Ho FK, Petermann-Rocha F, Gray SR, Jani B, Katikired S, Niedzwiedz C, et al. Is older age associated with COVID-19 mortality in the absence of other risk factors? General population cohort study of 470,034 participants. PLoS ONE. 2020;15:e0241824. https://doi.org/10.1371/journal.pone.0241824
Algunos artículos similares:
- José Moreno-Montoya, El desafío de comunicar y controlar la epidemia por coronavirus , Biomédica: Vol. 40 Núm. 1 (2020)
- Juan Pimentel, Neil Andersson, Cloroquina y sus derivados en el manejo de la COVID-19: una revisión sistemática exploratoria , Biomédica: Vol. 40 Núm. Supl. 2 (2020): SARS-CoV-2 y COVID-19
- Daniele Piovani , Georgios K. Nikolopoulos, Stefanos Bonovas, Escollos y peligros del análisis de supervivencia: el caso de los datos de COVID-19 , Biomédica: Vol. 41 Núm. Sp. 2 (2021): Octubre, Infecciones bacterianas y virales
Derechos de autor 2024 Biomédica

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























