Desafío diagnóstico: PCR anidada y en tiempo real frente a un sistema de puntuación en individuos con gran riesgo de neumonía por Pneumocystis jirovecii
Resumen
Introducción. Pneumocystis jirovecii es un hongo oportunista que afecta principalmente a personas con HIV (recuento de CD4 menor de 200 células/ml) y a otros pacientes inmunosuprimidos. Como P. jirovecii no crece en los medios micológicos de rutina, el diagnóstico de neumonía por P. jirovecii se basa en la evidencia presente en muestra respiratorias.
Objetivos. Asociar los resultados de la inmunofluorescencia directa y los de dos métodos moleculares con un puntaje para predecir la neumonía causada por P. jirovecii en pacientes con sida.
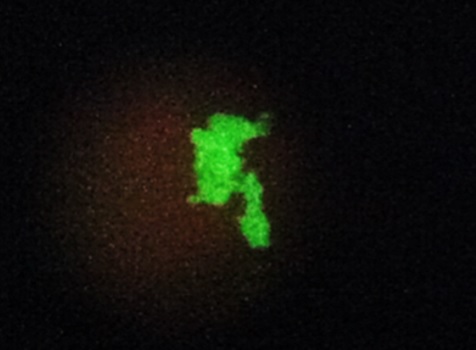
Materiales y métodos. Se realizó un estudio prospectivo de 40 pacientes. Se recolectó una muestra respiratoria antes del inicio de tratamiento y se sometió a una prueba de inmunofluorescencia directa con el kit Merifluor, una PCR anidada para la amplificación de la subunidad larga del ribosoma mitocondrial y una PCR en tiempo real usando el kit VIASURE.
Resultados. Estas tres técnicas evidenciaron la presencia de P. jirovecii en 6, 12 y 15 muestras, respectivamente. Todas las muestras positivas por inmunofluorescencia directa fueron positivas en la PCR anidada y todas las muestras positivas en la PCR anidada amplificaron por PCR en tiempo real. Se encontró una asociación estadística entre los valores de la neumonía causada por P. jirovecii y los métodos moleculares. Dos pacientes con diagnóstico temprano respondieron satisfactoriamente al tratamiento.
Conclusión. Se recomiendan los métodos moleculares, especialmente la PCR en tiempo real, para el diagnóstico temprano de neumonía causada por P. jirovecii en pacientes con sida.
Descargas
Referencias bibliográficas
Hoving JC, Brown GD, Gómez BL, Govender NP, Limper AH, May RC, et al. AIDS-related mycoses: Updated progress and future priorities. Trends Microbiol. 2020;28:425-8. https://doi.org/10.1016/j.tim.2020.01.009
Limper AH, Adenis A, Le T, Harrison TS. Fungal infections in HIV/AIDS. Lancet Infect Dis. 2017;17:e334-43. https://doi.org/10.1016/S1473-3099(17)30303-1
Meléndez-Rivera JG, Ciofoaia GA. Pneumocystis jirovecii prophylaxis. Treasure Island: StatPearls; 2021.
Thomas CF Jr., Limper AH. Pneumocystis pneumonia. N Engl J Med. 2004;350:2487-98. https://doi.org/10.1056/NEJMra032588
Peters SE, Wakefield AE, Sinclair K, Millard PR, Hopkin JM. A search for Pneumocystis carinii in post-mortem lungs by DNA amplification. J Pathol. 1992;166:195-8. https://doi.org/10.1002/path.1711660217
Truong J, Ashurst JV. Pneumocystis jirovecii pneumonia. Treasure Island: StatPearls; 2021.
Phair J, Muñoz A, Detels R, Kaslow R, Rinaldo C, Saah A. The risk of Pneumocystis carinii pneumonia among men infected with human immunodeficiency virus type 1. Multicenter AIDS Cohort Study Group. N Engl J Med. 1990;322:161-5. https://doi.org/10.1056/NEJM199001183220304
White PL, Price JS, Backx M. Pneumocystis jirovecii pneumonia: epidemiology, clinical manifestation and diagnosis. Curr Fungal Infect R. 2019;13:260‑73. https://doi.org/10.1007/s12281-019-00349-3
Schildgen V, Mai S, Khalfaoui S, Lusebrink J, Pieper M, Tillmann RL, et al. Pneumocystis jirovecii can be productively cultured in differentiated CuFi-8 airway cells. mBio. 2014;5:e01186-14. https://doi.org/10.1128/mBio.01186-14
Carmona EM, Limper AH. Update on the diagnosis and treatment of Pneumocystis pneumonia. Ther Adv Respir Dis. 2011;5:41-59. https://doi.org/10.1177/1753465810380102
Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-80. https://doi.org/10.1128/CMR.00053-10
Gingerich AD, Norris KA, Mousa JJ. Pneumocystis pneumonia: immunity, vaccines, and treatments. Pathogens. 2021;10. https://doi.org/10.3390/pathogens10020236
Daly K, Koch J, Respaldiza N, de la Horra C, Montes-Cano MA, Medrano FJ, et al. Geographical variation in serological responses to recombinant Pneumocystis jirovecii major surface glycoprotein antigens. Clin Microbiol Infect. 2009;15:937-42. https://doi.org/10.1111/j.1469-0691.2009.02716.x
Sabbagh W, Darwich NS. Pneumocystis jirovecii pneumonia and newly diagnosed human immunodeficiency virus (AIDS) in a 63-year-old woman. Am J Case Rep. 2018;19:927-31. https://doi.org/10.12659/AJCR.909612
Langevin B, Saleh M. Radiological presentation of Pneumocystis jirovecii pneumonia mimicking bacterial pneumonia. BMJ Case Rep. 2016;2016:bcr2016215207. https://doi.org/10.1136/bcr-2016-215207
Waks AG, Tolaney SM, Galar A, Arnaout A, Porter JB, Marty FM, et al. Pneumocystis jirovecii pneumonia (PCP) in patients receiving neoadjuvant and adjuvant anthracyclinebased chemotherapy for breast cancer: incidence and risk factors. Breast Cancer Res Treat. 2015;154:359-67. https://doi.org/10.1007/s10549-015-3573-2
Pandey S, Ghosh S, Halder P, Pal D, Modak DC, Guha SK. Pulmonary tuberculosis and Pneumocystis jirovecii concurrent pneumonia in HIV infected patients at a resource limited setting in Eastern India: A case series. Indian J Tuberc. 2020;67:378-82. https://doi.org/10.1016/j.ijtb.2019.08.003
Wahab A, Chaudhary S, Khan M, Smith SJ. Concurrent Pneumocystis jirovecii and pulmonary histoplasmosis in an undiagnosed HIV patient. BMJ Case Rep. 2018;2018:bcr2017223422. https://doi.org/10.1136/bcr-2017-223422
Obmann VC, Bickel F, Hosek N, Ebner L, Huber AT, Damonti L, et al. Radiological CT patterns and distribution of invasive pulmonary Aspergillus, non-Aspergillus, Cryptococcus and Pneumocystis jirovecii mold infections - A multicenter study. Rofo. 2021;193:1304-14. https://doi.org/10.1055/a-1482-8336
Nambu A, Ozawa K, Kobayashi N, Tago M. Imaging of community-acquired pneumonia: Roles of imaging examinations, imaging diagnosis of specific pathogens and discrimination from noninfectious diseases. World J Radiol. 2014;6:779‑93. https://doi.org/10.4329/wjr.v6.i10.779
Szydlowicz M, Matos O. Pneumocystis pneumonia in the COVID-19 pandemic era: Similarities and challenges. Trends Parasitol. 2021;37:859-62. https://doi.org/10.1016/j.pt.2021.07.010
Muñoz C, Zuluaga A, Restrepo A, Tobón A, Cano LE, González A. Molecular diagnosis and detection of Pneumocystis jirovecii DHPS and DHFR genotypes in respiratory specimens from Colombian patients. Diagn Microbiol Infect Dis. 2012;72:204-13. https://doi.org/10.1016/j.diagmicrobio.2011.11.015
Chotiprasitsakul D, Pewloungsawat P, Setthaudom C, Santanirand P, Pornsuriyasak P. Performance of real-time PCR and immunofluorescence assay for diagnosis of Pneumocystis pneumonia in real-world clinical practice. PLoS ONE. 2020;15:e0244023. https://doi.org/10.1371/journal.pone.0244023
Bateman M, Oladele R, Kolls JK. Diagnosing Pneumocystis jirovecii pneumonia: A review of current methods and novel approaches. Med Mycol. 2020;58:1015-28. https://doi.org/10.1093/mmy/myaa024
Perret T, Kritikos A, Hauser PM, Guiver M, Coste AT, Jaton K, et al. Ability of quantitative PCR to discriminate Pneumocystis jirovecii pneumonia from colonization. J Med Microbiol. 2020;69:705-11. https://doi.org/10.1099/jmm.0.001190
Gupta R, Mirdha BR, Guleria R, Kumar L, Samantaray JC, Agarwal SK, et al. Diagnostic significance of nested polymerase chain reaction for sensitive detection of Pneumocystis jirovecii in respiratory clinical specimens. Diagn Microbiol Infect Dis. 2009;64:381-8. https://doi.org/10.1016/j.diagmicrobio.2009.04.008
Robert-Gangneux F, Belaz S, Revest M, Tattevin P, Jouneau S, Decaux O, et al. Diagnosis of Pneumocystis jirovecii pneumonia in immunocompromised patients by real-time PCR: A 4-year prospective study. J Clin Microbiol. 2014;52:3370-6. https://doi.org/10.1128/JCM.01480-14
Smith D, Forbes A, Gazzard B. A simple scoring system to diagnose Pneumocystis carinii pneumonia in high-risk individuals. AIDS. 1992;6:337-8.
Nascimento CR, Lima MA, de Andrada-Serpa MJ, Espindola O, Leite AC, Echevarria-Lima J. Monocytes from HTLV-1-infected patients are unable to fully mature into dendritic cells. Blood. 2011;117:489-99. https://doi.org/10.1182/blood-2010-03-272690
Wakefield AE, Pixley FJ, Banerji S, Sinclair K, Miller RF, Moxon ER, et al. Amplification of mitochondrial ribosomal RNA sequences from Pneumocystis carinii DNA of rat and human origin. Mol Biochem Parasitol. 1990;43:69-76. https://doi.org/10.1016/0166-6851(90)90131-5
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159-74.
Elvin K, Linder E. Application and staining patterns of commercial anti-Pneumocystis carinii monoclonal antibodies. J Clin Microbiol. 1993;31:2222-4. https://doi.org/10.1128/jcm.31.8.2222-2224.1993
El Hodaky ES, Ouda N, Meshref M, El Bassiouny S, El Masry M, Gamal El Din H. Comparison of different methods to diagnose Pneumocystis jirovecii pneumonia in children with haematological malignancies. Egypt J Med Microbiol. 2008;17:151-9.
Robberts FJ, Liebowitz LD, Chalkley LJ. Polymerase chain reaction detection of Pneumocystis jirovecii: Evaluation of 9 assays. Diagn Microbiol Infect Dis. 2007;58:385-92. https://doi.org/10.1016/j.diagmicrobio.2007.02.014
Beard CB, Carter JL, Keely SP, Huang L, Pieniazek NJ, Moura IN, et al. Genetic variation in Pneumocystis carinii isolates from different geographic regions: implications for transmission. Emerg Infect Dis. 2000;6:265-72. https://doi.org/10.3201/eid0603.000306
Sing A, Roggenkamp A, Autenrieth IB, Heesemann J. Pneumocystis carinii carriage in immunocompetent patients with primary pulmonary disorders as detected by single or nested PCR. J Clin Microbiol. 1999;37:3409-10. https://doi.org/10.1128/JCM.37.10.3409-3410.1999
Tang X, Bartlett MS, Smith JW, Lu JJ, Lee CH. Determination of copy number of rRNA genes in Pneumocystis carinii f. sp. hominis. J Clin Microbiol. 1998;36:2491-4. https://doi.org/10.1128/JCM.36.9.2491-2494.1998
Derechos de autor 2023 Biomédica

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























